Field Trip 1 Hocking Hills State Park Saturday














- Slides: 19

Field Trip #1: Hocking Hills State Park Saturday, October 2 nd

MINERALS: The Building Blocks of Rocks

Definition of a Mineral • A Mineral is a naturally occurring, inorganic, solid, crystalline substance, with definite physical and chemical properties. • Inorganic – not composed of Carbon – Hydrogen molecules (usually part of living tissue) • Crystalline – atoms have a structured arrangement.

CRYSTAL SHAPES

How Do Crystals Form? Remember Atomic Structure

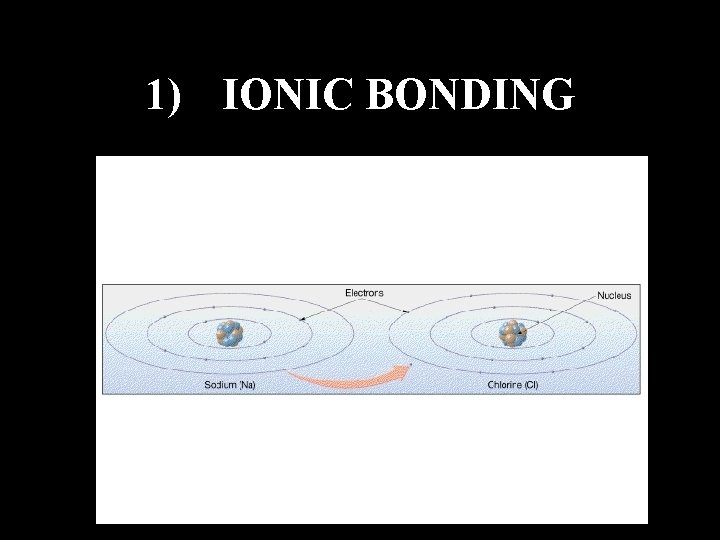
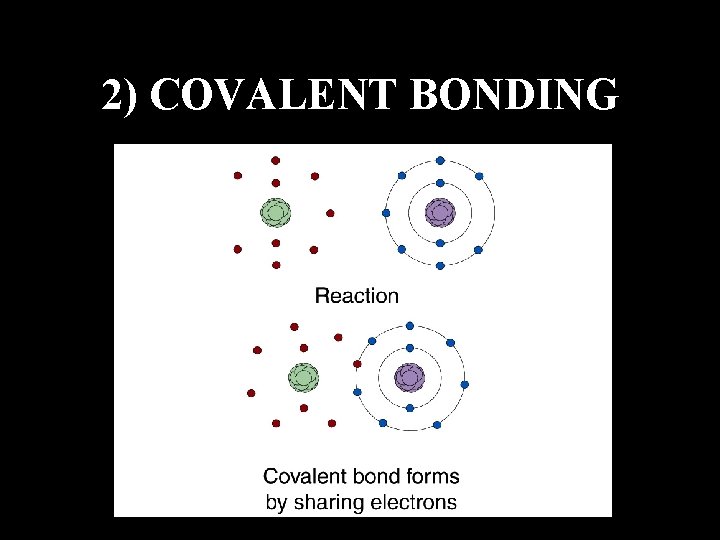
ATOMS DO NOT WANT TO BE NEUTRAL THEY WANT TO BE STABLE § Stable Means Having Their Outer Shell Of Electrons Full To Capacity § This Can Happen By Losing Or Gaining Electrons to Other Atoms §This Process causes Atoms to Join Together: A Process Called Bonding

1) IONIC BONDING

2) COVALENT BONDING

OTHER BONDS 3) Metallic – Electrons act as a mobile cloud that moves from atom to atom. Accounts for excellent electrical conductivity of metals Example: Copper 4) van der Waals – Very weak Bond due to slight polarity of charge on atoms

MINERAL PROPERTIES: HARDNESS

MINERAL PROPERTIES: CLEAVAGE Breakage along planes of crystal weakness

MINERAL PROPERTIES: Other Properties §Color §Streak – color of powdered mineral §Luster – the way light reflects of a mineral §Specific Gravity – Weight of Mineral Weight of same volume of water §Fracture – breakage through crystal §Reactivity – Reacts with Acid?

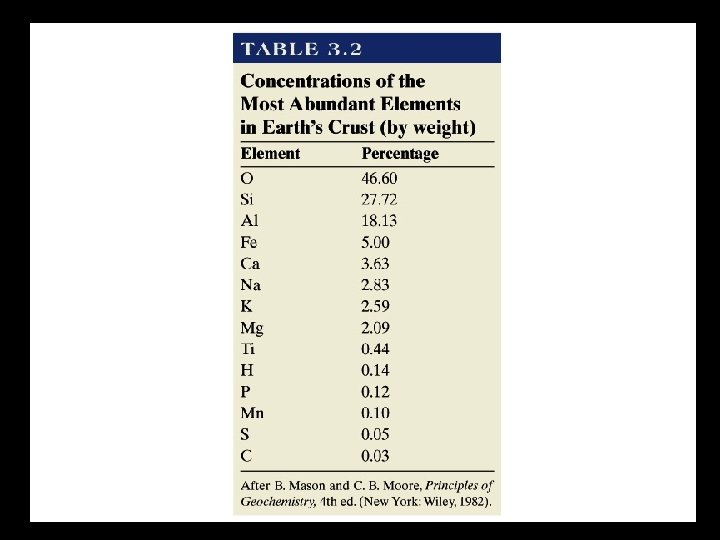
MAJOR MINERAL GROUPS What are the most common minerals in the Crust? 1) What are the most common elements?


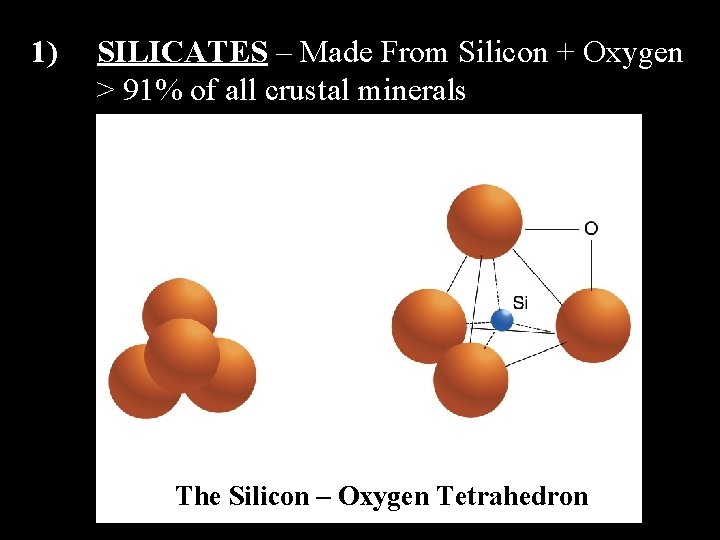
1) SILICATES – Made From Silicon + Oxygen > 91% of all crustal minerals The Silicon – Oxygen Tetrahedron

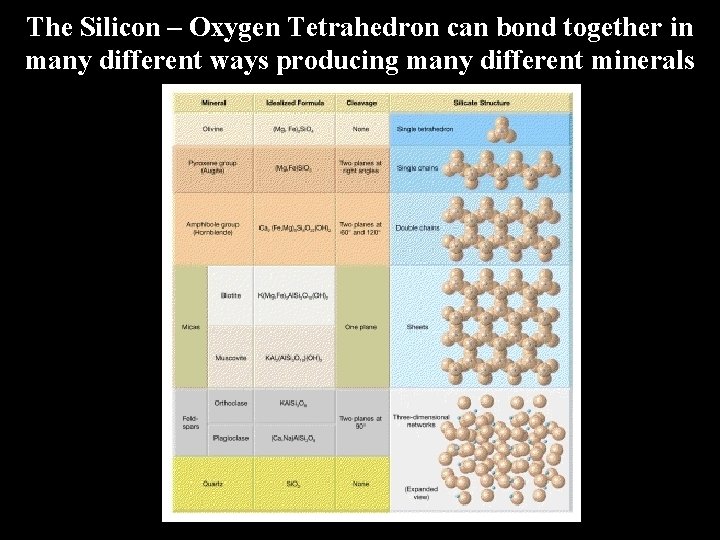
The Silicon – Oxygen Tetrahedron can bond together in many different ways producing many different minerals

Other Important Mineral Groups 2) Carbonates – based on Carbonate ion (CO 3) Calcite - Ca CO 3, Dolomite - Ca. Mg (CO 3)2 Main component of the rocks limestone & dolostone. 3) Oxides – usually metals + oxygen Hematite - Fe 2 O 3 (Iron Oxide or Rust), Corundum - Al 2 O 3 (Aluminum Oxide or Ruby). Excellent source of ore metals

Other Important Mineral Groups 4) Sulfides – based on sulfide ion (S 2 -) Pyrite - Fe. S 2 (“Fool’s Gold”) Excellent source of ore metals 5) Sulfates – based on sulfate ion (SO 4)Gypsum - Ca. SO 4 (Drywall) Used in the construction industry

So, why study minerals? 1) Building Blocks of Rocks – what the Earth is made of. 2) Important Economically – Industry & Agriculture. 3) Can be incredibly beautiful!!