Introduction to Forensic Toxicology John P Wetstein Toxicology


































































- Slides: 78

Introduction to Forensic Toxicology John P. Wetstein Toxicology Training Coordinator Illinois State Police Division of Forensic Services Forensic Sciences Command

Role of Forensic Toxicologist n To provide Quality analysis n Complete n Accurate n Timely n To provide interpretation of results n To testify effectively n To assist the trier of fact in understanding

Toxicology Testing n Different from clinical toxicology n Instrumentation overlap n Redundancy is increased n Speed is decreased n “Cheap, Good, or Fast – pick two”

Forensic Toxicology n Results are developed with the needs of the legal community in mind n Requires “second test “ philosophy Two tests based on a different chemical principle n One test more sensitive than the other n

Pharmacology n Pharmacokinetics – how the drug moves through the body Absorption n Distribution n Metabolism n Elimination n “What the body does to the drug”

Pharmacology n Pharmacodynamics n How the drug interacts with receptors in the brain n n What are the effects? How long does it last? “What the drug does to the body”

Ethanol Absorption n Dermal n Requires open abrasion or lesion n Elimination is faster than absorption for intact skin n Mucosal Membranes n Oral, vaginal, and rectal n Oral ingestion

Ethanol in the stomach n Only 25% of a dose is absorbed in the stomach n Ethanol is a secretagogue n More ethanol = more secretions= better barrier n 30% solution for maximal absorption n Retention in the stomach will delay absorption n At low concentration Michaelis-Menton kinetics apply

Ethanol in Small Intestine n Most absorption takes place here n Small intestine has roughly 300 square meters of surface area (over 3200 square feet!) n Colon can absorb ethanol – it usually never gets the chance

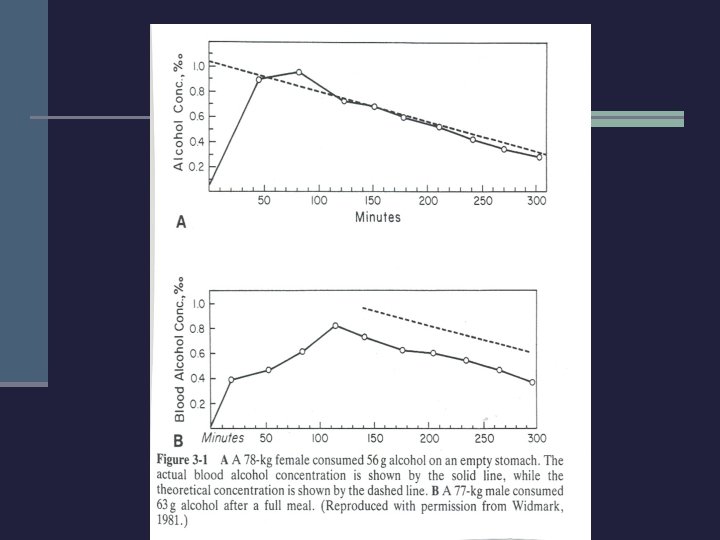
Volume of Distribution n Widmark derived the formula n n BAC = A/rp Where A is the alcohol dose R is the volume of distribution P is the body weight Widmark calculated r to be. 68 for males and. 55 for females n Later researchers found an average of. 70 and. 60 n

Algebra Refresher


Widmark Simplified r varies by individual but for males it is 0. 7 and 0. 6 for females

Using Volume of Distribution n Two significant elements can be derived n The alcohol dose n The estimated BAC n Example – How many grams of ethanol would be required to bring BAC up to. 08 g/d. L given a 220 lb. male? ?

Estimating Dose Required n Knowns: n Mass = 100 kg n Volume of distribution =. 70 n BAC =. 08 g/d. L (or. 8 g/kg) n Unknown = x grams ethanol

Algebra n 0. 7 = wbc/. 08 g/d. L n wbc =. 08 g/d. L x 0. 7 n wbc =. 056 g/d. L =. 56 g/kg n 0. 56 g/kg x 100 kg = 56 grams of ethanol n One drink = 11 grams ethanol

Estimating a BAC n Knowns n Mass = 55 kg (120 lbs) n Ethanol dose = 5 glasses of wine n Volume of distribution =. 60 n Unknown n BAC

Estimating BAC n 0. 6 = wbc / bac n 0. 6 = (55 grams etoh / 55 kg ) / bac n bac = (1 g/kg ) /0. 6 n bac = 0. 1 g /d. L / 0. 6 n bac =. 167 g / d. L

Units for the Equation n WBC must be expressed as grams/kilogram n Later converted to grams/d. L n 1 d. L weighs 0. 1 kilogram n BAC must be expressed as grams/d. L n Must convert body weight to kilograms n 1 lb =. 45 kg 1 kg = 2. 2 lb n Must know how many grams of ethanol in a standard drink. n Generally 11 grams

Practice Problem n Mary weighs 165 lbs. n What is her mass in kilograms? n She drinks four shots of whiskey. n How many grams of ethanol is that? n What is her “Whole Body Concentration? ” n What is her Rho or Volume of Distribution? n What is her maximum theoretical BAC

Ethanol Elimination n 95% of ingested ethanol is eliminated in the liver. n Dubowski reported these average rates in 1976 n n n . 015 g/d. L/hour for males, . 018 g/d. L/hour for females Both exhibited ranges of. 011 -. 022 Shajani and Dinn (1985) n n . 018 for males, . 020 for females Same ranges applied


Retrograde Extrapolation n At most alcohol concentrations, ethanol is eliminated at a steady state. n Given a known alcohol concentration, one can estimate a BAC at a previous point in time n Example: Driver at. 070 g/d. L measured 2 hours after an accident. What was BAC at the time?

Illustrative Question n If I drove 65 miles / hour for 2 hours, how far have I gone? n (2 hours) (65 miles/hour) = 130 miles n If all of my driving has been northbound on Interstate 55, and I stopped at the 235 mile marker, where did I start driving ? 235 miles – (2 hours) (65 miles/hour) n 235 – 135 = 100 n

Retrograde Extrapolation n Assumptions: n The driver is post absorptive at the times in question n 45 min to 2 hours after last drink – clinical Within 15 minutes in social situations The driver is in a “normal” state of health and metabolizes within normal ranges n n Normal ranges I use are. 010 to. 020 Alcoholics/chronic abusers. 030 +/-. 009

Retrograde Extrapolation n Driver drinks at a bar and leaves at 12: 30 am. At 1: 15, he is involved in a crash. At 3: 00 am, blood is drawn for alcohol testing. The BAC is measured at. 072 g/d. L. n What was the estimated BAC at 1: 15?

Retrograde Extrapolation n Low estimate: . 072 + (1. 75 hours *. 010 g/d. L/Hour) =. 072 +. 0175 =. 089 High Estimate: . 072 + (1. 75 *. 020 g/d. L/hour) =. 072 +. 035 =. 107

People vs Floyd n Defense won appeal n Expert assumed too many factors away n Factors listed irrelevant to metabolism n Factors never known n Not a bright line ruling against back extrapolation n Foundation question should address factors

DUI Kit Swabs n Contaminated swab defense n Ethanol and Isopropanol were detected in the iodine solution included in DUI kit n Concentrations were. 005 g/d. L n Factors to remember: n This can not increase a BAC of forensic significance – even in a worse case scenario n Both volatiles are present in equal amounts and both are readily detectable n Contamination would be evident by IPA peak

DUI Kit Swabs n Venipuncture requires drying of the site prior to needle insertion n Dubowski demonstrated contamination with wet ethanol sponge Placed directly on site n Needle under vacuum n n Defense will provide multiple references n These are legal, not scientific concerns

DUI Kit Swabs n Prior administrative rule stated “…does not contain alcohol” n Amended by emergency change in 2007 – later made permanent n Manufacturer is unable to detect this trace level n Source of the contamination is unknown

Drug Pharmacokinetics n Absorption n Route of administration n Oral, inhalation, intravenous, intranasal, intramuscular, transdermal Route effects the rapidity of onset Blood-Brain barrier § Crack vs cocaine hydrochloride

Drug Pharmacokinetics n Absorption n In order for the drug to exert action, the drug must be absorbed into the bloodstream and move through membranes to get to the appropriate receptor n Absorption is largely dictated by the chemical properties of the drug itself

Drug Pharmacokinetics n Distribution n Governed largely by solubility n n n Hydrophobic – into fats and tissues (THC) Hydrophillic – into body water Half Life is a function of solubility n n Half-Life is time taken to remove half of the concentration Hydrophillic substances are excreted quickly

Pharmacokinetics n Metabolism n Can go from active to inactive n n n THC to THC-COOH Cocaine to benzoylecgonine Can go from inactive to active n n Codeine to morphine Carisoprodal to meprobamate

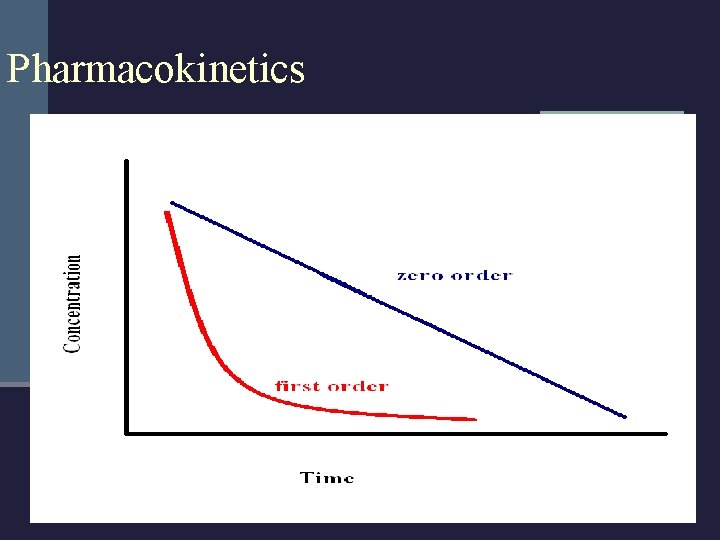
Pharmacokinetics n Elimination Process of removing the drug and or metabolites from the body. Usually by making the substance more polar and water soluble. Zero Order Kinetics – steady (ethanol) First Order Kinetics – not linear; uses half lives

Pharmacokinetics

Pharmacokinetics n With anything other than linear elimination, it is generally not possible to back extrapolate n Baseball n Maple Leaf

With the exception of ethanol, there is so far no widely accepted correlation between the drug concentration in blood and a corresponding level of driving impairment among the scientific community. What is more, factors such as tolerance can have a profound effect on the pharmacodynamic response in an individual. Sarah Kerrigan, Ph. D Drug Toxicology for Prosecutors American Prosecutors Research Institute

Driving and Impairment n Drugs can introduce impairment in the following ways Coordination n Judgment / Decision Making n Perception n Tracking n Reaction Time n Divided Attention / Multi-Tasking n

Driving and Impaiment n Coordination n Have to put hand foot movements together at appropriate times to effectively control the car n Drugs effect nerves and muscles n Once a decision is made to put into the vehicle, the body needs to respond at the right time and to the appropriate degree

Driving and Impairment n Judgment and Decision Making n Driving requires the ability to concentrate and assess risk appropriately n Stimulants may give a false sense of invincibility and induce riskier behavior

Driving and Impairment n Perception n Visual acumen can be diminished n n Blurred vision Ability to accurately determine distances and subtle changes in texture n Tracking n Required to maintain vehicle position on the roadway n May manifest in weaving

Driving and Impairment n Divided Attention and Multi-Tasking n Multiple visual cues that demand attention n n Road signs, other cars, pedestrians, changes in road surface Multiple activities that require attention n Gas, brake, steering input § Depressant drugs retard ability § Stimulant drugs may cause hypervigilance

Interpretation Issues n What does it mean? n A toxicology result by itself will not normally be sufficient to make a statement about impairment n A toxicologist can testify to the expected or anticipated effect of a particular drug n Can not state with complete certainty that all drivers would be impaired given a drug and/or concentration

Interpretation Issues n How long can it be detected? n Detection time n n n Dose Route of administration Elimination Rate Presence in urine may not be reliable indicator of recent usage n Metabolism from blood can be very rapid n

Drug Classes (functional) n Stimulants n Amphetamines, cocaine, MDMA n Depressants n Benzodiazepines, barbiturates, anti-convulsants, sleep inducers n Opiates n Oxy, Hydrocodone, codeine, morphine, Heroin n Hallucinogens n GHB, THC, LSD n Serotonin re-uptake inhibitors (SSRIs) n Prozac, paxil, lexapro, zoloft

Typical Tests n Immunoassays n Lack specificity n React to classes of drugs § Cocaine, THC, Amphetamines, Barbiturates, Benzodiazepines, PCP, Opiates n Individual drugs within class behave differently § Alprazolam vs Lorazepam n Can be used on blood or urine


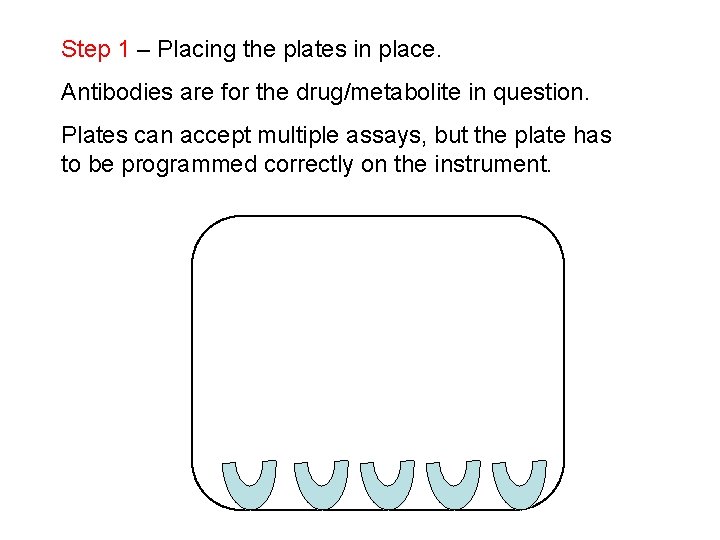
Step 1 – Placing the plates in place. Antibodies are for the drug/metabolite in question. Plates can accept multiple assays, but the plate has to be programmed correctly on the instrument.

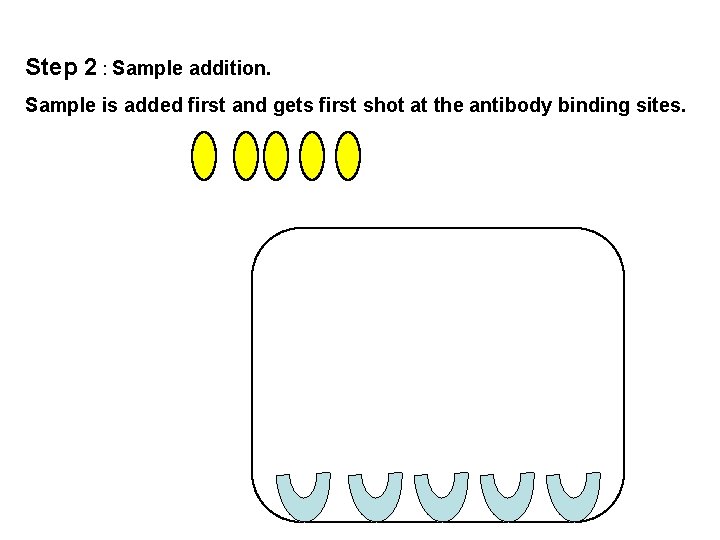
Step 2 : Sample addition. Sample is added first and gets first shot at the antibody binding sites.

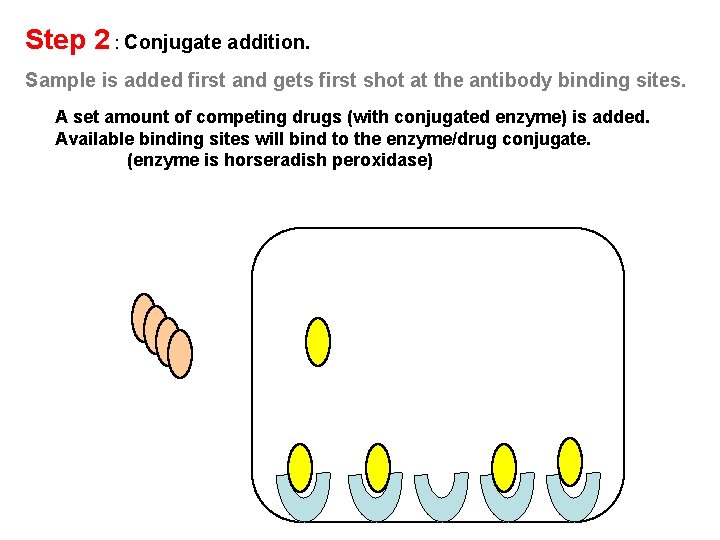
Step 2 : Conjugate addition. Sample is added first and gets first shot at the antibody binding sites. A set amount of competing drugs (with conjugated enzyme) is added. Available binding sites will bind to the enzyme/drug conjugate. (enzyme is horseradish peroxidase)

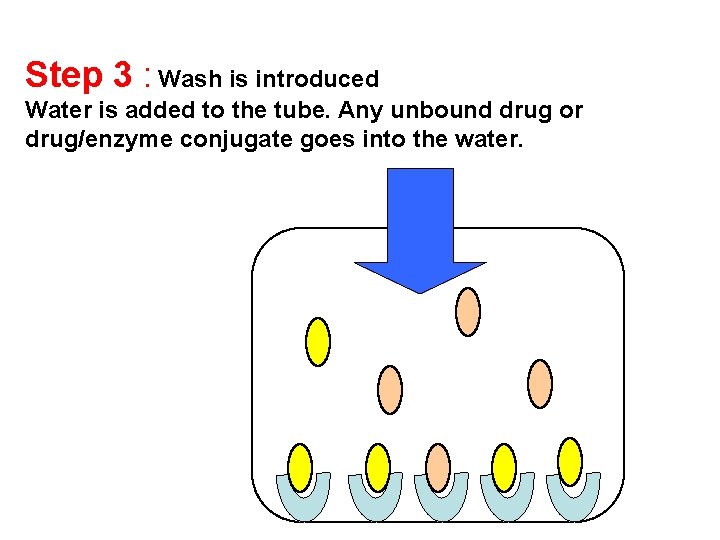
Step 3 : Wash is introduced Water is added to the tube. Any unbound drug or drug/enzyme conjugate goes into the water.

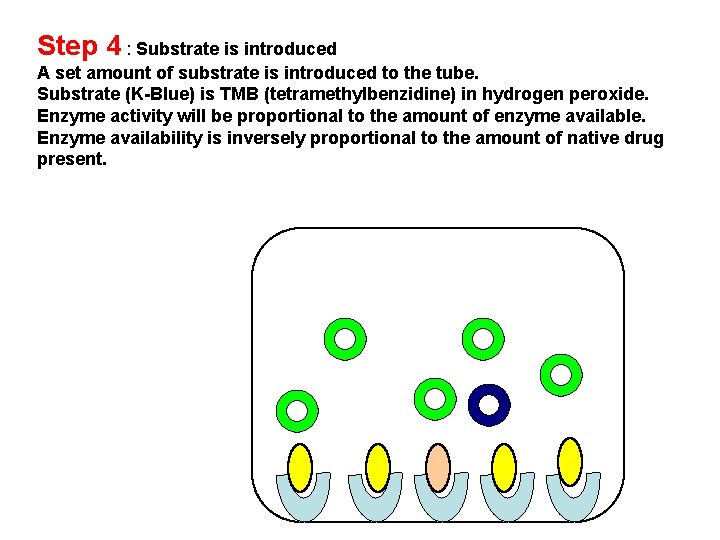
Step 4 : Substrate is introduced A set amount of substrate is introduced to the tube. Substrate (K-Blue) is TMB (tetramethylbenzidine) in hydrogen peroxide. Enzyme activity will be proportional to the amount of enzyme available. Enzyme availability is inversely proportional to the amount of native drug present.

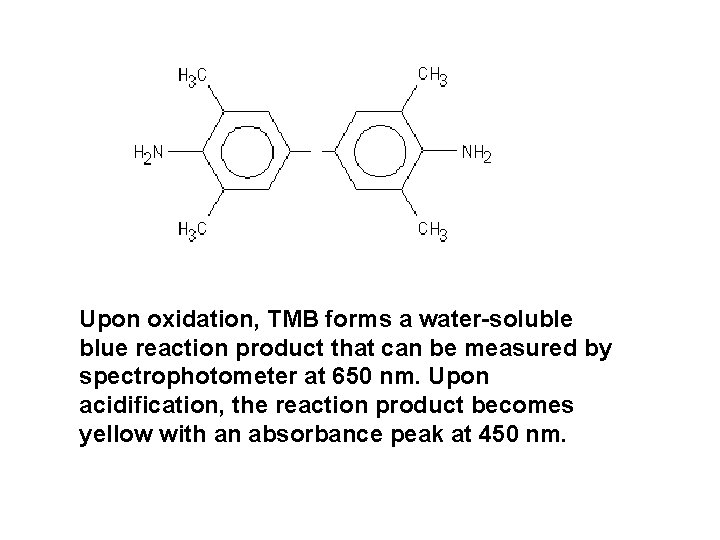
Upon oxidation, TMB forms a water-soluble blue reaction product that can be measured by spectrophotometer at 650 nm. Upon acidification, the reaction product becomes yellow with an absorbance peak at 450 nm.

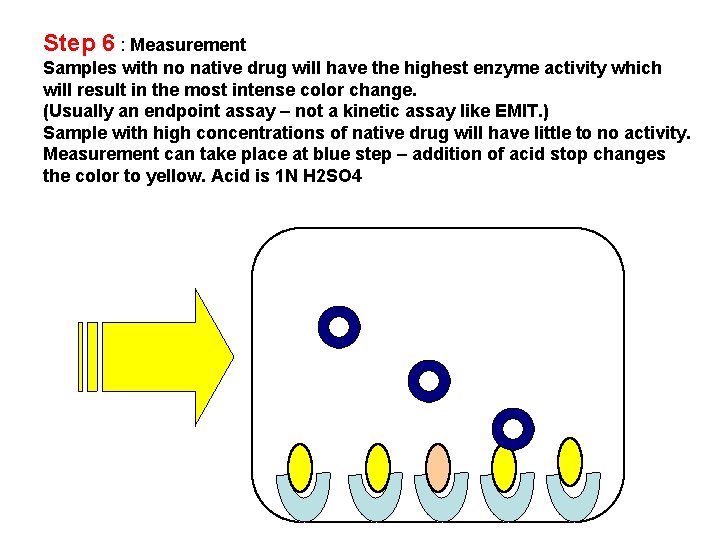
Step 6 : Measurement Samples with no native drug will have the highest enzyme activity which will result in the most intense color change. (Usually an endpoint assay – not a kinetic assay like EMIT. ) Sample with high concentrations of native drug will have little to no activity. Measurement can take place at blue step – addition of acid stop changes the color to yellow. Acid is 1 N H 2 SO 4

Chromatography n Separation of a mixture of compounds into individual components n Dependant on the interaction of a drug with a stationary and a mobile phase n Analogy: shoppers vs non-shoppers in a mall n Two basic types of mobile phase n Liquid (LC) n Gas (GC) n Silica (Thin Layer)

Chromatography n What causes the interactions? n Polarity of the drug n “like dissolves like” Structural components in the drug n With GC, the ability to be in a gaseous state n n n Takes heat to make steam With LC, the relative affinity for the mobile or stationary phase

Detectors n Once the individual compounds are separated, we have to have a way to count how much is there n The detector will plot the amount as a Peak on a chromatogram n The higher the peak, the more of that compound is in the sample


Types of detectors n Spectrophotometric n Based on the absorbance of light (1000 ug/L) n Flame ionization n Ability to excite a flame (500 ug/L) n Nitrogen/Phosphorous n Specific for elements commonly found in drug molecules (100 ug/L) n Mass Spectrometer n Provides a “fingerprint” of the molecule by looking at how the molecule breaks under stress (10 ug/L)

Measurement Uncertainty n LUDINGTON, Mich. — Blood samples tested by the Michigan State Police crime lab in drunken-driving cases are unreliable because the results don’t carry a margin of error, a judge said. District Court Judge Peter Wadel threw out blood evidence against a man in northern Michigan’s Mason County. His decision is not binding on similar cases in other state courts but it’s certain to get attention.

Measurement Uncertainty n Cited in 2009 NAS Report on Forensic Science n All testing procedures have some degree of imprecision n Measurement uncertainty is a way of expressing that imprecision

Example of Measurement Uncertainty: Bathroom Scale n 150 pound male n n n 150 152 151 148 149 n 150 pound male n n n 125 144 151 170 160 Both scales give the same average value, but one does it with more precision

Example of Measurement Uncertainty: Bathroom Scale n The man weighs 150 +/- 3. 2 lbs Uncertainty is reported when: the agency asks a compliance limit is bracketed interpretation is affected +/- 34 lbs.

Reporting Uncertainty n Blood Ethanol. 082 g/d. L “The estimated uncertainty for ethanol analysis is 3. 0% at the 95% confidence interval, resulting in an ethanol range of. 079 to. 084 g/d. L. ”

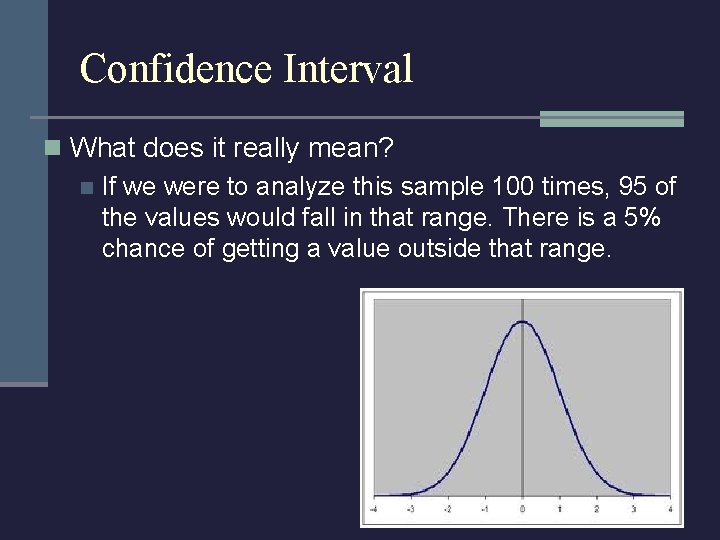
Confidence Interval n A confidence interval gives an estimated range of values which is likely to include an unknown population parameter, the estimated range being calculated from a given set of sample data. (Definition taken from Valerie J. Easton and John H. Mc. Coll's Statistics Glossary v 1. 1)

Confidence Interval n What does it really mean? n If we were to analyze this sample 100 times, 95 of the values would fall in that range. There is a 5% chance of getting a value outside that range.

Example of Measurement Uncertainty: Bathroom Scale n The man weighs 150 +/- 3. 2 lbs +/- 34 lbs. (146. 8 – 153. 2) (116 – 184) Observed range 148 -152 Observed range 125 -170

How is uncertainty estimated? n In toxicology, large data set of controls which are analyzed like case sample n An imprecision is tracked and acceptability limits are established n Imprecision of controls is applied to case samples

Measurement Uncertainty n Variety of methods exist n Some are rigorous mathematical n Others are more empirical (our approach) n n Historical familiarity Passes the jury “common sense” test Defense may pick from journal articles n No direct guidance is available n

The alphabet soup n GC/FID n Alcohols, drugs with relatively high dosages n GC/NPD n Drugs with nitrogen and phosphorous n GC/MS n Most drugs except those that break down under high temperatures n LC/MS n Wide variety of drugs (ID is complicated)

Specimen Types n Blood, serum or plasma n Used for quantitation n Used to interpret therapeutic drug levels n Urine n Used for qualitative identification n Not useful for quantitation since no contact with nervous system

Urine Formation n Urine is a product of blood being filtered by the kidney Drugs and metabolites and toxins are removed from the blood and brought to the bladder for excretion n Filtering and pooling tends to concentrate drugs and metabolites into an aqueous (waterlike) solution n n Urine is the ideal fluid for identifying drugs

Blood n Has direct contact with the central nervous system, and is therefore the most relevant for drug quantitation. n Drug concentrations in blood will significantly less than in urine Factor of 10, 100 or possibly 1000 n Drug may be below detection limit in blood yet still found in urine n n Poor sample for drug screening

Other Sample Types n Restricted to post-mortem analysis n Vitreous Humor : eye fluid n Liver n Brain n Bile

Testing Protocol n Typical DUI case with Blood and Urine n Blood gets tested for alcohol n n Report if >0. 08. No further testing. Urine gets immunoassay for THC and Cocaine metabolites. Confirm and report. n If negative expand immunoassays. Confirm and report. GC/MS drug screen if immunoassay screen is negative n Agency can always request more analysis n

Testing Protocol n Typical Aggravated DUI case with Blood and Urine Blood gets tested for alcohol n Urine gets immunoassay for PCP, Opiates, Barbiturates, Benzodiazepines, Amphetamines, THC and Cocaine n GC/MS drug screen n Drugs identified are quantitated in blood as necessary by request n