Radioactivity Radioactivity l When an atom of an















- Slides: 16

Radioactivity

Radioactivity l When an atom of an element has an unstable nucleus, the nucleus can disintegrate, forming subatomic particles and releasing large amounts of energy. l This release is called radioactivity l Alpha and beta particles may be released

Alpha Particles l An alpha particle is a helium nucleus l 2 protons l 2 neutrons l Symbol is α l Mass is 4 amu l Charge is +2

Beta Particles l. A beta particle is an electron l Symbol is β l Mass is insignificant 0. 0006 amu l Charge is -1

Nuclear Reactions During nuclear reactions, an unstable element will transform or decay into another element with a lower mass l In this reaction, an alpha particle has been released l This is called an alpha decay reaction l

Nuclear Reactions In the second step of this reaction, an electron is released l This is a beta decay nuclear reaction l It takes 12 more decay reactions to reach a stable nuclear structure l

Gamma Rays l Gamma rays are powerful electromagnetic waves that are emitted when a nucleus disintegrates l They do not transform an unstable element into another element or alter its mass

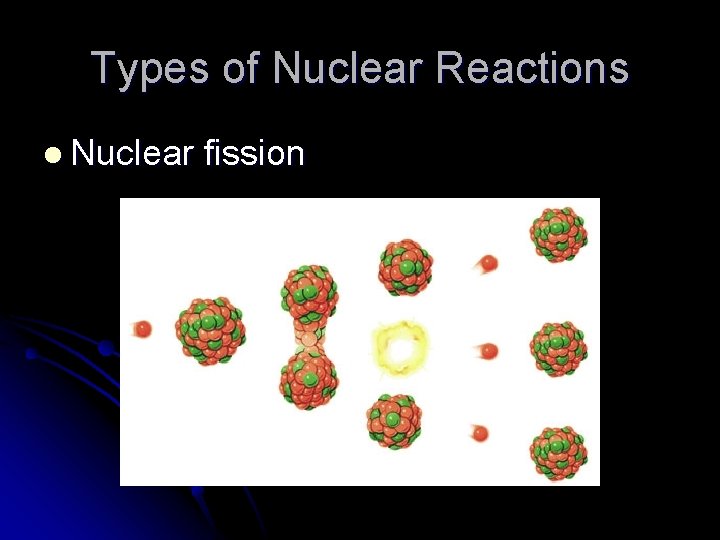
Types of Nuclear Reactions l Nuclear fission l The splitting of a nucleus into two smaller nuclei with a tremendous release of energy l The energy comes from the mass lost when the nucleus is split l Often, neutrons are ejected when the nucleus is split

Types of Nuclear Reactions l Nuclear fission

Types of Nuclear Reactions l Nuclear fusion l The combining of two smaller nuclei to form a larger single nucleus with a tremendous release of energy – this energy also comes from lost mass l Fusion requires a huge amount of energy and pressure l Occurs naturally in stars like the sun

Types of Nuclear Reactions l Nuclear fusion

Pros of Nuclear Technology l Used in medicine, weapons and electricity production l Medicine – cancer treatments and diagnostic tests l Electricity – nuclear power plants l. Produce large amounts of energy l. Little air pollutio

Cons of Nuclear Technology l Cooling of electrical plants can result in thermal pollution l Radioactive leaks possible l Fukushima, Japan l Three Mile Island l Dangerous waste must be stored for long periods of time

Half-life l Radioactive materials decay in a predictable way l The rate of decay is called the halflife of a substance l The half-life is the amount of time it takes half of the substance to decay l Different substances have different half-lives

Half-life l For carbon-14 (C-14), the half life is 5370 years l If you start with 1000 g of C-14, in 5370 years, you will have only 500 g left (half has decayed) l After another 5370 years, you will have 250 g (half of the 500 g)

Half-life Activity l We are going to use Skittles to simulate what happens with atoms of radioactive elements