IDIOPATHIC PULMONARY FIBROSIS MONITORING THE CLINICAL COURSE OF

IDIOPATHIC PULMONARY FIBROSIS

MONITORING THE CLINICAL COURSE OF DISEASE
IPF Prognosis At Time of Diagnosis (Baseline) Physiologic • Gas exchange • Desaturation • Pulmonary hemodynamics Radiologic • HRCT score • HRCT pattern Pathologic • Pattern • Extent Follow Up (Dynamic) Physiologic • Forced vital capacity • Gas exchange • Dyspnea

Clinical Predictors IPF Age: Poorer prognosis Median survival for patients younger than age 50 years was more compared with those between 50– 60 years, 6070 years and above 70 years 1 Sex : More common in men, but sex differences in survival have been inconsistent 1. Am J Respir Crit Care Med 2001; 164: 1171– 1181.

Clinical Predictors IPF Ethnicity: Limited data; earlier study suggested higher mortality of whites compared with blacks Smoking status: Non-smokers had a higher survival rate than former smokers and all smokers (current and former) Am J Respir Crit Care Med 2011; 183: 431– 440.

Clinical Predictors IPF Dyspnea: MRC chronic dyspnea score and the CRP dyspnea score at baseline and change in score at 6 and 12 months shown to be significant Physical findings: Digital clubbing significantly associated with reduced survival; BMI has shown an inverse association with survival Impact of comorbidities: pulmonary hypertension, emphysema, bronchogenic carcinoma, gastroesophageal reflux and significant coronary artery disease may also affect outcome in IPF Am J Respir Crit Care Med 2011; 183: 431– 440.

Physiologic Predictors Most consistently associated with prognosis are FVC, TLC, and DLCO Confounding by obstructive lung disease, especially emphysema Composite physiologic index : may account for emphysema in IPF combining FVC, DLCO, and FEV 1 into a formula that correlates better with disease extent by CT than any individual pulmonary function test ; may be a more accurate predictor of survival
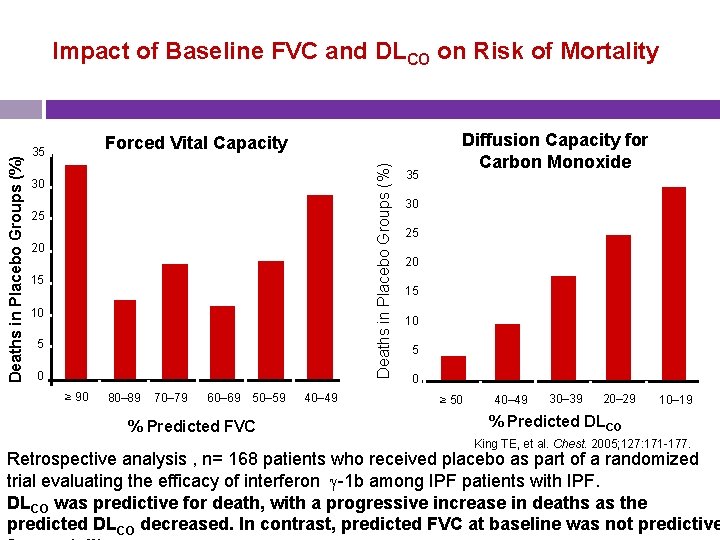
Forced Vital Capacity 35 Deaths in Placebo Groups (%) Impact of Baseline FVC and DLCO on Risk of Mortality 30 25 20 15 10 5 0 ≥ 90 80– 89 70– 79 60– 69 50– 59 % Predicted FVC 40– 49 35 Diffusion Capacity for Carbon Monoxide 30 25 20 15 10 5 0 ≥ 50 40– 49 30– 39 20– 29 10– 19 % Predicted DLCO King TE, et al. Chest. 2005; 127: 171 -177. Retrospective analysis , n= 168 patients who received placebo as part of a randomized trial evaluating the efficacy of interferon -1 b among IPF patients with IPF. DLCO was predictive for death, with a progressive increase in deaths as the predicted DLCO decreased. In contrast, predicted FVC at baseline was not predictive
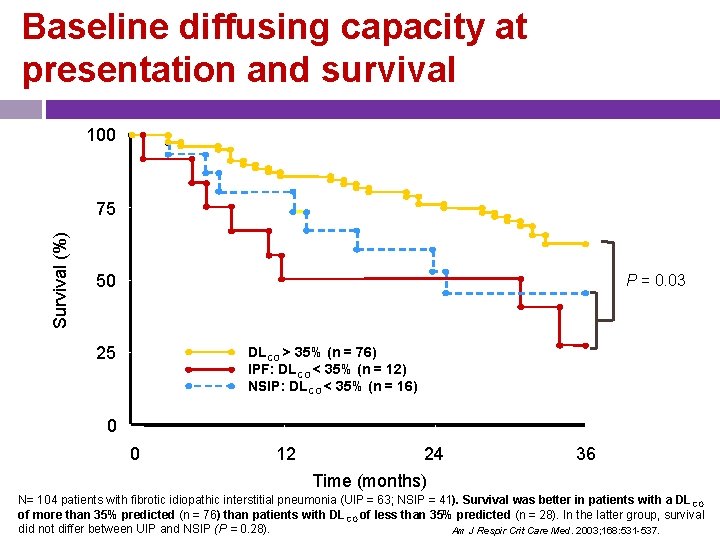
Baseline diffusing capacity at presentation and survival 100 Survival (%) 75 50 P = 0. 03 25 DLCO > 35% (n = 76) IPF: DLCO < 35% (n = 12) NSIP: DLCO < 35% (n = 16) 0 0 12 24 36 Time (months) N= 104 patients with fibrotic idiopathic interstitial pneumonia (UIP = 63; NSIP = 41). Survival was better in patients with a DL CO of more than 35% predicted (n = 76) than patients with DLCO of less than 35% predicted (n = 28). In the latter group, survival did not differ between UIP and NSIP (P = 0. 28). Am J Respir Crit Care Med. 2003; 168: 531 -537.

Survival in relation to the magnitude of serial change in FVC Studies have demonstrated that 6 - to 12 -month changes in FVC and DLCO are highly predictive of outcome Clinically significant changes in FVC and DLCO have typically been considered greater than 10% and greater than 15%, respectively Even marginal declines in FVC at 6 months (5– 10%) are associated with higher risk for mortality
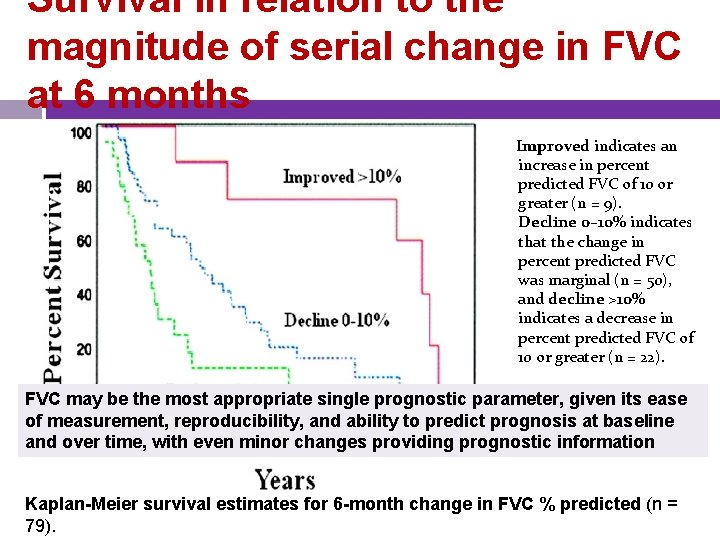
Survival in relation to the magnitude of serial change in FVC at 6 months Improved indicates an increase in percent predicted FVC of 10 or greater (n = 9). Decline 0– 10% indicates that the change in percent predicted FVC was marginal (n = 50), and decline >10% indicates a decrease in percent predicted FVC of 10 or greater (n = 22). FVC may be the most appropriate single prognostic parameter, given its ease of measurement, reproducibility, and ability to predict prognosis at baseline and over time, with even minor changes providing prognostic information Kaplan-Meier survival estimates for 6 -month change in FVC % predicted (n = 79).
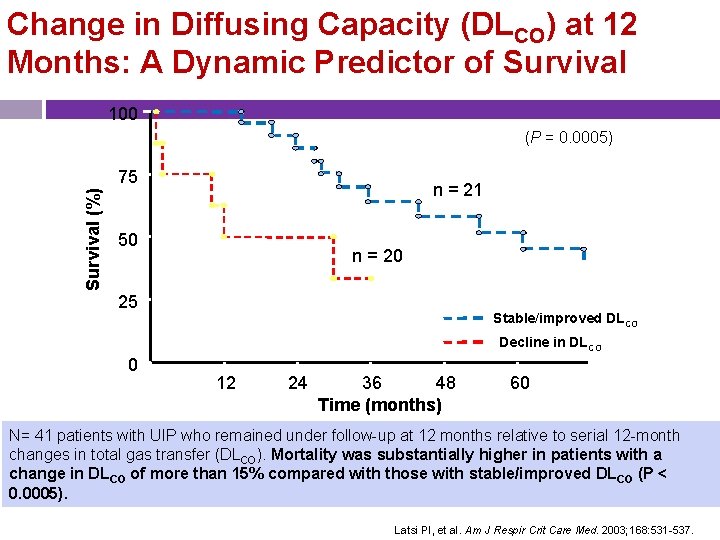
Change in Diffusing Capacity (DLCO) at 12 Months: A Dynamic Predictor of Survival 100 (P = 0. 0005) Survival (%) 75 n = 21 50 n = 20 25 Stable/improved DLCO Decline in DLCO 0 0 12 24 36 48 Time (months) 60 72 N= 41 patients with UIP who remained under follow-up at 12 months relative to serial 12 -month changes in total gas transfer (DLCO). Mortality was substantially higher in patients with a change in DLCO of more than 15% compared with those with stable/improved DLCO (P < 0. 0005). Latsi PI, et al. Am J Respir Crit Care Med. 2003; 168: 531 -537.
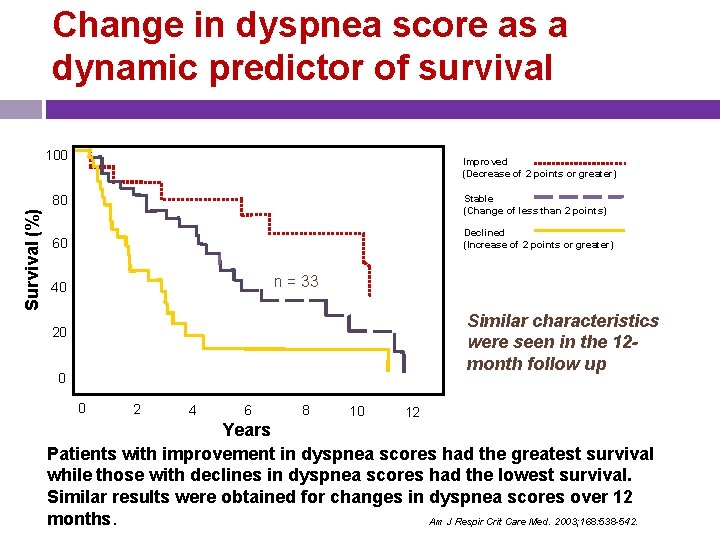
Change in dyspnea score as a dynamic predictor of survival 100 Improved (Decrease of 2 points or greater) Survival (%) 80 Stable (Change of less than 2 points) n = 15 Declined (Increase of 2 points or greater) 60 n = 33 40 20 Similar characteristics were seen in the 12 month follow up n = 31 0 0 2 4 6 8 10 12 Years Patients with improvement in dyspnea scores had the greatest survival while those with declines in dyspnea scores had the lowest survival. Similar results were obtained for changes in dyspnea scores over 12 months. Am J Respir Crit Care Med. 2003; 168: 538 -542.

Physiologic Predictors: Exercise testing Both distance walked and desaturation during the 6 MWT found to predict mortality, and a composite of the product of distance and desaturation in one study predicted mortality better than either measure alone 1 Change in 6 MWD is highly predictive of mortality (decline in 6 MWD>50 m over 24 wk is associated with a fourfold increase in risk of death at 1 year [P , 0. 001])2 Abnormal heart rate recovery after 1 minute of rest after the 6 MWT may be a novel and powerful predictor of mortality 3 1. Respir Med 2006; 100: 1734– 1741. 2. Am J Respir Crit Care Med 2010; 181: A 1103. 3. Chest 2009; 136: 841– 848.

Radiographic Predictors HRCT : overall extent of fibrosis has been consistently shown to correlate with disease severity parameters on pulmonary function tests and prognosis UIP pattern on HRCT: worse prognosis in patients with IPF compared with those with atypical HRCT findings Am J Respir Crit Care Med 2008; 177: 433– 439 Thorax 2003; 58: 143– 148.

Survival Time Is Influenced by Both Histologic and HRCT Appearance at Presentation HRCT Biopsy Median Survival Definite/probable UIP 2. 08 years Indeterminate UIP or definite/probable NSIP UIP 5. 76 years NSIP > 9 years Definite/probable NSIP worst prognosis Fibroblastic foci and their quantification has been shown to predict survival Flaherty KR, et al. Thorax. 2003; 58: 143 -148.

Biomarker Predictors Blood B-type natriuretic peptide was shown to be a better predictor of survival 1 Albumin levels negatively correlate with prognosis in many diseases and predict survival 2 Circulating fibrocyte levels are elevated in IPF and increase further during acute exacerbations 3 Higher Krebs von den Lungen-6 (KL-6 )levels may have reduced survival 4 High serum levels of both surfactant proteins A and D (SP-A and SP -D) - increased mortality 5 Serum chemokines CCL-18 levels 6 1. Respir Med 2009; 103: 180– 186. 2. Chest 2009; 135: 929– 935. 3. Respir Crit Care Med 2009; 179: 588– 594. 4. Respirology 2006; 11: 164– 168. 5. Am J Respir Crit Care Med 1999; 160: 1843– 1850. 6. Am J Respir Crit Care Med 2009; 179: 717– 723.

Biomarker Predictors BAL fluid Neutrophilia in BAL at baseline shown to independently predict 1 -year mortality 1 Matrix metalloproteinases (MMPs- 3, 7, 8, and 9) appear to be elevated in both blood and BAL fluid in patients with IPF 2 BAL fluid elevated CCL-2, 17, 22 produced by alveolar macrophages, SP-A and SP-D 4 shown to be a strong and independent predictor of mortality 3 1. Eur Respir J 2009; 33: 77– 84. 2. Respiration 2009; 78: 285– 292. 3. Chest 2008; 133: 226– 232. 4. Am J Respir Crit Care. Med 2000; 162: 1109– 1114.

Monitoring Patients with IPF Every 3 to 6 months monitor for : � Worsening Symptoms � Worsening Oxygenation (Spirometry, DLco , 6 MWT) � Progression of Fibrosis from Baseline on HRCT � Complications and Comorbidities Presence of any of the following changes is consistent with progressive disease: Progressive dyspnea (objectively assessed) Progressive, sustained decrease from baseline in absolute FVC and/or absolute DLCO (corrected for hemoglobin) Progression of fibrosis from baseline on HRCT Acute exacerbation Death from respiratory failure Am J Respir Crit Care Med 2011; 183: 788– 824.
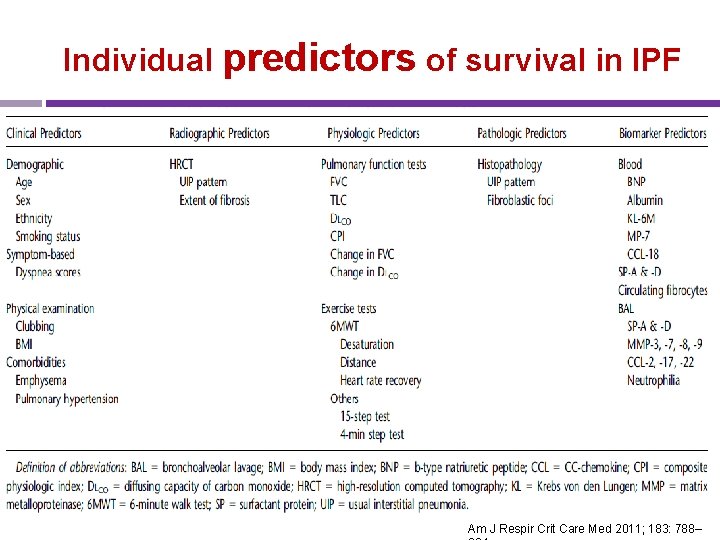
Individual predictors of survival in IPF Am J Respir Crit Care Med 2011; 183: 788–
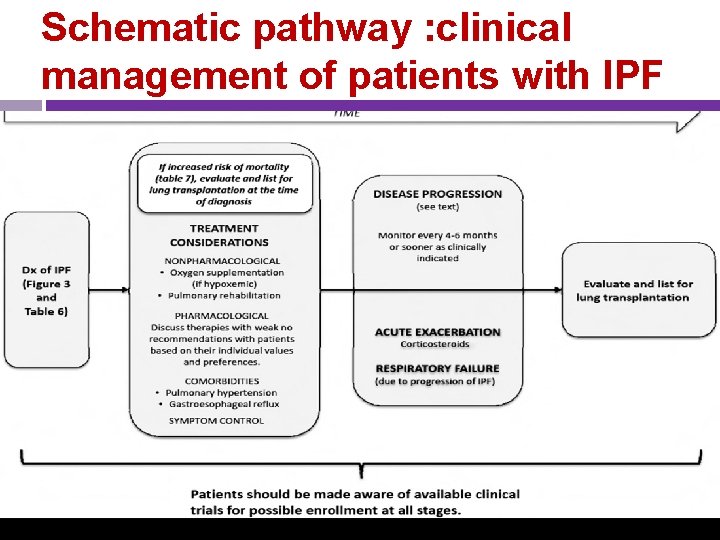
Schematic pathway : clinical management of patients with IPF Am J Respir Crit Care Med 2011; 183: 788– 824.

Summary IPF is a disease of multiple pathways ; combination therapy likely to be most successful. Clinical predictors useful in describing natural history of IPF, however disease progression remains difficult to predict FVC may be the most appropriate single prognostic parameter providing prognostic information Similar to the diagnosis of IPF, prognosis of individual patients will likely best be determined using a multidisciplinary approach

THE END
- Slides: 23