Pharmacovigilance Programme Of India Dr Reshmi TR Pharmacovigilance



















- Slides: 23

Pharmacovigilance Programme Of India Dr. Reshmi. TR

Pharmacovigilance Pharmakon Drug, Vigilare to be observant Continuous monitoring for unwanted effects of marketed drugs • WHO definition • Science and activities relating to Detection, Assessment, Understanding , Prevention of adverse effects and any other drug related problems

History • 10 th century Salerno Medical School • “whosoever shall sell any noxious drug shall be hanged” 18 th century William Withering 1 st systematic paper in medicinal drug with detailed description of ADR associated with Digitalis

20 th century Thalidomide disaster

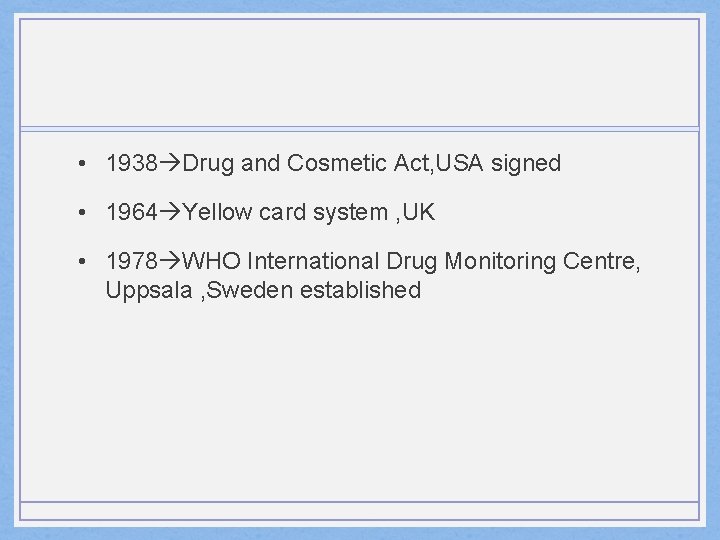
• 1938 Drug and Cosmetic Act, USA signed • 1964 Yellow card system , UK • 1978 WHO International Drug Monitoring Centre, Uppsala , Sweden established

WHO-UMC • Suggested guidelines for setting up Pharmacovigilance centre in every country • Uppsala Monitoring System , Sweden manages international drug monitoring

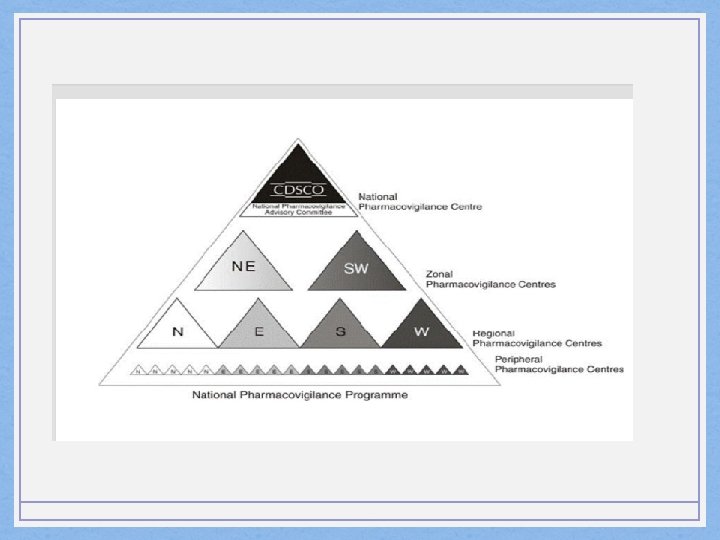
PVPI • Pharmacovigilance programme of India • July, 2010 at AIIMS, Delhi • April, 2011 shifted to IPC, Ghaziabad • Forward data to WHO-UMC through software Vigiflow

Mission Ensure that benefit outweigh risk

Objective • To create nation wide system for safety reporting • Analyse risk benefit ratio • Promote rational use of medicines

Goal • Expansion of Pharmacovigilance programme to all healthcare institutions • Develop reporting culture

Activities involved in Pharmacovigilance • Post marketing surveillance • Periodic Safety Update Report • During the first 4 years of marketing a new drug the manufacturer has to submit PSUR every 6 months for first 2 years and then once annually for the next 2 years • Dissemination of ADR data drug alert • Changes in labelling of medicines statutary warning /withdrawal of drug

Reporting Øwww. ipc. gov. in Øpvpi. ipcindia@gmail. com Ø 1800 -180 -3024 (9 am to 5. 30 pm)

Types of Reporting • Active Surveillance • Passive Surveillance • Suspected adverse drug reaction reporting form



Causality assessment • Structured and standardized assessment of ADRs as reported through ICSR (Individual Case Safety Report ) for the likelihood of involvement of suspected drug in causing particular event in a given patient.

• Causality is assessed based on: • Temporal relationship Time sequence of event relation to drug administration • Previous knowledge Whether drug produced the event in earlier cases • Dechallenge: Event subsided on stopping the drug • Rechallenge: Event reappeared on readminstration of the drug


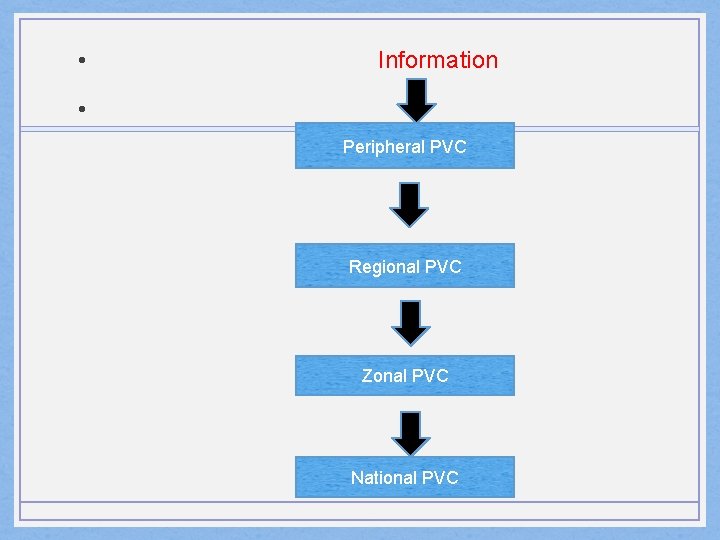
• Information • Peripheral PVC Regional PVC Zonal PVC National PVC


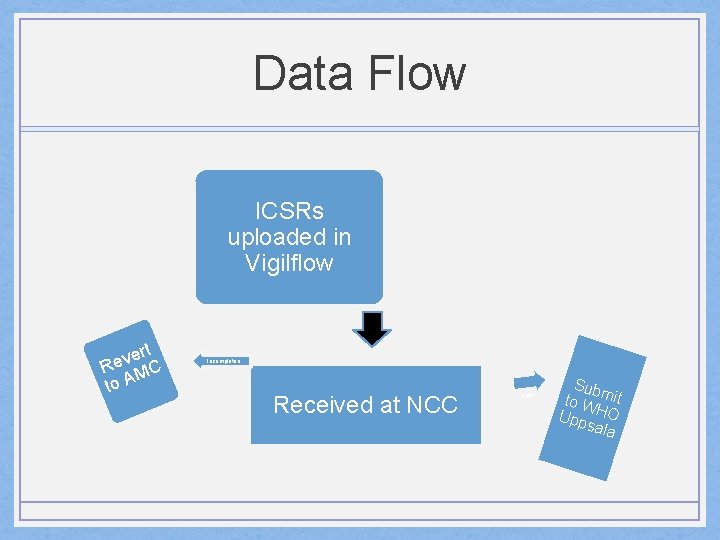
Data Flow ICSRs uploaded in Vigilflow Incompletee Received at NCC valid rt e v Re MC to A Sub to W mit Upp HO sala

Utilisation of Data • Risk management • Drug regulation • Education
