Author Netherlands Pharmacovigilance Centre Lareb Version date 14

Author: Netherlands Pharmacovigilance Centre Lareb Version date: 14 Nov 2017 Content: This lecture describes Clinical Pharmacological aspects of ADRS, by R. van Eekeren, Pharm. D, The Netherlands. This lecture can be used for teaching key aspect 2, 3, 4: preventing, recognizing, managing ADRs.

Clinical pharmacology of adverse drug reactions Course Pharmacovigilance 2017 Drs R (Rike) van Eekeren Pharmacist

Learning objectives • In this lecture, you learn to - apply the pathophysiologic principles that underlie to the occurrence of side effects • Characterize adverse drug reactions based different formats: - type A/B classification - Do. TS classification • Discuss which patients have an increased risk of developing side effects

Dosis sola facit venenum “All substances are poisons; there is none which is not a poison. The right dose differentiates a poison and a remedy” Paracelsus (1493 -1541)
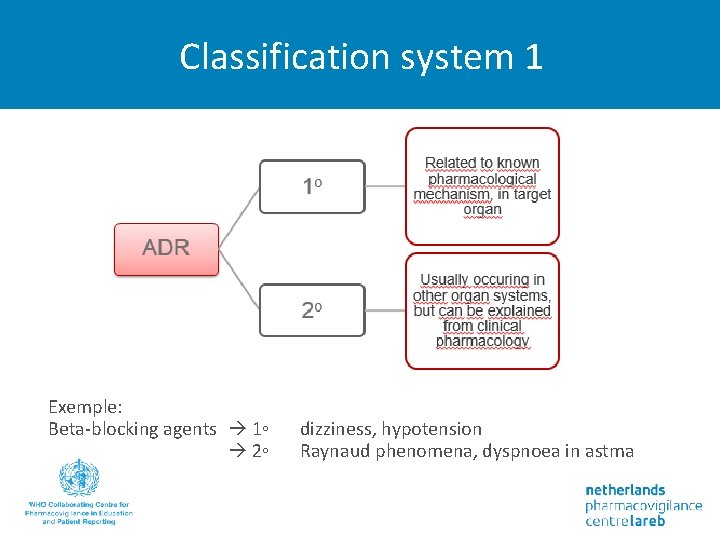
Classification system 1 Exemple: Beta-blocking agents 1◦ 2◦ dizziness, hypotension Raynaud phenomena, dyspnoea in astma
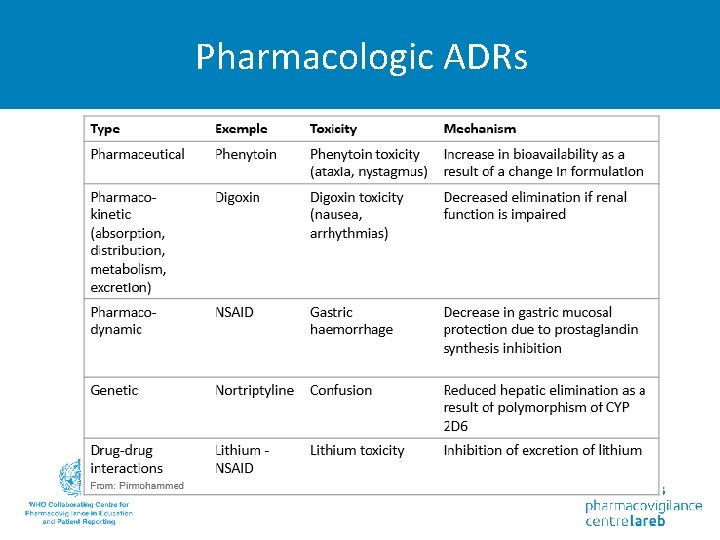
Pharmacologic ADRs

Classification system 2: Type A/B A “Augmented” B “Bizarre” • Pharmacological effect • Idiosyncratic • Dose dependent • Rare • Occurs frequently • Serious • Often detected in clinical trials • Not detected in clinical trials Talbot J, Waller P. Stephen’s detection of new adverse drug reactions. 5 e editie, 2004. John Wiley &Sons Ltd. Chichester. P. 92
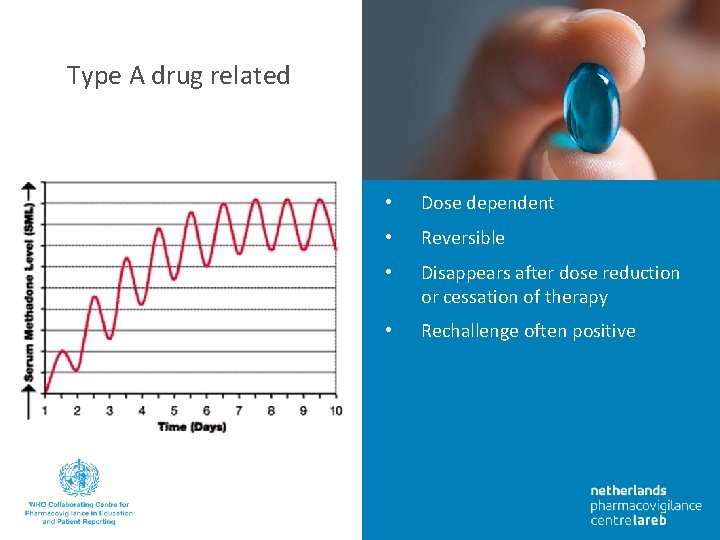
Type A drug related • Dose dependent • Reversible • Disappears after dose reduction or cessation of therapy • Rechallenge often positive
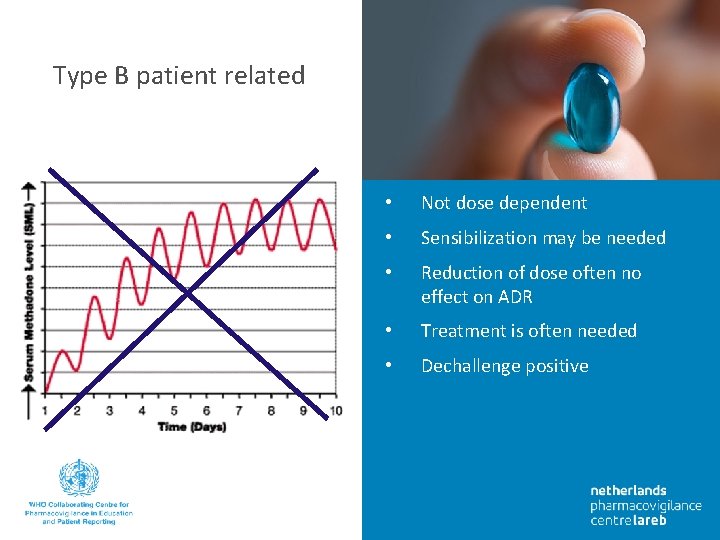
Type B patient related • Not dose dependent • Sensibilization may be needed • Reduction of dose often no effect on ADR • Treatment is often needed • Dechallenge positive

Type B patient related • Allergic / immunological reactions Examples: Blood: serious blood disorders Liver: hepatitis Kidney: nephritis Lung: pneumonia, alveolitis Skin: urticaria, angioedema, anaphylaxis • Other patient-related factors - Idiosyncrasy (undefined susceptibility) - Genetic factors (enzyme deficiencies, HLA-types)

AB types: No sharp distinction? When we do not know the exact mechanism: - Nausea in antibiotics type A, but unknown pharmacology? When the immune systems is triggered by toxicity mechanism - Toxic hepatitis Often, auto-immune component after initial hepatocellular damage In pseudo-allergy: allergic-like symptoms due to pharmacological effect - ACE-inhibitor induced angio-edema Time relationship? Patient susceptibility? ADR’s after chronic use… Why some patients develop ADR
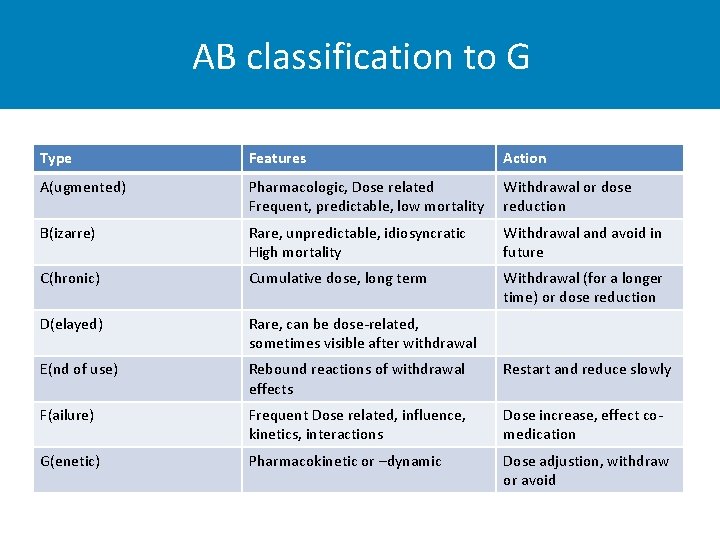
AB classification to G Type Features Action A(ugmented) Pharmacologic, Dose related Frequent, predictable, low mortality Withdrawal or dose reduction B(izarre) Rare, unpredictable, idiosyncratic High mortality Withdrawal and avoid in future C(hronic) Cumulative dose, long term Withdrawal (for a longer time) or dose reduction D(elayed) Rare, can be dose-related, sometimes visible after withdrawal E(nd of use) Rebound reactions of withdrawal effects Restart and reduce slowly F(ailure) Frequent Dose related, influence, kinetics, interactions Dose increase, effect comedication G(enetic) Pharmacokinetic or –dynamic Dose adjustion, withdraw or avoid

Classification ADRs • Text Mydriasis by amitriptyline • Text Type? A(ugmented)
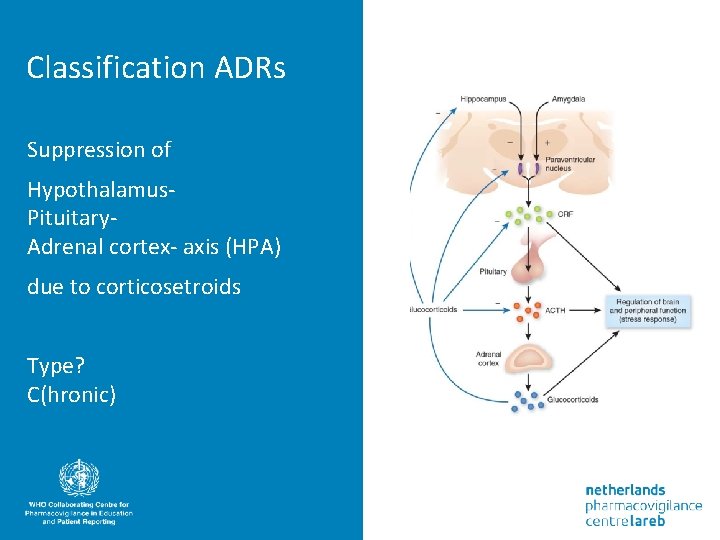
Classification ADRs Suppression of Hypothalamus. Pituitary. Adrenal cortex- axis (HPA) due to corticosetroids Type? C(hronic)

Classification system 3: Do. TS • Do Dose relatedness • T Time relatedness • S Susceptibility

Dose relatedness • Effects of medicines are based on interactions between chemical substances, to which law of mass action is applicable • Even for immunological disorders like - Hay Fever! - Desensitisation - Allergic skin reactions
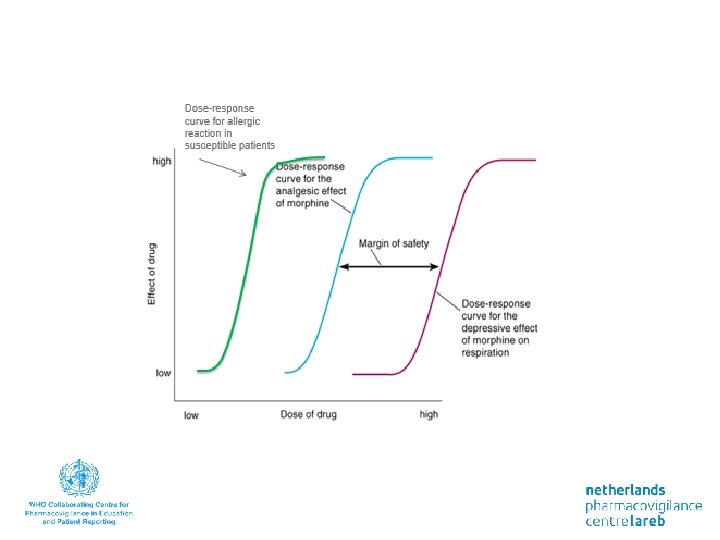

Dose relatedness Alternative classification relates to reactions that occur in • Supra-therapeutic doses (toxic dose) • Standard therapeutic doses (collateral effects) • Subtherapeutic doses (allergic reactions)

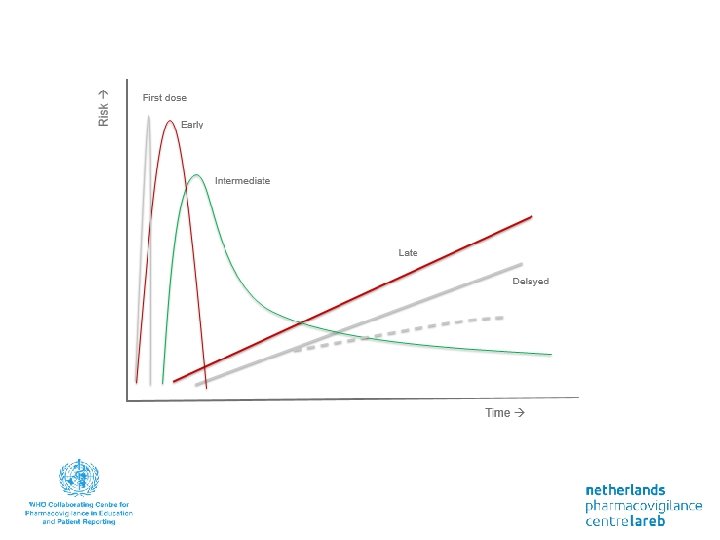
Time relatedness Time-independent reactions can occur at any time; independent of the duration of the treatment treat the cause if possible • Change in concentration at site of action - Dose - Renal function - Drug interaction (kinetic) -… • When pharmacological response alters due to other causes - Comorbidity - Ageing - Drug interaction -…

Timing • Time dependent reactions 1. Rapid reactions only when a drug is administered rapidly - Red man syndrome on vancomycin - administer slowly 2. First dose reaction. In particular, at first dose, not necessarily therafter - Hypotension ACE inhibitors, alfa-1 -blockers precautions for 1 st dose - Type I allergic reactions avoid re-exposure

Timing 3. Early reactions - early in treatment but then disappears through adaptation (tolerance) - Example: nitrate-dependent headache predictable, monitor, reassurance of patient 4. Intermediate reaction. Risk increases at first (days to month) and than diminishes - Hypersensitivity reactions II-IV - Venous thrombo embolism – antipsychotic drugs monitoring not needed after risk period withdraw drug if reaction develops susceptible population

Timing 5. Late reaction. The risk of the onset of the side effect increases with time - Osteoporosis in use of corticosteroids - Tardive dyskinesia at dopamine antagonists asses base-line function, monitor during treatment - Withdrawal reactions (like myocardial infarction after discontinuation of ß-blockers) withdraw slowly 6. Delayed reaction - Especially after prolonged use or repeated exposure - Increased risk of breast carcinoma after the use of estrogens in menopause avoud drug of screen; counseling patient

Individual susceptibility Risks of an adverse reaction depends on various factors such as • Age • Seks • Genetic factors • Renal function • Hepatic function • Exogenous factors
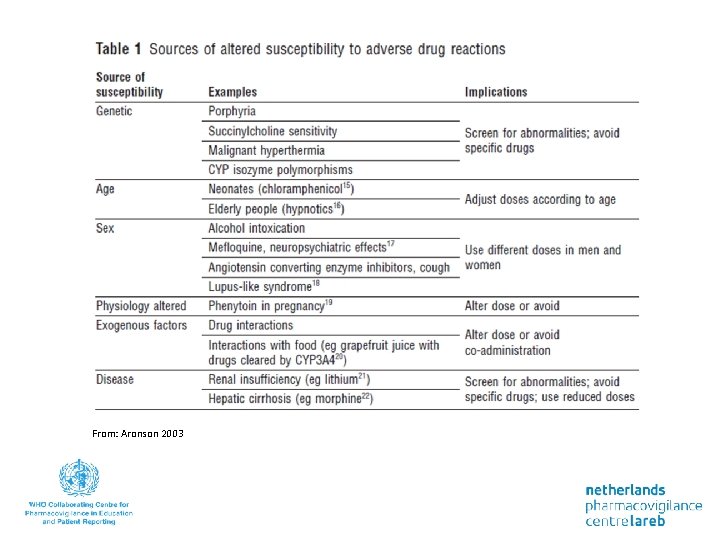
From: Aronson 2003

Children
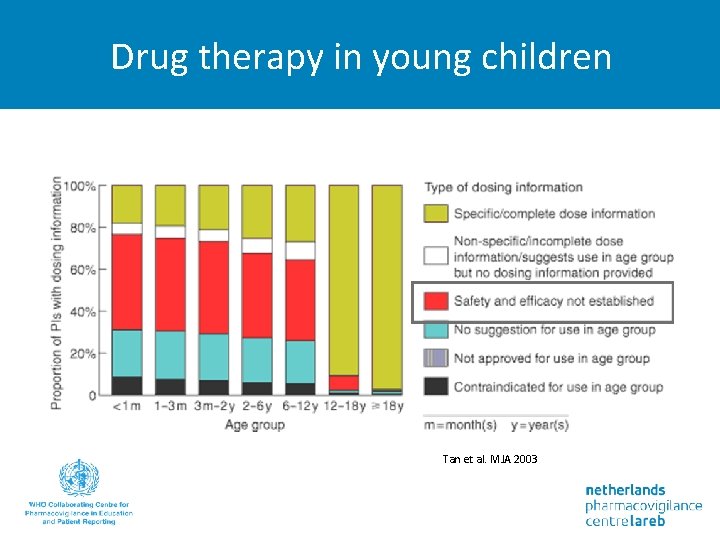
Drug therapy in young children Tan et al. MJA 2003

Adverse drug reactions in children • Different patterns compared to adults • Sometimes higher frequency - Liver failure and valproate - Stevens Johnson Syndrome and lamotrigine • Alterations in pharmacokinetics and pharmacodynamics
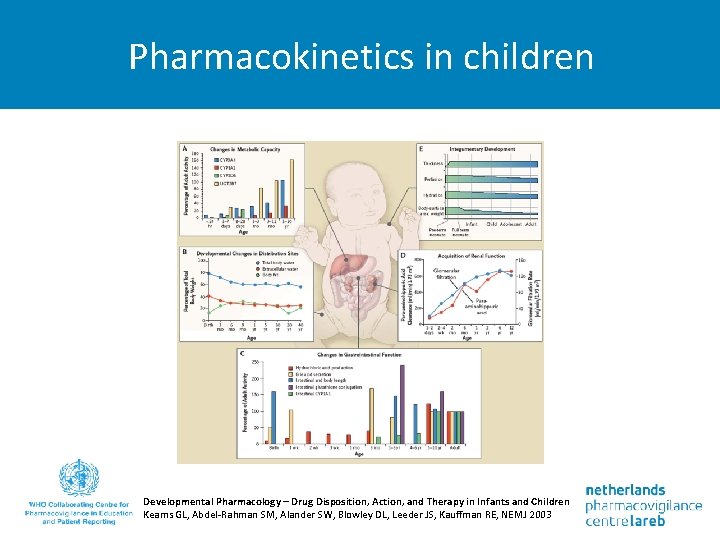
Pharmacokinetics in children Developmental Pharmacology – Drug Disposition, Action, and Therapy in Infants and Children Keams GL, Abdel-Rahman SM, Alander SW, Blowley DL, Leeder JS, Kauffman RE, NEMJ 2003
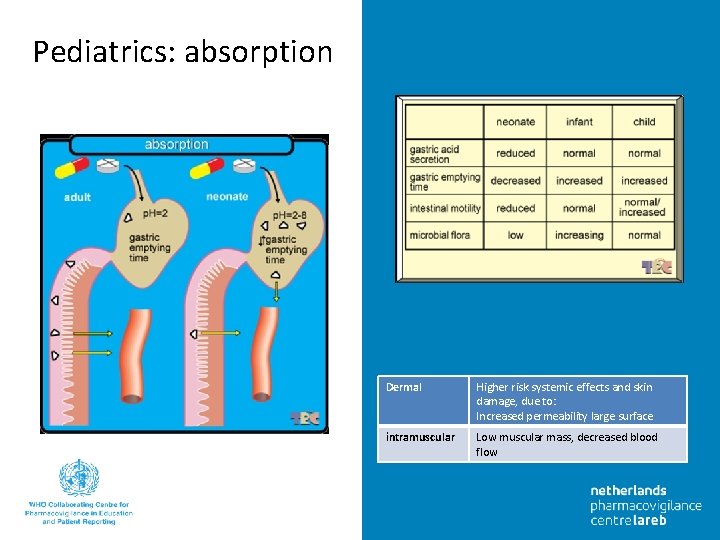
Pediatrics: absorption Dermal Higher risk systemic effects and skin damage, due to: Increased permeability large surface intramuscular Low muscular mass, decreased blood flow
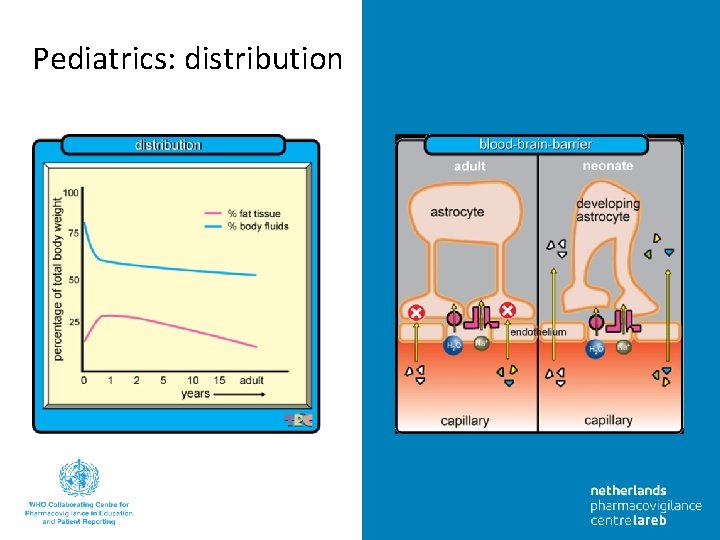
Pediatrics: distribution
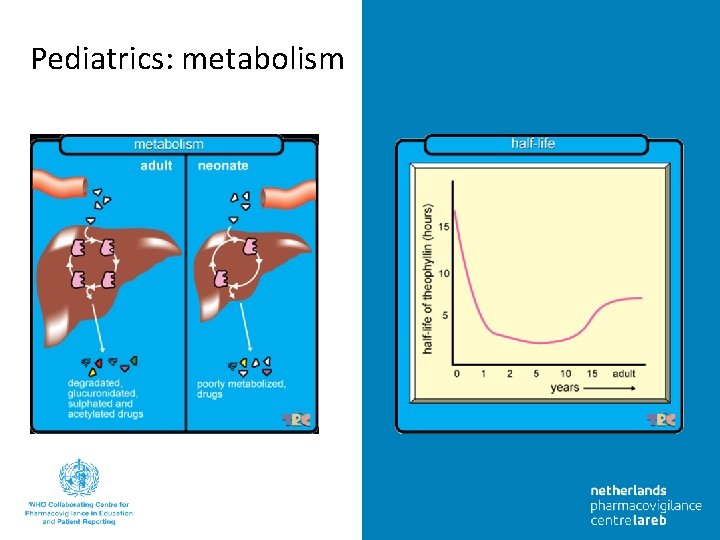
Pediatrics: metabolism
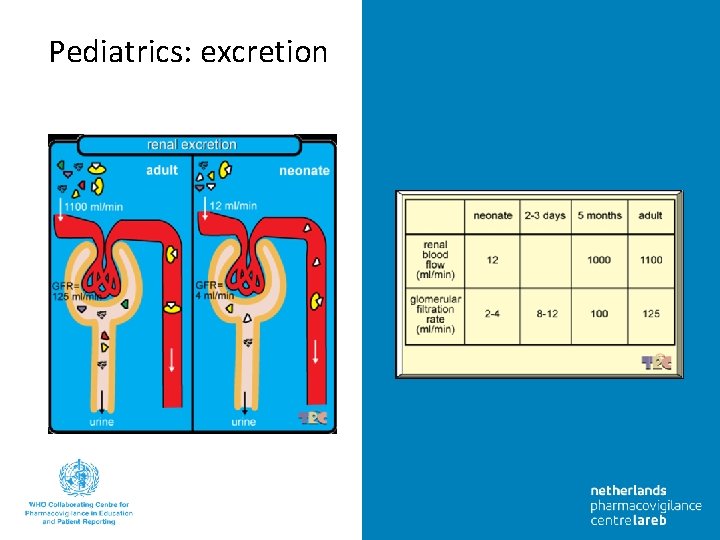
Pediatrics: excretion

Pharmacodynamics • Increased or decreased sensitivity • Antihistamines: central stimulating effects • Benzodiazepines: paradoxical effects • Ketotifen: aggressive/hyperactive behaviour • Cyclosporin: High immunosuppressive response • Effects of growth and development

Elderly

Drug therapy in the elderly • • Aging process differs among individuals Aging of various tissues may differ (within individuals) Multiple morbidity Polypharmacy (interactions) Long term use of drugs Compliance Practical problems
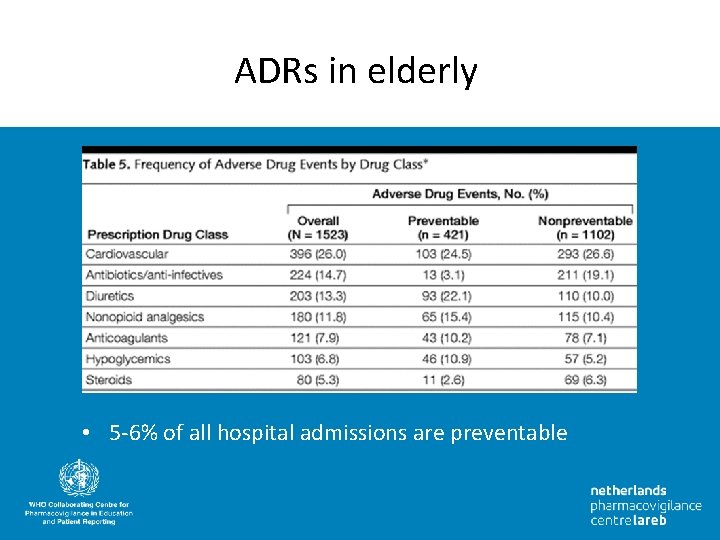
ADRs in elderly • 5 -6% of all hospital admissions are preventable
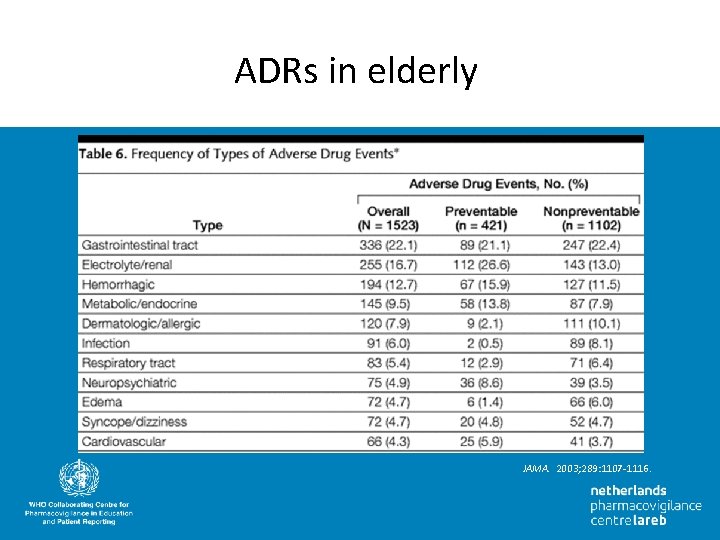
ADRs in elderly JAMA. 2003; 289: 1107 -1116.
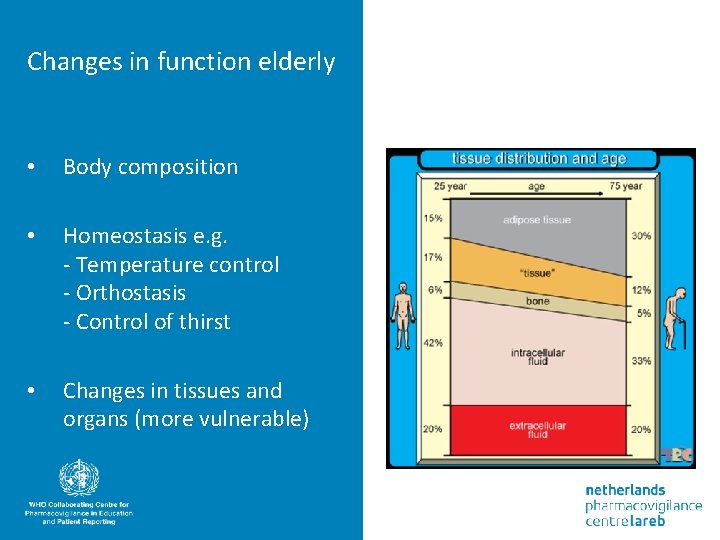
Changes in function elderly • Body composition • Homeostasis e. g. - Temperature control - Orthostasis - Control of thirst • Changes in tissues and organs (more vulnerable)
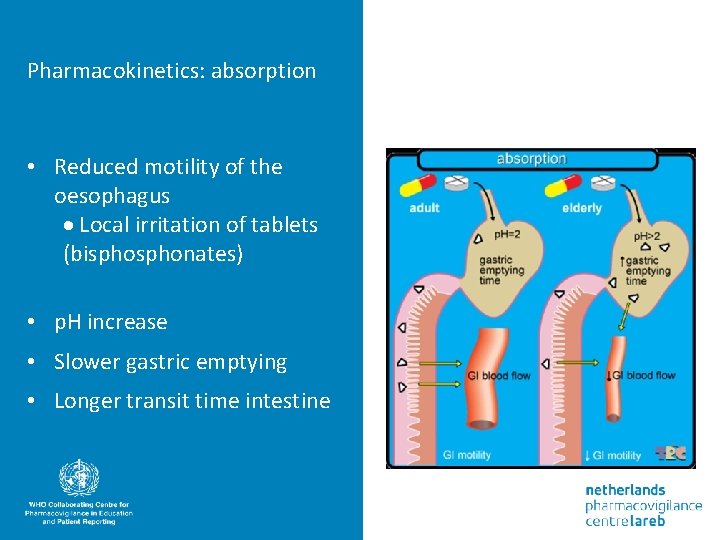
Pharmacokinetics: absorption • Reduced motility of the oesophagus Local irritation of tablets (bisphonates) • p. H increase • Slower gastric emptying • Longer transit time intestine
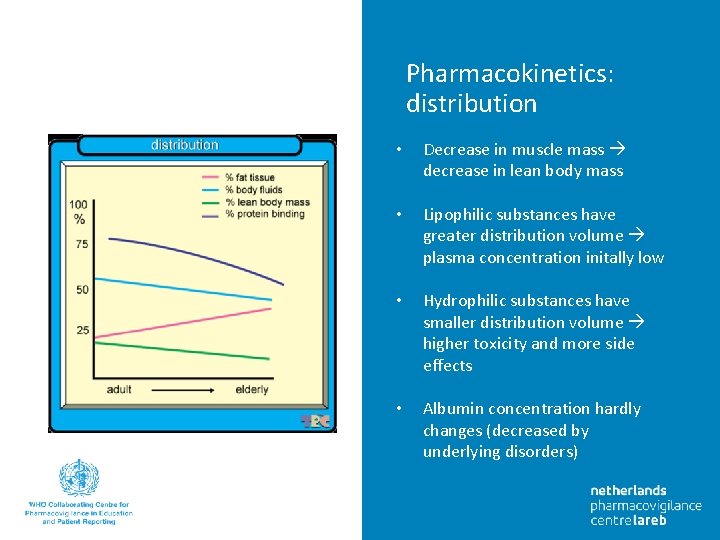
Pharmacokinetics: distribution • Decrease in muscle mass decrease in lean body mass • Lipophilic substances have greater distribution volume plasma concentration initally low • Hydrophilic substances have smaller distribution volume higher toxicity and more side effects • Albumin concentration hardly changes (decreased by underlying disorders)
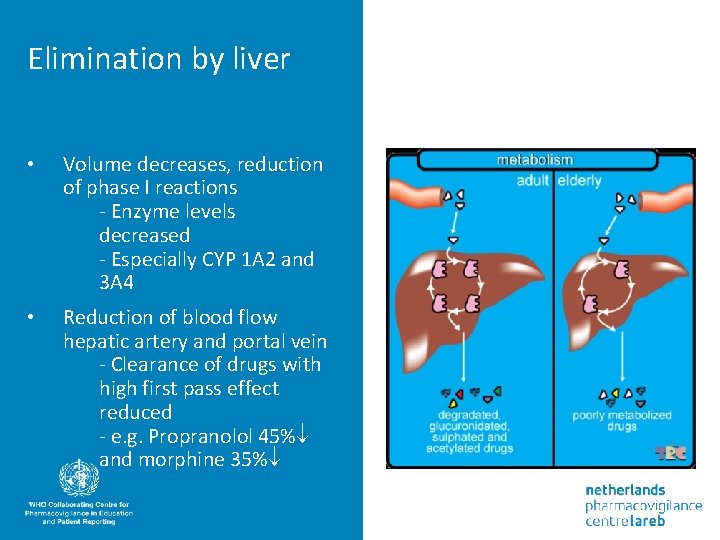
Elimination by liver • Volume decreases, reduction of phase I reactions - Enzyme levels decreased - Especially CYP 1 A 2 and 3 A 4 • Reduction of blood flow hepatic artery and portal vein - Clearance of drugs with high first pass effect reduced - e. g. Propranolol 45% and morphine 35%
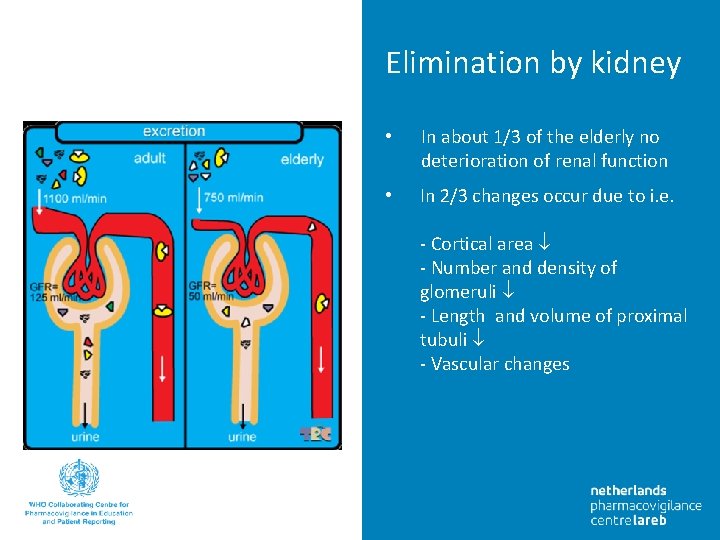
Elimination by kidney • In about 1/3 of the elderly no deterioration of renal function • In 2/3 changes occur due to i. e. - Cortical area - Number and density of glomeruli - Length and volume of proximal tubuli - Vascular changes
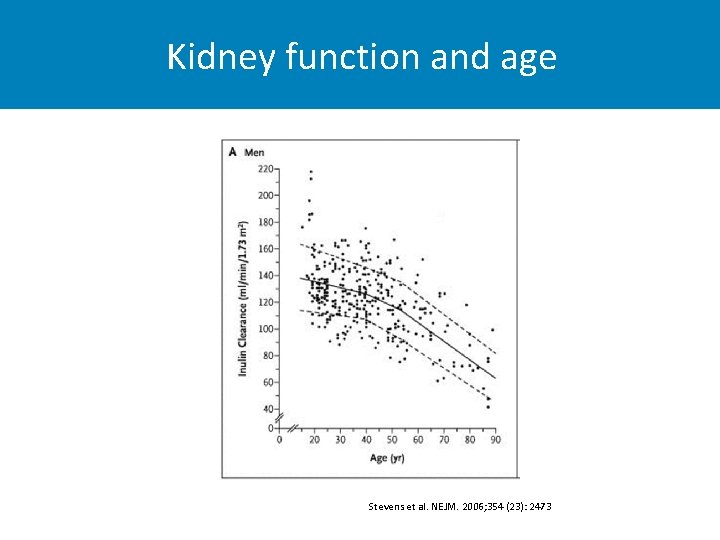
Kidney function and age Stevens et al. NEJM. 2006; 354 (23): 2473

Pharmacodynamics • Changes may occur in: - Receptor density - Receptor structure - Propagation of signals • Sensitivity for various drugs - Increased ( ): psychiatric drugs, opioids, dopamine, agonists, parasympaticolytics - Decreased ( ): ß-blockers and insulin

ADRs in elderly: risk of falling • Balance is complex mechanism controlled by - Vestibular system, Vision, Proprioception (orientation and movement), Muscle responses • Falling may occur due to - Cardiovascular effects: Orthostatic effects - Inadequate response due to vasodilatation - Reduction in function of baroreceptors - Insufficient fluid intake - α- or ß blocking agents, duiretics, ACE inhibitors, … • Psycho active drugs - Sedation, dizziness, confusion - Effect on extrapyramidal system - Muscle relaxants (benzodiazepines)
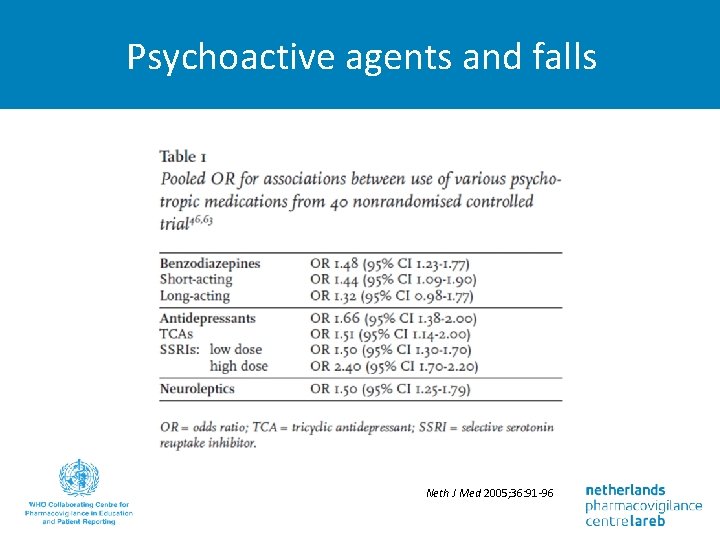
Psychoactive agents and falls Neth J Med 2005; 36: 91 -96

ADRs in elderly: thermoregulation • Problems in thermoregulation - Homeostasis for maintaining core temperature is changed - Ability to increase temperature (shivering) - Risk for hypothermia/poikilothermia • Antipsychotics may increase risk!

ADRs in elderly: cerebral function • Loss of neurons and receptors for cholinergic transmission • Additional neurologic disorders (i. e. stroke) • Anticholinergic drugs may influence cerebral function to large extent

Summary: Do. TS and basic aspects • Dose - Pharmacological mechanism - Drug properties • Time • Susceptibility - Patient properties - Pharmacodynamics - Pharmacokinetics

Summary / Exam question (1) Two patients have developed an ADR while taking a drug: • Patient 1 developed an anaphylactic shock, while using amoxicillin • Patient 2 suffered from stomach bleeding, while using aspirin. Answer which type of adverse drug reaction (A or B) patient 1 and 2 are suffering from: a) The ADRs in patient 1 and 2 are both type-A-reactions b) The ADRs in patient 1 and 2 are both type-B-reaction c) The ADR in patient 1 is a type-A, and in patient 2 a type-Breaction d) The ADR in patient 1 is a type-B, and in patient 2 a type-Areaction

Summary / Exam question (2) An elderly lady (80) experienced dizziness due to orthostatic hypotension after start of enalapril (ACE-inhibitor) for hypertension 1. Type ADR A to G, and 1 O or 2 O? 2. Classify the appropriate Do. TS a) Do: therapeutic, T: time independent, S: age, gender b) Do: supratherapeutic, T: early, S: age, renal function c) Do: therapeutic, T: first dose, S: age, renal function d) Do: subtherapeutic, T; rapid, S: age, renal function

Learning objectives • In this lecture, you learn to - apply the pathophysiologic principles that underlie the occurrence of side effects • Characterize adverse drug reactions based different formats: - type A/B classification and Do. TS classification - aspects which play a role in the development of side effects • Discuss which patients have an increased risk of developing side effects
- Slides: 53