Particle Theory of Matter SNC 1 D Chemistry














- Slides: 19

Particle Theory of Matter SNC 1 D - Chemistry Unit

Particle Theory • A theory is a series of statements which are developed to explain several related observations • A model is used as a guide for the imagination when using a theory

Particle Theory The particle theory states that… 1. All matter is made of small particles

Particle Theory 2. All particles of the same substance are made up of the same particles, regardless of state (solid, liquid, gas)

Particle Theory 3. The particles attract one another. These forces get stronger as the particles get closer

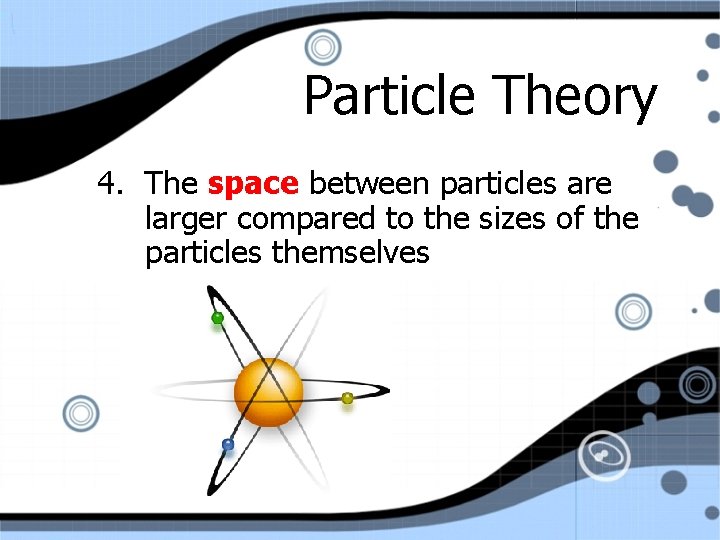
Particle Theory 4. The space between particles are larger compared to the sizes of the particles themselves

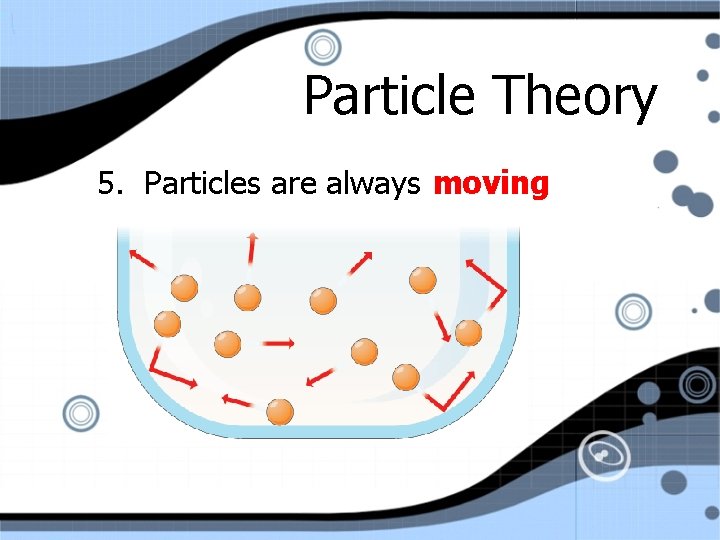
Particle Theory 5. Particles are always moving

States of Matter

States of Matter • Some substances exist only in one state of matter

States of Matter • Some substances exist in more than one state of matter. Water is a common example

Solids • attraction between particles is strong • space between particles is small (particles are close together) • there is little motion of particles - movement is mostly vibrational

Liquids • attraction between particles is medium • space between particles is medium • movement is vibrational and rotational

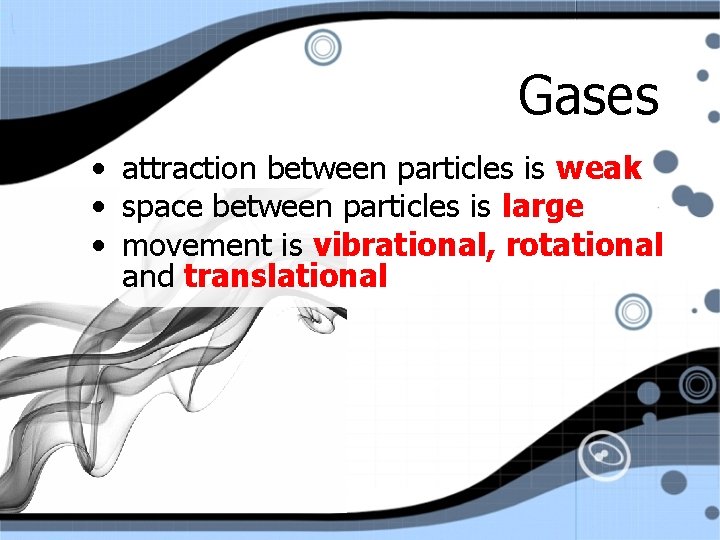
Gases • attraction between particles is weak • space between particles is large • movement is vibrational, rotational and translational

Gas vs. Vapour gas = gas at room temperature vapour = the gaseous state of a substance that is liquid at room temperature

Using the particle theory, explain what happens when you heat a substance.

Heat • heat = energy • added energy breaks the attractive forces between the particles • particles spread apart and movement increases

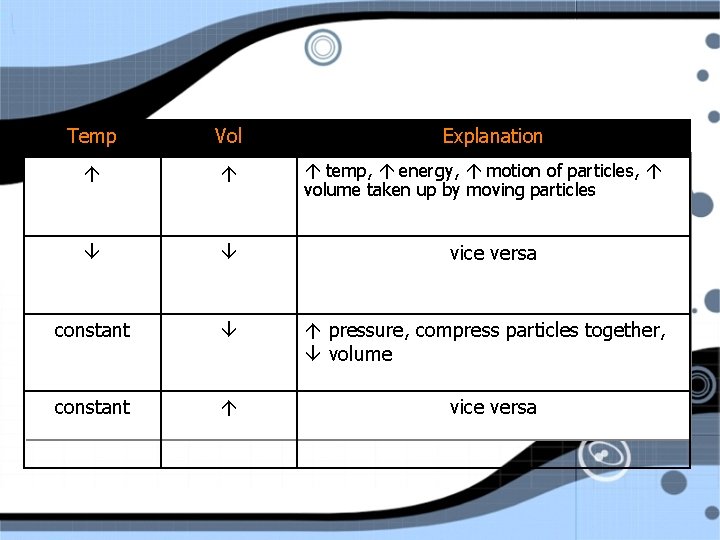
Temp Vol constant Explanation temp, energy, motion of particles, volume taken up by moving particles vice versa pressure, compress particles together, volume vice versa

Diffusion is the movement of particles from an area of high concentration to an area of low concentration

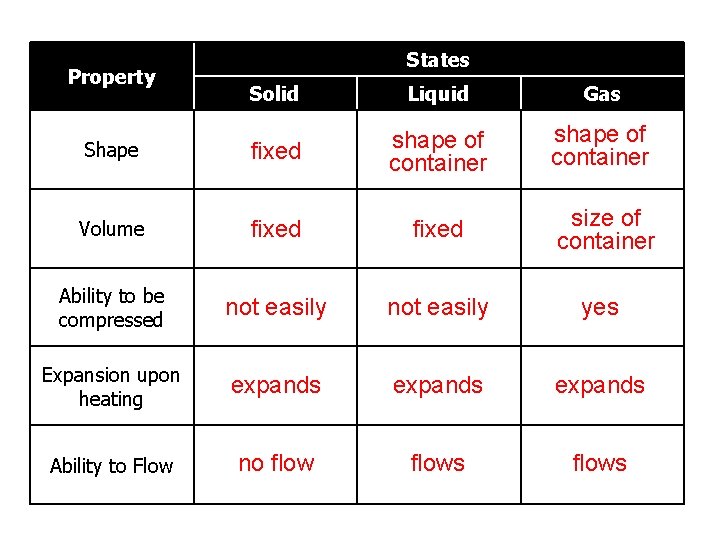
Property States Solid Liquid Gas Shape fixed shape of container Volume fixed size of container Ability to be compressed not easily yes Expansion upon heating expands Ability to Flow no flows