Nintedanib in Idiopathic Pulmonary Fibrosis IPF Milano 25

Nintedanib in Idiopathic Pulmonary Fibrosis (IPF) Milano, 25 Novembre 2015 Premio Galeno Patrizia Lessi, MD Therapeutic Area Head

2

What is IPF? q IPF is a progressive, fatal lung disease with a variable and unpredictable clinical course and a median survival time of only 2– 3 years from diagnosis 1 Risk factors for IPF It affects more men than women and most patients are over 501 Cause of the condition is unknown (idiopathic) but some risk factors have been identified 2, 3 IPF prevalence is increasing Smoking, environmental exposures, abnormal acid reflux and family history of the disease 2, 3 1. Pulmonary Fibrosis Foundation. Pulmonary Fibrosis Information Guide. pulmonaryfibrosis. org. Accessed March 2015 2. Raghu G, et al. Am J Respir Crit Care Med. 2011; 183: 788– 824 3. NHLBI, NIH. What Is Idiopathic Pulmonary Fibrosis? nhlbi. nih. gov/health-topics/ipf /. Accessed February 2015
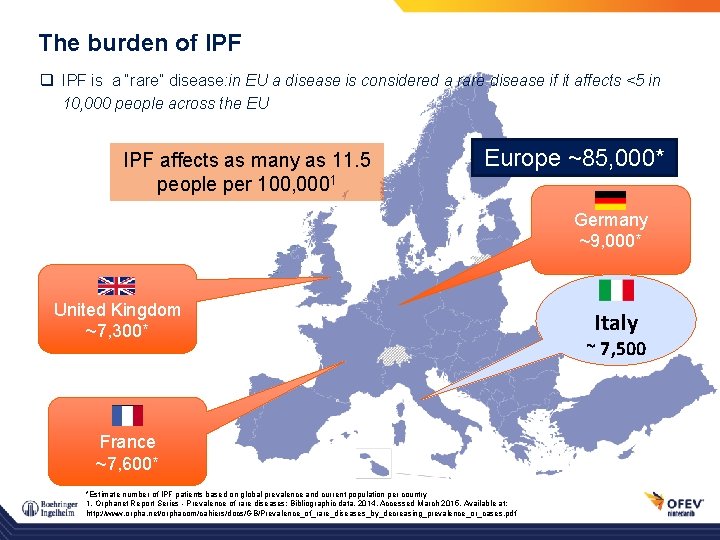
The burden of IPF q IPF is a “rare” disease: in EU a disease is considered a rare disease if it affects <5 in 10, 000 people across the EU IPF affects as many as 11. 5 people per 100, 0001 Europe ~85, 000* Germany ~9, 000* United Kingdom ~7, 300* France ~7, 600* *Estimate number of IPF patients based on global prevalence and current population per country 1. Orphanet Report Series - Prevalence of rare diseases: Bibliographic data, 2014. Accessed March 2015. Available at: http: //www. orpha. net/orphacom/cahiers/docs/GB/Prevalence_of_rare_diseases_by_decreasing_prevalence_or_cases. pdf Italy ~ 7, 500
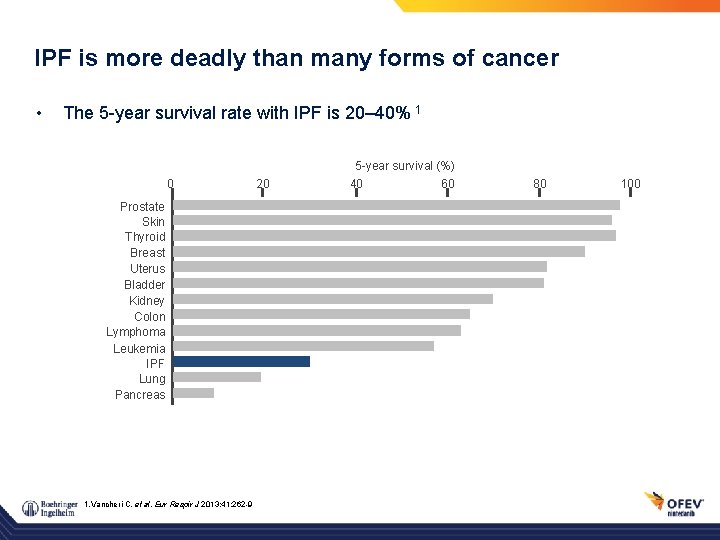
IPF is more deadly than many forms of cancer • The 5 -year survival rate with IPF is 20– 40% 1 5 -year survival (%) 0 Prostate Skin Thyroid Breast Uterus Bladder Kidney Colon Lymphoma Leukemia IPF Lung Pancreas 1. Vancheri C, et al. Eur Respir J 2013; 41: 262 -9 20 40 60 80 100

A fibrotic disease Fibrosis of lung tissue causes loss of lung function 1, 2 Lungs lose their ability to take in and transfer oxygen into the bloodstream 3 Those with IPF experience shortness of breath, cough and often have difficulty with everyday physical activities 4 1. Selman M, et al. Ann Intern Med. 2001; 134: 136– 51 2. NHLBI, NIH. What Is Idiopathic Pulmonary Fibrosis? nhlbi. nih. gov/health-topics/ipf # Accessed March 2015 3. Collard H, et al. Am J Respir Crit Care Med. 2007; 176: 636– 643 4. Pulmonary Fibrosis Foundation. Symptoms. pulmonaryfibrosis. org/life-with-pf/about-pf. Accessed March 2015 7

Diagnosis • More than 80% of patients with IPF have a distinctive, Velcro-like crackle, easily detected through a stethoscope 2, 3, 4 • High resolution CT scan: honeycombing 1 • Lung biopsy (in some cases)1 Adapted from Meltzer & Noble Orphanet Journal of Rare Diseases 1. Raghu G, et al. Am J Respir. Crit Care Med. 2011; 183: 788– 824 2. Borchers A et al. Clin Rev Allergy Immunol 2011; 40: 117– 34. 3. Ley B, et al. AJRCCM 2011; 183: 431– 40. 4. Cottin V, et al. Eur Respir J. 2012 40: 519– 512

Nintedanib in Idiopathic Pulmonary Fibrosis Mechanism of Action and Clinical Benefits
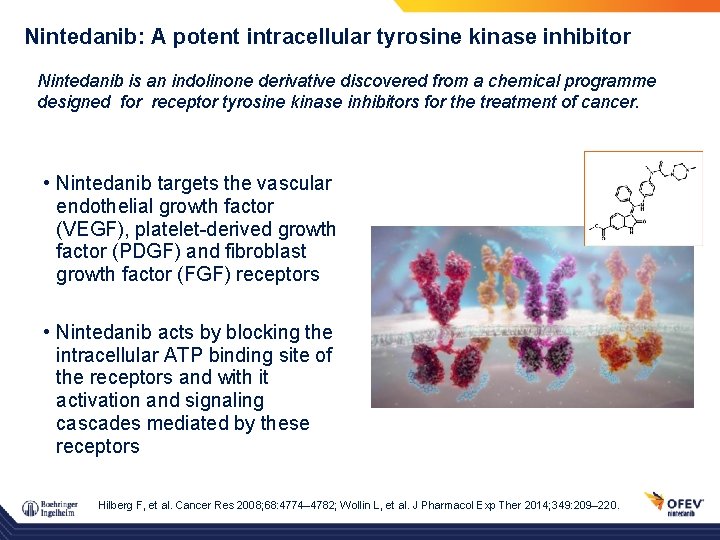
Nintedanib: A potent intracellular tyrosine kinase inhibitor Nintedanib is an indolinone derivative discovered from a chemical programme designed for receptor tyrosine kinase inhibitors for the treatment of cancer. • Nintedanib targets the vascular endothelial growth factor (VEGF), platelet-derived growth factor (PDGF) and fibroblast growth factor (FGF) receptors • Nintedanib acts by blocking the intracellular ATP binding site of the receptors and with it activation and signaling cascades mediated by these receptors Hilberg F, et al. Cancer Res 2008; 68: 4774– 4782; Wollin L, et al. J Pharmacol Exp Ther 2014; 349: 209– 220.
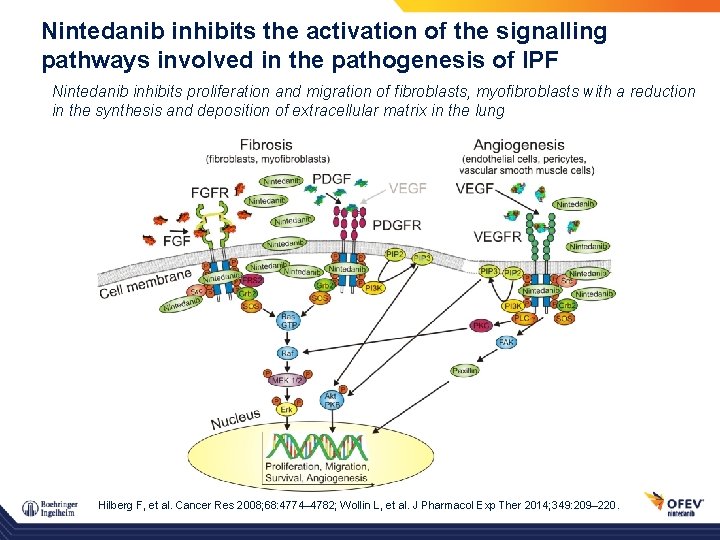
Nintedanib inhibits the activation of the signalling pathways involved in the pathogenesis of IPF Nintedanib inhibits proliferation and migration of fibroblasts, myofibroblasts with a reduction in the synthesis and deposition of extracellular matrix in the lung Hilberg F, et al. Cancer Res 2008; 68: 4774– 4782; Wollin L, et al. J Pharmacol Exp Ther 2014; 349: 209– 220.
The OFEV® (nintedanib) story BI first investigates nintedanib Nintedanib enters Phase III IPF clinical trials 2007 2011 1998 2014 Nintedanib enters Phase II IPF clinical trials INPULSIS® data presented at ATS FDA approves OFEV® in the US 2014 OFEV® approved in the EU 2015

16

INPULSIS® trials (2 phase III trials): 1 year 1066 patients Summary of efficacy findings from the INPULSIS® trials Nintedanib 150 mg bid is the first treatment for IPF that has consistently been shown to slow disease progression in all subgroup patients with IPF independently of the severity of lung function impairment by significantly reducing the annual decline in lung function by approximately 50% This clinically relevant effect of nintedanib on disease progression was supported by: • A significant reduction in the risk of adjudicated confirmed exacerbations of 68% in a pre-specified sensitivity analysis • • • A numerical reduction in all-cause mortality of 30% Consistent results across a range of lung function endpoints A manageable side-effect profile

Nintedanib a major contribution to patient care and a new therapeutic approach for IPF 18
- Slides: 14