Mepact mifamurtide Fulfilling an unmet need in osteosarcoma
























- Slides: 42

Mepact® (mifamurtide) Fulfilling an unmet need in osteosarcoma treatment Prescribing information can be found at end of the presentation EUCAN/MEP/2013 -10019 September 2013

Osteosarcoma ● Second most common type of primary bone cancer 1 ● Disease of children and young adults 2 – Most common between ages of 10 -20 years 2 – Accounts for >10% of all solid tumours in adolescents aged 15 -19 years 3 ● More common in males than females 3 ● Incidence: 0. 2 -0. 3 per 100, 000 population in Europe 3 – Estimated 1, 135 new cases every year in Europe 1 1. Stiller CA et al. Eur J Cancer 2013; 49: 684 -695. 2. Meyers PA, Gorlick R. Pediatr Clin North Am 1997; 44(4): 973 -990. 3. Bone sarcomas: ESMO Clinical Practice Guidelines. Ann Oncol 2012; 23(7): vii 100 -vii 109. EUCAN/MEP/2013 -10019 September 2013

Osteosarcoma ● Most common sites are ends of long bones 1 – Approximately half of tumours begin around the knee ● Around 20% of newly diagnosed patients have detectable lung metastases 1 – Almost all patients have subclinical microscopic metastases – Death from osteosarcoma is almost always the result of progressive pulmonary metastases ● Relapse rate in newly diagnosed patients can be as high as 30% 2 – Most recurrences occur as lung metastases 1 1. Meyers PA, Gorlick R. Pediatr Clin North Am 1997; 44(4): 973 -990. 2. Grimer RJ. Lancet Oncol 2005: 6: 85 -92. EUCAN/MEP/2013 -10019 September 2013

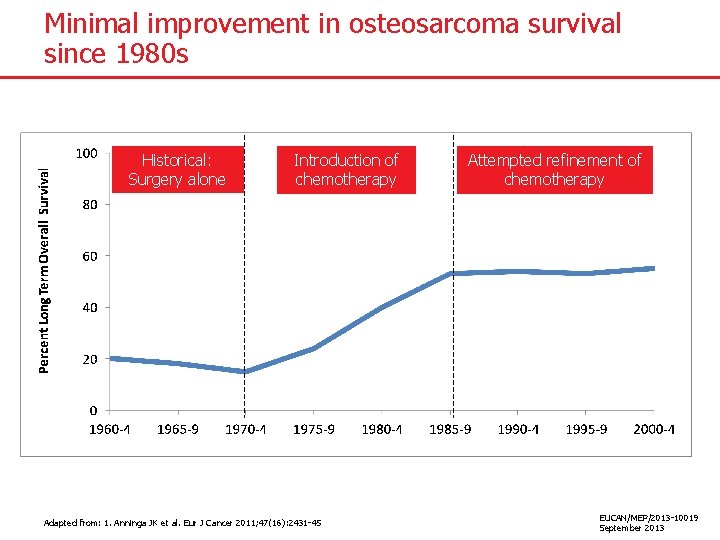
Minimal improvement in osteosarcoma survival since 1980 s Historical: Surgery alone Introduction of chemotherapy Adapted from: 1. Anninga JK et al. Eur J Cancer 2011; 47(16): 2431 -45 Attempted refinement of chemotherapy EUCAN/MEP/2013 -10019 September 2013

Minimal improvement in osteosarcoma survival since 1980 s ● Before the 1970 s patients underwent amputation and no chemotherapy – Long term overall survival was <20%1 ● By the mid 1980 s long term overall survival up to 50 -60% 1 ● Some studies showed better rates, but overall little significant improvement for over three decades 1 ● Meta-analysis of chemotherapy studies showed 1: – 5 year overall survival of 60% with 2 -drug regimen compared with 66% for 3 -drug regimen (p=0. 04) – No benefit of a fourth drug ● Most investigators agree treatment has reached a plateau 2 1. Anninga JK et al. Eur J Cancer 2011; 47(16): 2431 -45. 2. Meyers PA. Expert Revs Anticancer Ther 2009; 9: 1035 -1049. EUCAN/MEP/2013 -10019 September 2013

Significant unmet need in osteosarcoma treatment ● Today, long-term (5 -year) survival for localised disease is 50 -60% 1 ● Continuing improvements in survival have been seen for many childhood cancers, but the news is less positive for osteosarcoma 2 − No improvement in past 30 years despite the introduction of multiagent chemotherapy 1 ● Therefore, improving overall survival for osteosarcoma patients represents a significant unmet need 1. Anninga JK et al. Eur J Cancer 2011; 47(16): 2431 -45 2. Gatta G et al. J Clin Oncol 2005; 23: 3742 -3751 EUCAN/MEP/2013 -10019 September 2013

The potential role of immunotherapy in osteosarcoma ● Immunotherapy is a treatment strategy which upregulates the body’s immune response to cancer cells and thus increases tumouricidal activity 1 ● There is a growing body of evidence to support immunotherapy as an effective treatment in osteosarcoma 1 ● Postoperative infection after endoprosthetic surgery has been shown to improve survival outcome which suggested a role for infectioninduced anti-tumour immune response 2 1. Mori K et al. Oncology Reports 2006; 15: 693 -700 2. Jeys LM et al. Ann Surg Oncol. 2007; 14(10): 2887 -95 EUCAN/MEP/2013 -10019 September 2013

The evidence for immunotherapy in osteosarcoma ● Potential strategies targeting the host immune system include: - Immune modulation (interferon-α 1, muramyl tripeptide phosphatidyl ethanolamine MTP-PE 2) - Cellular therapy (cytotoxic T-cells, primed dendritic cells 3) ● Study results cannot be extrapolated across immunotherapeutic agents: – Immunotherapy with interferon-α as an adjuvant therapy has been evaluated following initial encouraging results 1 however preliminary data from the EURAMOS-1 trial concluded that there is no evidence to support interferon-α in this setting 4 – Mepact has demonstrated a proven benefit on overall survival 2 1. 2. 3. 4. Müller C et al. Acta Oncologica 2005; 44: 475 -480 Meyers PA et al. J Clin Oncol 2008; 26(4): 633 -638 Mori K et al. Oncology Reports 2006; 15: 693 -700 Bielack S et al. J Clin Oncol 31, 2013 (suppl; abstr LBA 10504) EUCAN/MEP/2013 -10019 September 2013

Introduction - What is Mepact? ● Mepact (mifamurtide) is L-MTP-PE, a liposomal formulation of Muramyl Tripeptide Phosphatidyl Ethanolamine (MTP-PE)1 – Liposomal formulation facilitates uptake by tissue macrophages after i. v. infusion – A potent activator of macrophages ● L-MTP-PE was initially developed as an immunostimulant with significant anti-tumour effects in preclinical models 1, 2 ● MTP-PE is a synthetic analog of muramyl dipeptide (MDP)3 – The smallest natural immune stimulatory component of the mycobacterium cell wall 1 1. Nardin A. et al. Current Cancer Drug Targets. 2006; 6: 123 -133 2. Fidler IJ. et al. Proc. Natl Acad. Sci. USA. 1981; 78: 1680 -1684 3. Mepact Sm. PC, March 2013 http: //www. medicines. org. uk/emc/medicine/22763 EUCAN/MEP/2013 -10019 September 2013

Mepact is well studied and characterised ● Extensive supporting data at the time of Marketing Authorisation submission (November 2006) – Administered to more than 700 patients in clinical trials 1 – More than 100 pre-clinical studies 1 ● 86 pharmacology studies, 12 pharmacokinetic studies and 20 toxicology studies were submitted as part of the EMA dossier 1 – Extensive clinical development program ● 18 Phase I and II studies were submitted as part of EMA dossier 1 1. EMA Assessment Report for Mepact EMEA/H/C/000802. EUCAN/MEP/2013 -10019 September 2013

Mepact is well studied and characterised ● Robust Phase III clinical data 1 – Leading paediatric cooperative study group (COG*) clinical trial – One of the largest studies ever completed – Consistent, significant and long-term survival benefit ● Almost one third reduction in relative risk of death with an absolute risk reduction of 8% (p=0. 03)1 – Favourable benefit/risk profile 2 ● Granted Orphan Medicinal Product Status in 20043 ● One of most studied paediatric oncology drugs * COG – Children’s Oncology Group 1. Meyers PA et al. J Clin Oncol. 2008; 26: 633 -638. 2. Meyers PA. Expert Revs Anticancer Ther 2009; 9: 1035 -1049. 3. EMA. EMEA/COMP/373/04 EUCAN/MEP/2013 -10019 September 2013

Mepact development pathway ● In pre-clinical models, Mepact was proven to be an immunostimulant with significant anti-tumour effects 1 – Anti-tumour effects are macrophage mediated ● Direct (activated macrophage tumour killing) ● Indirect (cytokine mediated, activation of immune cells) ● Liposomal formulation facilitates uptake by tissue macrophages after i. v. injection 1 – Particularly in lungs, nasopharynx, spleen, liver 2 ● Following pre-clinical models, results in dogs stimulated additional interest in Mepact 3 – Comparable model for human osteosarcoma 1. Nardin A et al. Current Cancer Drug Targets. 2006; 6: 123 -133. 2. Murray JL et al. J Clin Oncol. 1989; 7: 1915 -1925. 3. Meyers PA. Expert Revs Anticancer Ther 2009; 9: 1035 -1049. EUCAN/MEP/2013 -10019 September 2013

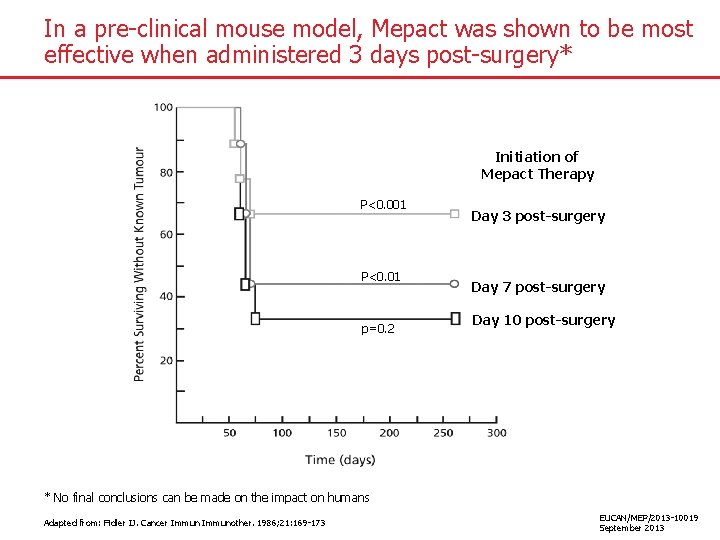
In a pre-clinical mouse model, Mepact was shown to be most effective when administered 3 days post-surgery* Initiation of Mepact Therapy P<0. 001 P<0. 01 p=0. 2 Day 3 post-surgery Day 7 post-surgery Day 10 10 post-surgery Day * No final conclusions can be made on the impact on humans Adapted from: Fidler IJ. Cancer Immunother. 1986; 21: 169 -173 EUCAN/MEP/2013 -10019 September 2013

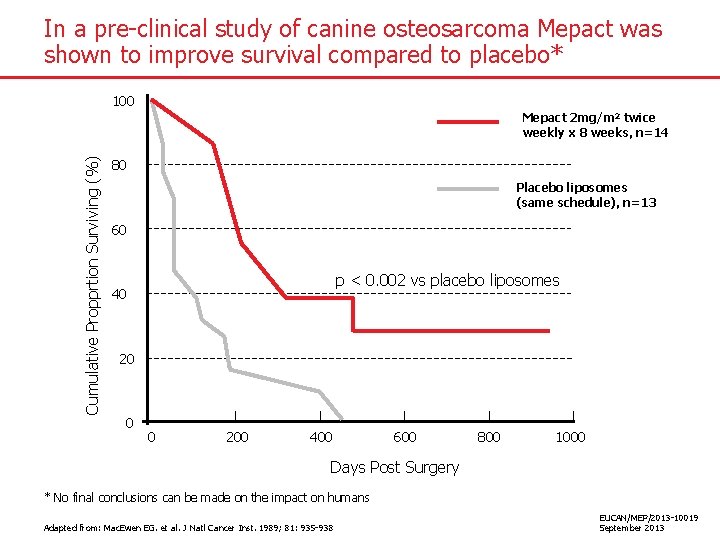
In a pre-clinical study of canine osteosarcoma Mepact was shown to improve survival compared to placebo* 100 Cumulative Propprtion Surviving (%) Mepact 2 mg/m 2 twice weekly x 8 weeks, n=14 80 Placebo liposomes (same schedule), n=13 60 p < 0. 002 vs placebo liposomes 40 20 0 0 200 400 600 800 1000 Days Post Surgery * No final conclusions can be made on the impact on humans Adapted from: Mac. Ewen EG. et al. J Natl Cancer Inst. 1989; 81: 935 -938 EUCAN/MEP/2013 -10019 September 2013

Phase I studies determined maximum tolerated dose and optimal biological dose ● Maximum Tolerated Dose 1, 2, 3 is 4 -6 mg/m 2 – Patients received doses from 0. 01 -12 mg/m 2 ● Optimal Biological Dose 3 – Induction of monocyte mediated tumouricidal activity ● Within hours of administration ● Wide range of doses assessed ● Optimal range between 0. 5 -2. 0 mg/m 2 – Doses above 2 mg/m 2 provide little increased effect – Similarity to other biologicals (IFN ) that activate monocytes/ macrophages ● Recommendation for future studies to be conducted using Optimal Biological Dose 3 1. Murray JL et al. JCO 1989; 7: 1915 -25. 2. Creaven PJ et al. J Biol Res Mod, 1990; 9: 492 -8. 3. Kleinerman ES et al. Cancer Res 1989; 49: 4665 -4670 EUCAN/MEP/2013 -10019 September 2013

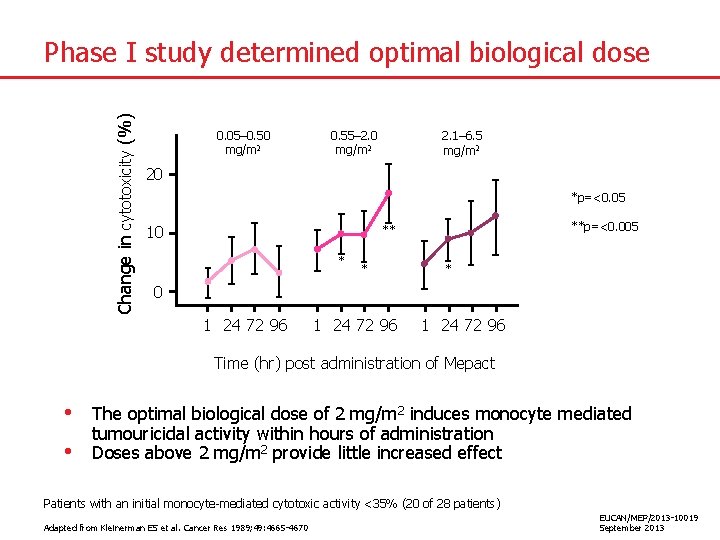
Change in cytotoxicity (%) Phase I study determined optimal biological dose 0. 05– 0. 50 mg/m 2 0. 55– 2. 0 mg/m 2 2. 1– 6. 5 mg/m 2 20 *p=<0. 05 10 **p=<0. 005 ** * 0 1 24 72 96 Time (hr) post administration of Mepact • • The optimal biological dose of 2 mg/m 2 induces monocyte mediated tumouricidal activity within hours of administration Doses above 2 mg/m 2 provide little increased effect Patients with an initial monocyte-mediated cytotoxic activity <35% (20 of 28 patients) Adapted from Kleinerman ES et al. Cancer Res 1989; 49: 4665 -4670 EUCAN/MEP/2013 -10019 September 2013

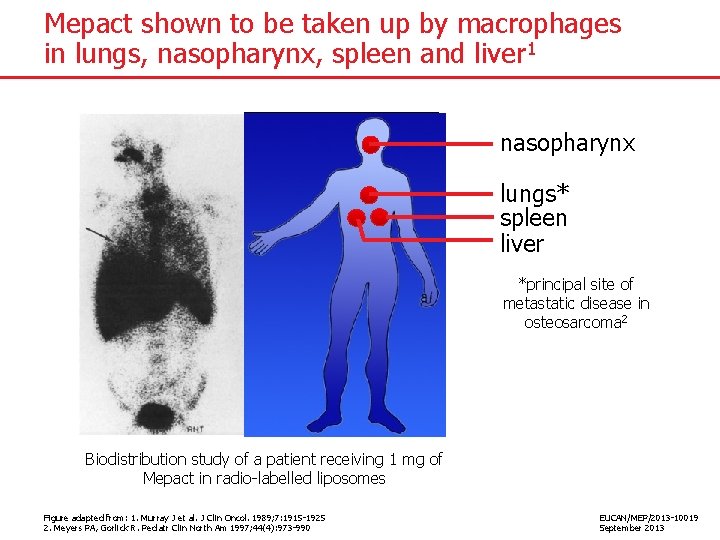
Mepact shown to be taken up by macrophages in lungs, nasopharynx, spleen and liver 1 nasopharynx lungs* spleen liver *principal site of metastatic disease in osteosarcoma 2 Biodistribution study of a patient receiving 1 mg of Mepact in radio-labelled liposomes Figure adapted from: 1. Murray J et al. J Clin Oncol. 1989; 7: 1915 -1925 2. Meyers PA, Gorlick R. Pediatr Clin North Am 1997; 44(4): 973 -990 EUCAN/MEP/2013 -10019 September 2013

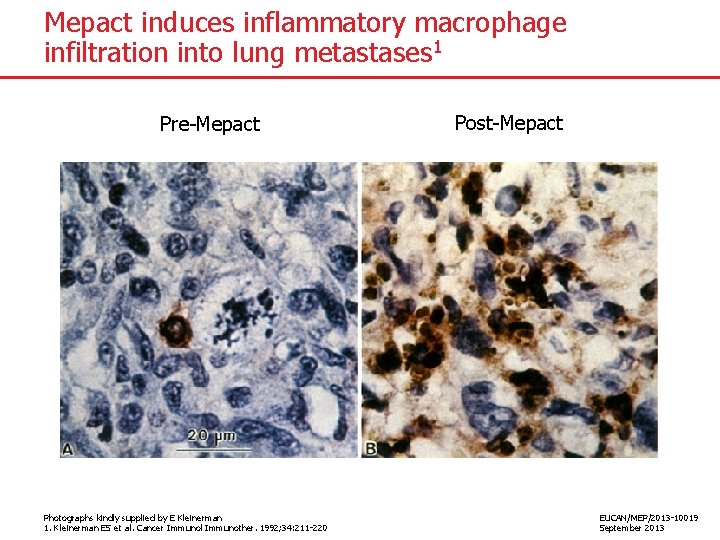
Mepact induces inflammatory macrophage infiltration into lung metastases 1 Pre-Mepact Photographs kindly supplied by E Kleinerman 1. Kleinerman ES et al. Cancer Immunol Immunother. 1992; 34: 211 -220 Post-Mepact EUCAN/MEP/2013 -10019 September 2013

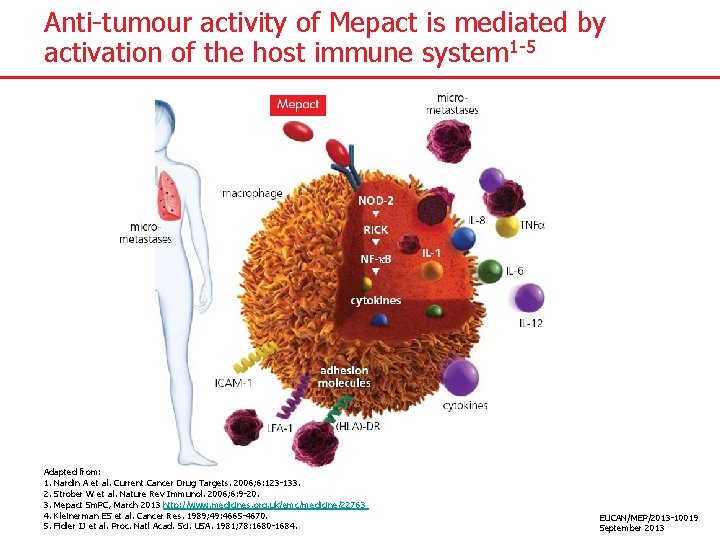
Measured immunological effects ● Activation of macrophages 1 ● Induction of tumouricidal monocytes 2 ● Elevated levels: – IL-12, 3 −IL-24 – IL-61, 3 -7 −IL-83, 7 – IL-123 −TNF- 1, 3, 4, 7 – C-reactive protein (CRP)1, 4, 7 −Neopterin 4, 6 -8 ● Increased levels of β 2 -microglobulin 6 ● Leukocytosis 8 1. 2. 3. 4. 5. 6. 7. 8. Kleinerman ES et al. J Clin Onco. 1992; 10: 1310 -1316. Kleinerman ES et al. Cancer Research 1989; 49: 4665 -4670. Mepact Sm. PC, March 2013 http: //www. medicines. org. uk/emc/medicine/22763 Sculier JP et al. Drug Invest. 1993; 6: 276 -284. Frost H et al. J Biol Response Modifiers. 1990; 9: 160 -166. Liebes L et al. J Natl Cancer Inst. 1992; 84: 694 -699. Asano T. Kleinerman ES. J Immunother. 1993; 14: 286 -292. Landmann R et al. Biotherapy. 1994; 7: 1 -12. EUCAN/MEP/2013 -10019 September 2013

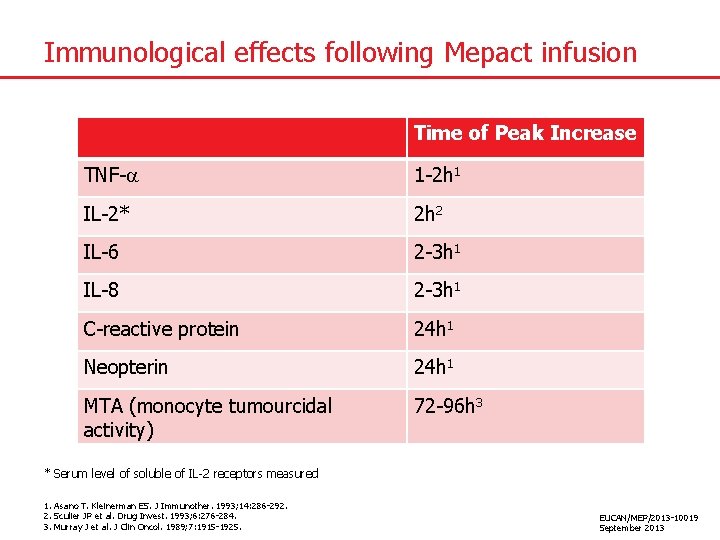
Immunological effects following Mepact infusion Time of Peak Increase TNF- 1 -2 h 1 IL-2* 2 h 2 IL-6 2 -3 h 1 IL-8 2 -3 h 1 C-reactive protein 24 h 1 Neopterin 24 h 1 MTA (monocyte tumourcidal activity) 72 -96 h 3 * Serum level of soluble of IL-2 receptors measured 1. Asano T. Kleinerman ES. J Immunother. 1993; 14: 286 -292. 2. Sculier JP et al. Drug Invest. 1993; 6: 276 -284. 3. Murray J et al. J Clin Oncol. 1989; 7: 1915 -1925. EUCAN/MEP/2013 -10019 September 2013

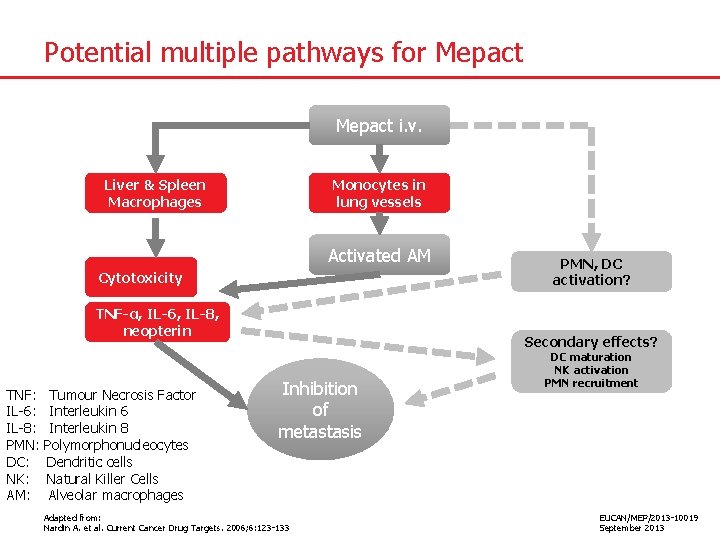
Potential multiple pathways for Mepact i. v. Liver & Spleen Macrophages Monocytes in lung vessels Activated AM Cytotoxicity TNF-α, IL-6, IL-8, neopterin TNF: IL-6: IL-8: PMN: DC: NK: AM: Tumour Necrosis Factor Interleukin 6 Interleukin 8 Polymorphonucleocytes Dendritic cells Natural Killer Cells Alveolar macrophages PMN, DC activation? Secondary effects? Inhibition of metastasis Adapted from: Nardin A. et al. Current Cancer Drug Targets. 2006; 6: 123 -133 DC maturation NK activation PMN recruitment EUCAN/MEP/2013 -10019 September 2013

Anti-tumour activity of Mepact is mediated by activation of the host immune system 1 -5 Adapted from: 1. Nardin A et al. Current Cancer Drug Targets. 2006; 6: 123 -133. 2. Strober W et al. Nature Rev Immunol. 2006; 6: 9 -20. 3. Mepact Sm. PC, March 2013 http: //www. medicines. org. uk/emc/medicine/22763 4. Kleinerman ES et al. Cancer Res. 1989; 49: 4665 -4670. 5. Fidler IJ et al. Proc. Natl Acad. Sci. USA. 1981; 78: 1680 -1684. EUCAN/MEP/2013 -10019 September 2013

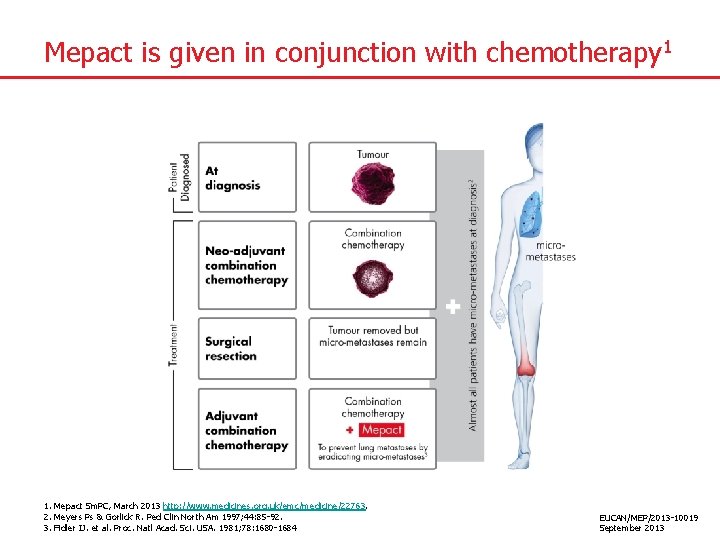
Mepact is given in conjunction with chemotherapy 1 1. Mepact Sm. PC, March 2013 http: //www. medicines. org. uk/emc/medicine/22763. 2. Meyers Ps & Gorlick R. Ped Clin North Am 1997; 44: 85 -92. 3. Fidler IJ. et al. Proc. Natl Acad. Sci. USA. 1981; 78: 1680 -1684 EUCAN/MEP/2013 -10019 September 2013

Mepact administration ● Supplied as lyophilised powder – Liposome suspensions are formed after hydration of lyophilisate ● 48 Doses over 36 weeks – 2 mg/m 2 – One hour i. v. infusion Examples only – replace with vial and pack shots appropriate to your market – First 12 weeks: ● Twice weekly ● Total 24 doses – Next 24 weeks: ● Once weekly ● Total 24 infusions Mepact Sm. PC, March 2013 http: //www. medicines. org. uk/emc/medicine/22763 EUCAN/MEP/2013 -10019 September 2013

The Children’s Oncology Group Phase III study of Mepact ● National Cancer Institute sponsored Children’s Oncology Group (COG) study 1, 2 – Designed and conducted independently of corporate sponsor – 1993 -1997 enrollment – 1993 -2007 follow-up ● One of the largest studies completed in osteosarcoma ● Newly diagnosed patients (enrolled n=793*; age <31)2 – 662 non-metastatic resectable disease – 115 with more metastatic or unresectable disease ● Independently analysed and published survival benefit 2 – Meyers PA, Schwartz CL, Krailo MK et al. Osteosarcoma: the addition of muramyl tripeptide to chemotherapy improves overall survival - A report from the Children’s Oncology Group. J Clin Oncol 2008; 26: 633 -638 * 777 patients entered the study. 16 were ineligible for treatment 1. 2. Meyers PA. et al. J Clin Oncol. 2005; 23: 2004 -2011. Meyers PA. et al. J Clin Oncol. 2008; 26: 633 -638. EUCAN/MEP/2013 -10019 September 2013

The COG Phase III study Objectives The study posed two questions in a 2 x 2 factorial design 1. To compare overall survival and event free survival of a three drug chemotherapy regimen of cisplatin, doxorubicin and high-dose methotrexate with a four drug regimen using these agents with ifosfamide 2. Whether the addition of Mepact to chemotherapy would improve overall survival or event free survival Meyers PA et al. J Clin Oncol 2008; 26: 633 -638 EUCAN/MEP/2013 -10019 September 2013

The COG Phase III study Factorial design Induction 3 drugs Regimen A Cisplatin Doxorubicin High-Dose Methotrexate 3 drugs Regimen B Ifosfamide Doxorubicin High-Dose Methotrexate Maintenance 3 drugs CIS, DOX, HDMTX Without Mepact (A-) With Mepact (A+) 4 drugs IFOS, CIS, DOX, HDMTX Without Mepact (B-) With Mepact (B+) During induction phase Regimen A patients received 2 doses of cisplatin (CIS) and Regimen B patients received 2 courses of ifosfamide (IFOS) During maintenance therapy all patients received doxorubicin (DOX) and high-dose methotrexate (HDMTX) plus: • Regimen A: 2 doses of cisplatin • Regimen B: 3 courses ifosfamide + 4 doses cisplatin 1. Meyers PA et al. J Clin Oncol 2005; 23: 2004 -2011 2. Meyers PA et al. J Clin Oncol 2008; 26: 633 -638 EUCAN/MEP/2013 -10019 September 2013

The COG Phase III study Design overview INDUCTION R A N D O M I S A T I O N A Cisplatin Doxorubicin HDMTX B Ifosfamide Doxorubicin HDMTX D E F I N I T I V E S U R G E R Y H I S T O L O G I C A L E V A L U A T I O N MAINTENANCE Cisplatin, Doxorubicin, HDMTX A- Cisplatin, Doxorubicin, HDMTX, Mepact A+ B- Ifosfamide, Cisplatin, Doxorubicin, HDMTX, Mepact Weeks 20 27 B+ 36 Regimens A and B: Doxorubicin x 6 ea (25 mg/m 2/day x 3), Cisplatin x 4 ea (120 mg/m 2), and Methotrexate x 12 ea (12 g/m 2) Regimen B only: Ifosfamide x 5 ea (1. 8 g/m 2/day x 5) HDMTX = High-Dose Methotrexate; 1. Meyers PA et al. J Clin Oncol 2005; 23: 2004 -2011 2. Meyers PA et al. J Clin Oncol 2008; 26: 633 -638 EUCAN/MEP/2013 -10019 September 2013

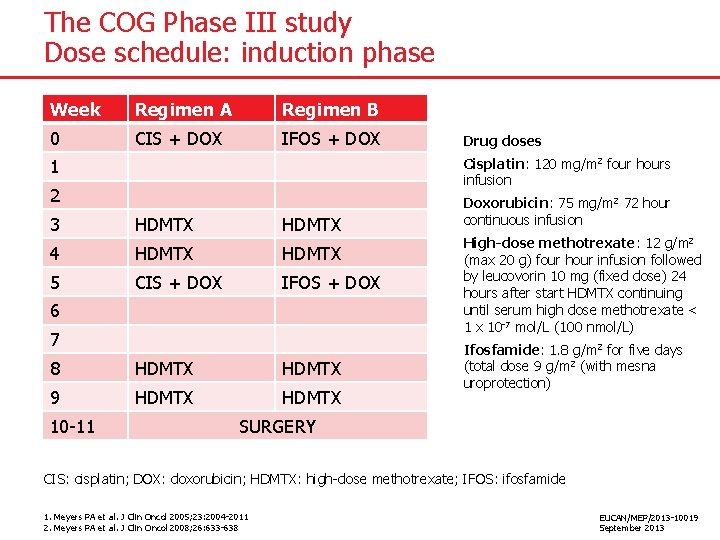
The COG Phase III study Dose schedule: induction phase Week Regimen A Regimen B 0 CIS + DOX IFOS + DOX Drug doses Cisplatin: 120 mg/m 2 four hours infusion 1 2 Doxorubicin: 75 mg/m 2 72 hour continuous infusion 3 HDMTX 4 HDMTX 5 CIS + DOX IFOS + DOX High-dose methotrexate: 12 g/m 2 (max 20 g) four hour infusion followed by leucovorin 10 mg (fixed dose) 24 hours after start HDMTX continuing until serum high dose methotrexate < 1 x 10 -7 mol/L (100 nmol/L) Ifosfamide: 1. 8 g/m 2 for five days (total dose 9 g/m 2 (with mesna uroprotection) 6 7 8 HDMTX 9 HDMTX 10 -11 SURGERY CIS: cisplatin; DOX: doxorubicin; HDMTX: high-dose methotrexate; IFOS: ifosfamide 1. Meyers PA et al. J Clin Oncol 2005; 23: 2004 -2011 2. Meyers PA et al. J Clin Oncol 2008; 26: 633 -638 EUCAN/MEP/2013 -10019 September 2013

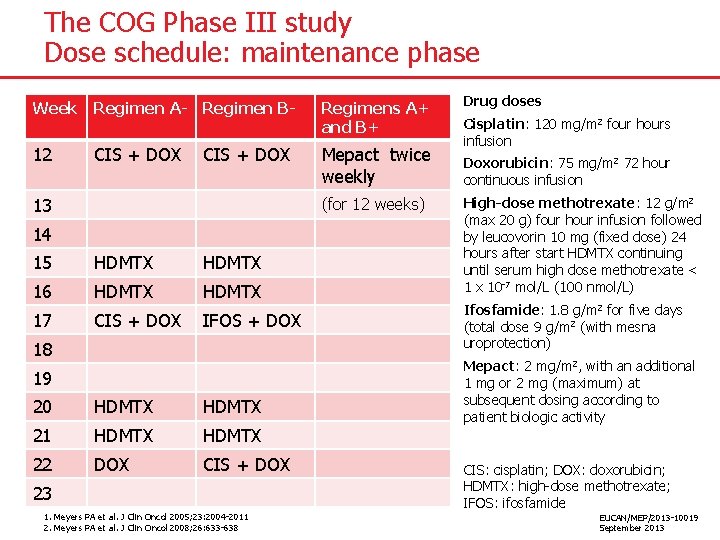
The COG Phase III study Dose schedule: maintenance phase Week Regimen A- Regimen B- Regimens A+ and B+ 12 CIS + DOX Mepact twice weekly CIS + DOX 13 (for 12 weeks) 14 15 HDMTX 16 HDMTX 17 CIS + DOX IFOS + DOX 18 19 20 HDMTX 21 HDMTX 22 DOX CIS + DOX 23 1. Meyers PA et al. J Clin Oncol 2005; 23: 2004 -2011 2. Meyers PA et al. J Clin Oncol 2008; 26: 633 -638 Drug doses Cisplatin: 120 mg/m 2 four hours infusion Doxorubicin: 75 mg/m 2 72 hour continuous infusion High-dose methotrexate: 12 g/m 2 (max 20 g) four hour infusion followed by leucovorin 10 mg (fixed dose) 24 hours after start HDMTX continuing until serum high dose methotrexate < 1 x 10 -7 mol/L (100 nmol/L) Ifosfamide: 1. 8 g/m 2 for five days (total dose 9 g/m 2 (with mesna uroprotection) Mepact: 2 mg/m 2, with an additional 1 mg or 2 mg (maximum) at subsequent dosing according to patient biologic activity CIS: cisplatin; DOX: doxorubicin; HDMTX: high-dose methotrexate; IFOS: ifosfamide EUCAN/MEP/2013 -10019 September 2013

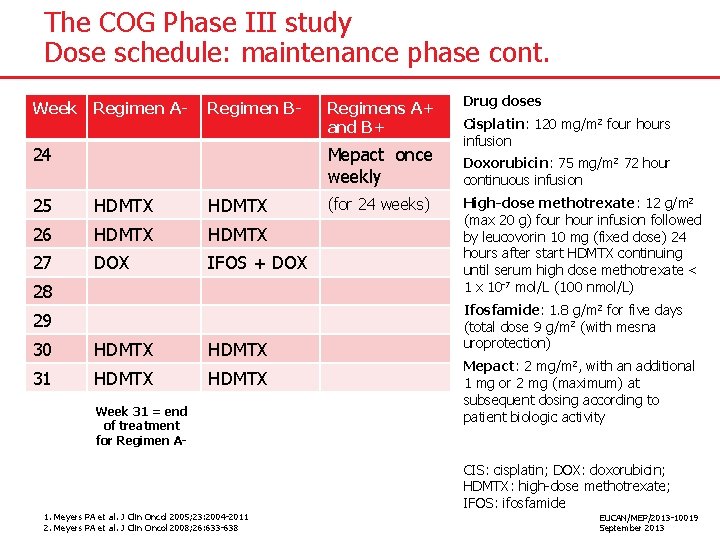
The COG Phase III study Dose schedule: maintenance phase cont. Week Regimen A- Regimen B- 24 Regimens A+ and B+ Mepact once weekly 25 HDMTX 26 HDMTX 27 DOX IFOS + DOX 28 29 30 HDMTX 31 HDMTX Week 31 = end of treatment for Regimen A- 1. Meyers PA et al. J Clin Oncol 2005; 23: 2004 -2011 2. Meyers PA et al. J Clin Oncol 2008; 26: 633 -638 (for 24 weeks) Drug doses Cisplatin: 120 mg/m 2 four hours infusion Doxorubicin: 75 mg/m 2 72 hour continuous infusion High-dose methotrexate: 12 g/m 2 (max 20 g) four hour infusion followed by leucovorin 10 mg (fixed dose) 24 hours after start HDMTX continuing until serum high dose methotrexate < 1 x 10 -7 mol/L (100 nmol/L) Ifosfamide: 1. 8 g/m 2 for five days (total dose 9 g/m 2 (with mesna uroprotection) Mepact: 2 mg/m 2, with an additional 1 mg or 2 mg (maximum) at subsequent dosing according to patient biologic activity CIS: cisplatin; DOX: doxorubicin; HDMTX: high-dose methotrexate; IFOS: ifosfamide EUCAN/MEP/2013 -10019 September 2013

The COG Phase III study Dose schedule: maintenance phase cont. Week Regimen A- Regimen B- Regimens A+ and B+ 32 CIS Mepact once weekly 33 (for 24 weeks) 34 35 IFOS 36 CIS Week 38 = end of treatment for Regimen B- 1. Meyers PA et al. J Clin Oncol 2005; 23: 2004 -2011 2. Meyers PA et al. J Clin Oncol 2008; 26: 633 -638 Cisplatin: 120 mg/m 2 four hours infusion Doxorubicin: 75 mg/m 2 72 hour continuous infusion High-dose methotrexate: 12 g/m 2 (max 20 g) four hour infusion followed by leucovorin 10 mg (fixed dose) 24 hours after start HDMTX continuing until serum high dose methotrexate < 1 x 10 -7 mol/L (100 nmol/L) Ifosfamide: 1. 8 g/m 2 for five days (total dose 9 g/m 2 (with mesna uroprotection) 37 38 Drug doses Regimens A+ and B+ Mepact treatment continued until week 47 Mepact: 2 mg/m 2, with an additional 1 mg or 2 mg (maximum) at subsequent dosing according to patient biologic activity CIS: cisplatin; DOX: doxorubicin; HDMTX: high-dose methotrexate; IFOS: ifosfamide EUCAN/MEP/2013 -10019 September 2013

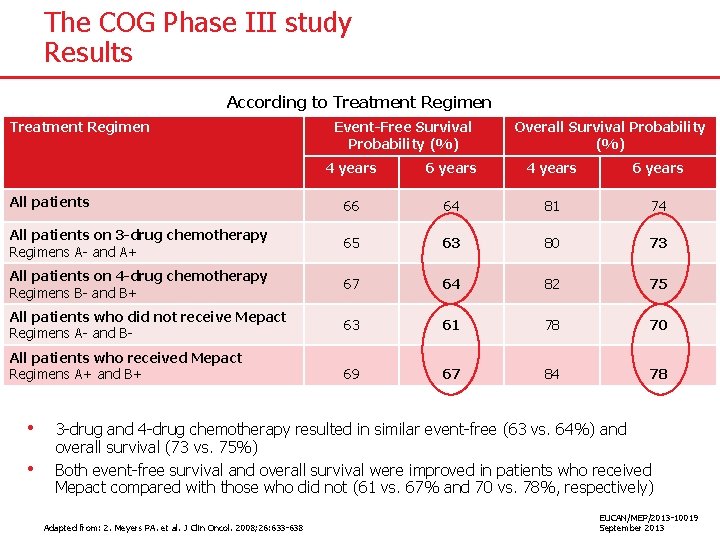
The COG Phase III study Results According to Treatment Regimen Event-Free Survival Probability (%) Overall Survival Probability (%) 4 years 6 years All patients 66 64 81 74 All patients on 3 -drug chemotherapy Regimens A- and A+ 65 63 80 73 All patients on 4 -drug chemotherapy Regimens B- and B+ 67 64 82 75 All patients who did not receive Mepact Regimens A- and B- 63 61 78 70 69 67 84 78 All patients who received Mepact Regimens A+ and B+ • • 3 -drug and 4 -drug chemotherapy resulted in similar event-free (63 vs. 64%) and overall survival (73 vs. 75%) Both event-free survival and overall survival were improved in patients who received Mepact compared with those who did not (61 vs. 67% and 70 vs. 78%, respectively) Adapted from: 2. Meyers PA. et al. J Clin Oncol. 2008; 26: 633 -638 EUCAN/MEP/2013 -10019 September 2013

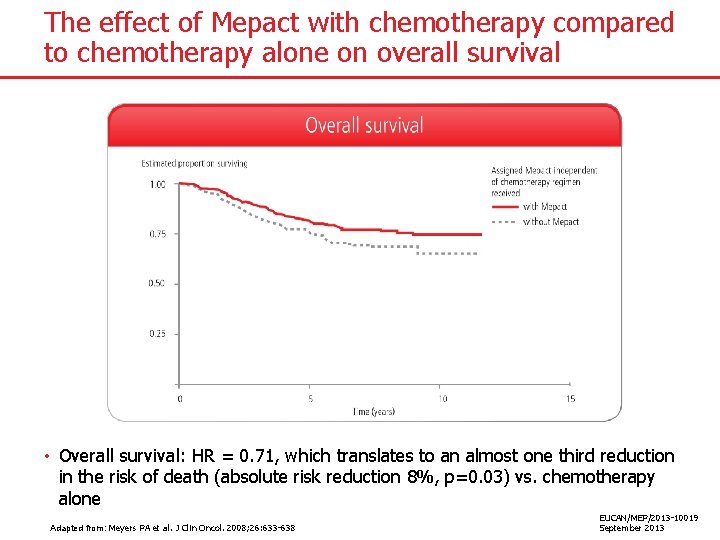
The effect of Mepact with chemotherapy compared to chemotherapy alone on overall survival • Overall survival: HR = 0. 71, which translates to an almost one third reduction in the risk of death (absolute risk reduction 8%, p=0. 03) vs. chemotherapy alone Adapted from: Meyers PA et al. J Clin Oncol. 2008; 26: 633 -638 EUCAN/MEP/2013 -10019 September 2013

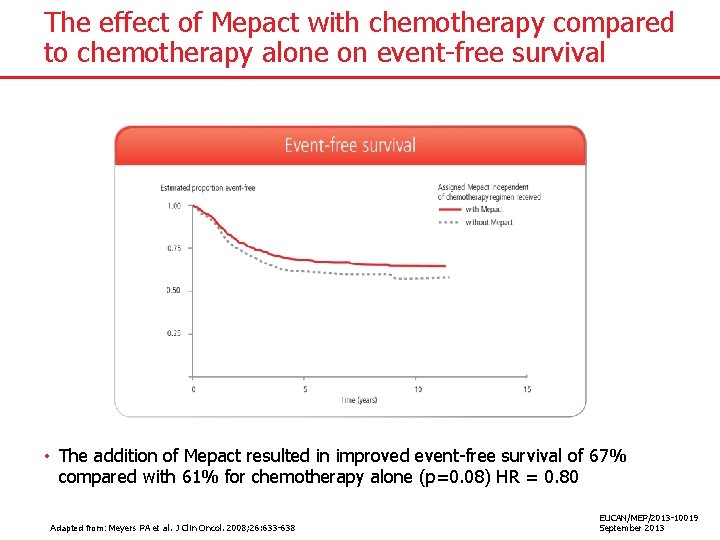
The effect of Mepact with chemotherapy compared to chemotherapy alone on event-free survival • The addition of Mepact resulted in improved event-free survival of 67% compared with 61% for chemotherapy alone (p=0. 08) HR = 0. 80 Adapted from: Meyers PA et al. J Clin Oncol. 2008; 26: 633 -638 EUCAN/MEP/2013 -10019 September 2013

Results of the Mepact with our without multi-agent chemotherapy on overall survival ● The addition of Mepact to chemotherapy: – Improved 6 -year overall survival from 70% to 78% (p=0. 03) – Improved 6 -year event-free survival from 61% to 67% (p=0. 08) – Reduced the risk of death by almost one third (relative risk=0. 71, absolute risk reduction 8%) Meyers PA. et al. J Clin Oncol. 2008; 26: 633 -638 EUCAN/MEP/2013 -10019 September 2013

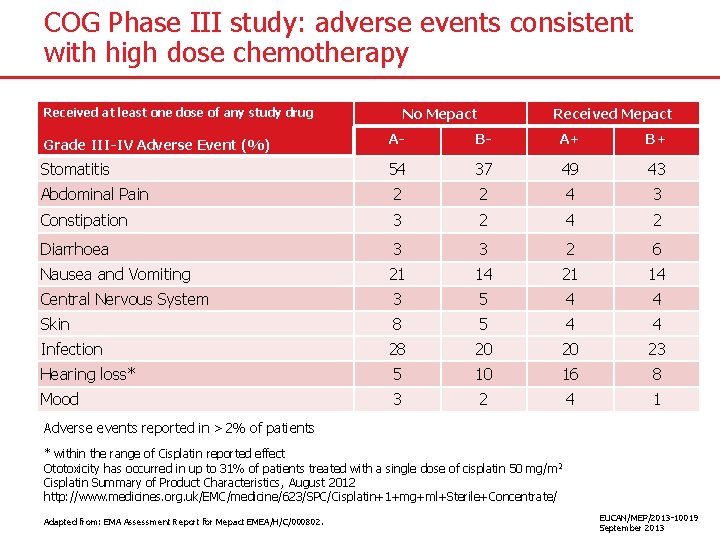
COG Phase III study: adverse events consistent with high dose chemotherapy No Mepact Received at least one dose of any study drug Received Mepact Grade III-IV Adverse Event (%) A- B- A+ B+ Stomatitis 54 37 49 43 Abdominal Pain 2 2 4 3 Constipation 3 2 4 2 Diarrhoea 3 3 2 6 Nausea and Vomiting 21 14 Central Nervous System 3 5 4 4 Skin 8 5 4 4 Infection 28 20 20 23 Hearing loss* 5 10 16 8 Mood 3 2 4 1 Adverse events reported in >2% of patients * within the range of Cisplatin reported effect Ototoxicity has occurred in up to 31% of patients treated with a single dose of cisplatin 50 mg/m 2 Cisplatin Summary of Product Characteristics, August 2012 http: //www. medicines. org. uk/EMC/medicine/623/SPC/Cisplatin+1+mg+ml+Sterile+Concentrate/ Adapted from: EMA Assessment Report for Mepact EMEA/H/C/000802. EUCAN/MEP/2013 -10019 September 2013

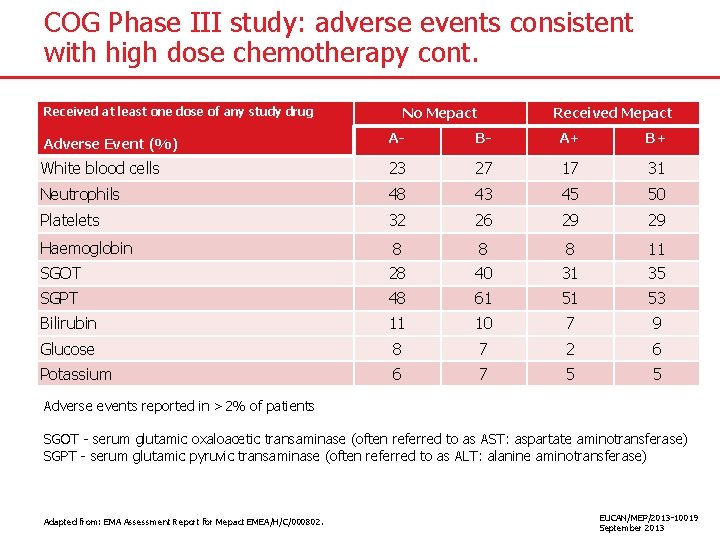
COG Phase III study: adverse events consistent with high dose chemotherapy cont. No Mepact Received at least one dose of any study drug Received Mepact Adverse Event (%) A- B- A+ B+ White blood cells 23 27 17 31 Neutrophils 48 43 45 50 Platelets 32 26 29 29 Haemoglobin 8 8 8 11 SGOT 28 40 31 35 SGPT 48 61 51 53 Bilirubin 11 10 7 9 Glucose 8 7 2 6 Potassium 6 7 5 5 Adverse events reported in >2% of patients SGOT - serum glutamic oxaloacetic transaminase (often referred to as AST: aspartate aminotransferase) SGPT - serum glutamic pyruvic transaminase (often referred to as ALT: alanine aminotransferase) Adapted from: EMA Assessment Report for Mepact EMEA/H/C/000802. EUCAN/MEP/2013 -10019 September 2013

Mepact adverse event profile Very Common Side Effects (>1/10) ● Anaemia ● Anorexia ● Headache, dizziness ● Tachycardia ● Hypertension, hypotension ● Dyspnoea, tachypnoea, cough ● Vomiting, diarrhoea, constipation, abdominal pain, nausea ● Hyperhidrosis ● Myalgia, arthralgia, back pain, pain in extremity ● Fever, chills, fatigue, hypothermia, pain, malaise, asthenia, chest pain Selected Common Side Effects (>1/100 to <1/10) ● Sepsis ● Leukopenia ● Thrombocytopenia ● Granulocytopenia ● Hypokalaemia ● Vertigo ● Tinnitus ● Hearing loss Please refer to Summary of Product Characteristics (Sm. PC) for full list of adverse events Mepact Sm. PC, March 2013 http: //www. medicines. org. uk/emc/medicine/22763 EUCAN/MEP/2013 -10019 September 2013

Mepact contraindications ● Hypersensitivity to the active substance or to any of the excipients ● Concurrent use with ciclosporin or other calcineurin inhibitors ● Concurrent use with high-dose non-steroidal anti-inflammatory drugs (NSAIDs, cyclooxygenase inhibitors) Please refer to Summary of Product Characteristics (Sm. PC) for full list of adverse events Mepact Sm. PC, March 2013 http: //www. medicines. org. uk/emc/medicine/22763 EUCAN/MEP/2013 -10019 September 2013

Mepact EMA licensed indication Mepact is indicated in children, adolescents and young adults for the treatment of high-grade resectable non-metastatic osteosarcoma after macroscopically complete surgical resection. It is used in combination with post-operative multi-agent chemotherapy. Safety and efficacy have been assessed in studies of patients 2 to 30 years of age at initial diagnosis Mepact Sm. PC, March 2013 http: //www. medicines. org. uk/emc/medicine/22763 EUCAN/MEP/2013 -10019 September 2013

MEPACT® Prescribing Information Please refer to Summary of Product Characteristics (Sm. PC) before prescribing Insert Local Prescribing Information and replace TPI Gmb. H (below)with LOC company name © 2013 – Takeda Pharmaceuticals International Gmb. H EUCAN/MEP/2013 -10019 September 2013