Chemistry for Changing Times Thirteenth Edition Lecture Outlines

















- Slides: 25

Chemistry for Changing Times, Thirteenth Edition Lecture Outlines Chapter 6 Gases, Liquids, Solids, and Intermolecular Forces John Singer, Jackson Community College © 2013 Pearson Education, Inc.

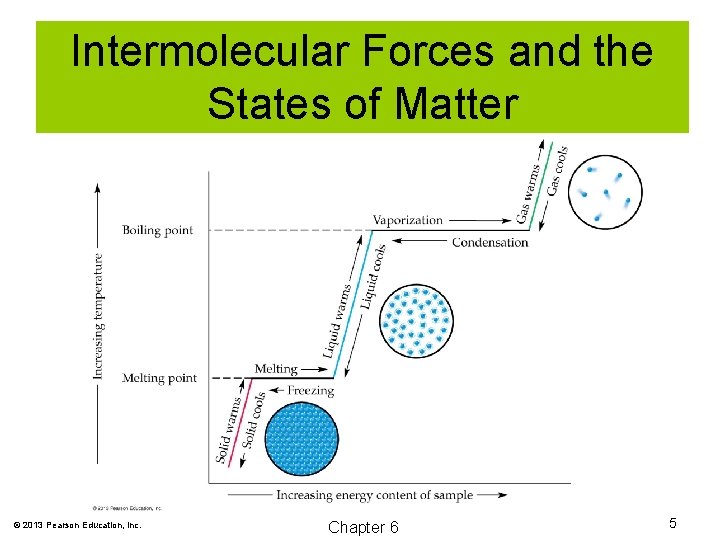
Intermolecular Forces and the States of Matter Solids: The particles of a solid have fixed positions and exhibit motions of vibration. Liquids: The particles of a liquid are free to move within the confines of the liquid. Gas: The particles of a gas are far apart and move randomly and rapidly. © 2013 Pearson Education, Inc. Chapter 6 2

Intermolecular Forces and the States of Matter Melting point: The temperature at which a solid becomes a liquid. Vaporization: The process of a liquid becoming a gas. Boiling point: The temperature at which the particles of a liquid escape and become a gas. © 2013 Pearson Education, Inc. Chapter 6 3

Intermolecular Forces and the States of Matter Condensation: The process by which a gas becomes a liquid. Freezing: The process by which a liquid becomes a solid. This occurs at the freezing point, which is the same as the melting point. Sublimation: When a solid changes directly from the solid to the gaseous state. © 2013 Pearson Education, Inc. Chapter 6 4

Intermolecular Forces and the States of Matter © 2013 Pearson Education, Inc. Chapter 6 5

Intermolecular Forces and the States of Matter Ionic bonds: Ionic bonds are the strongest of forces that hold matter in the condensed states. © 2013 Pearson Education, Inc. Chapter 6 6

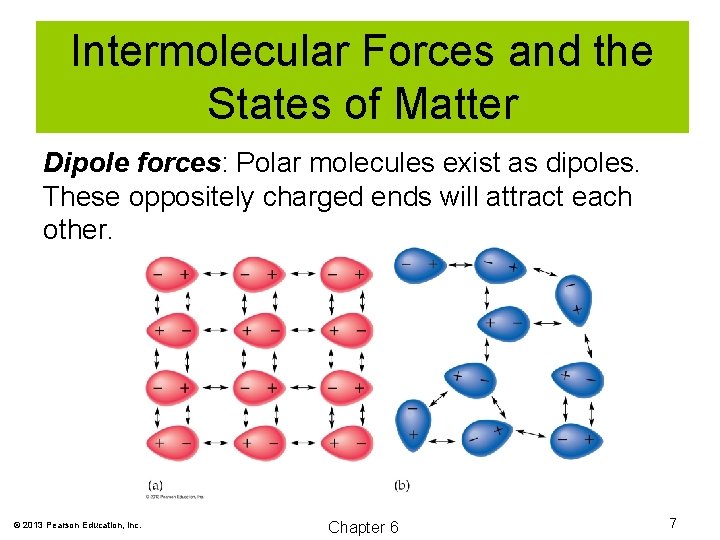
Intermolecular Forces and the States of Matter Dipole forces: Polar molecules exist as dipoles. These oppositely charged ends will attract each other. © 2013 Pearson Education, Inc. Chapter 6 7

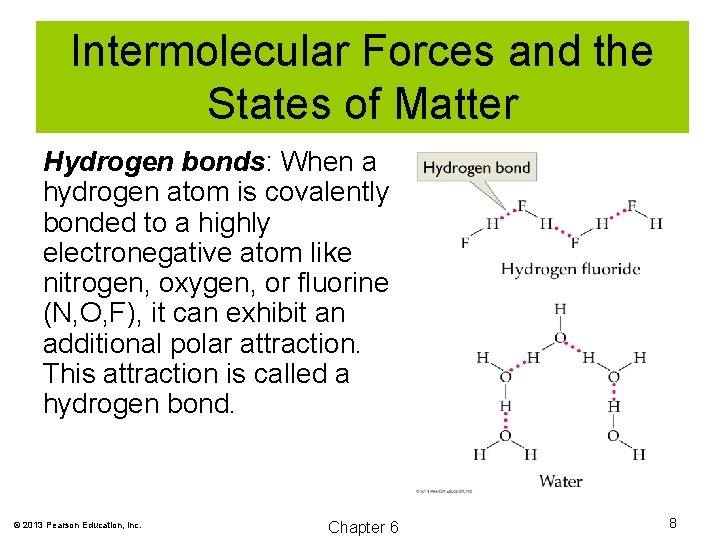
Intermolecular Forces and the States of Matter Hydrogen bonds: When a hydrogen atom is covalently bonded to a highly electronegative atom like nitrogen, oxygen, or fluorine (N, O, F), it can exhibit an additional polar attraction. This attraction is called a hydrogen bond. © 2013 Pearson Education, Inc. Chapter 6 8

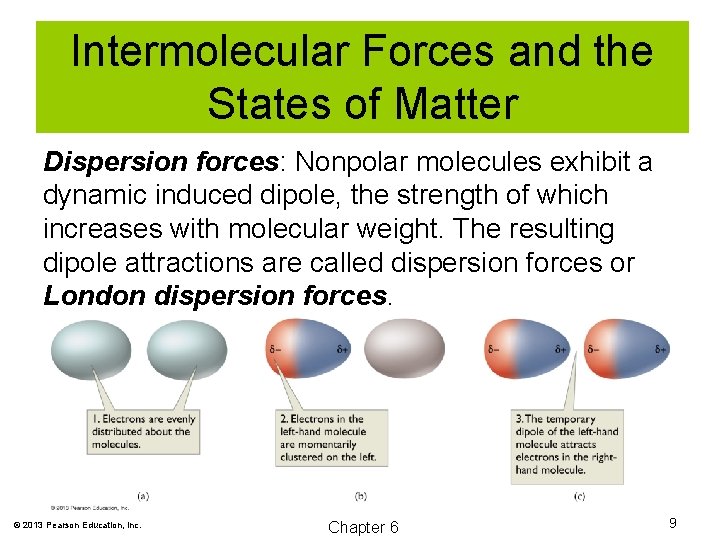
Intermolecular Forces and the States of Matter Dispersion forces: Nonpolar molecules exhibit a dynamic induced dipole, the strength of which increases with molecular weight. The resulting dipole attractions are called dispersion forces or London dispersion forces. © 2013 Pearson Education, Inc. Chapter 6 9

Intermolecular Forces and the States of Matter Solution: A homogeneous mixture of two or more substances. Solute: A substance that is dispersed in a solution. Solvent: The substance doing the dissolving, usually present in greatest quantity. © 2013 Pearson Education, Inc. Chapter 6 10

Intermolecular Forces and the States of Matter © 2013 Pearson Education, Inc. Chapter 6 11

Intermolecular Forces and the States of Matter “Like dissolves like”: Solutions form most readily when both the solute and solvent have similar intermolecular forces. © 2013 Pearson Education, Inc. Chapter 6 12

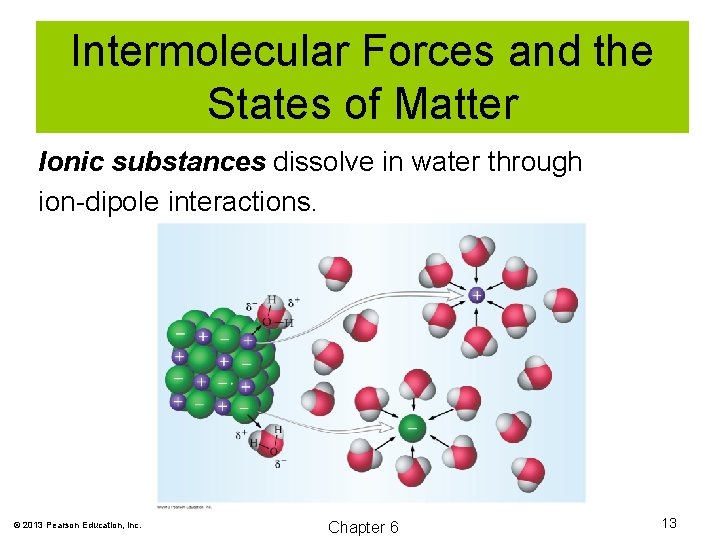
Intermolecular Forces and the States of Matter Ionic substances dissolve in water through ion-dipole interactions. © 2013 Pearson Education, Inc. Chapter 6 13

The Gas Laws Kinetic Molecular Theory of a Gas Postulates: 1. The particles of a gas are in rapid constant motion. 2. The particles of a gas are tiny compared to the distance between them. 3. There is little attraction between the particles of a gas. 4. Collisions between gas molecules are perfectly elastic. 5. Temperature is a measure of the average kinetic energy of gas molecules. © 2013 Pearson Education, Inc. Chapter 6 14

The Gas Laws © 2013 Pearson Education, Inc. Chapter 6 15

The Gas Laws Boyle’s law: At constant temperature, the volume of a gas is inversely proportional to its pressure. V α 1/P V = a/P PV = a V 1 P 1 = V 2 P 2 © 2013 Pearson Education, Inc. Chapter 6 16

The Gas Laws Boyle’s law: At constant temperature, the volume of a gas is inversely proportional to its pressure. © 2013 Pearson Education, Inc. Chapter 6 17

The Gas Laws Boyle’s law: At constant temperature, the volume of a gas is inversely proportional to its pressure. © 2013 Pearson Education, Inc. Chapter 6 18

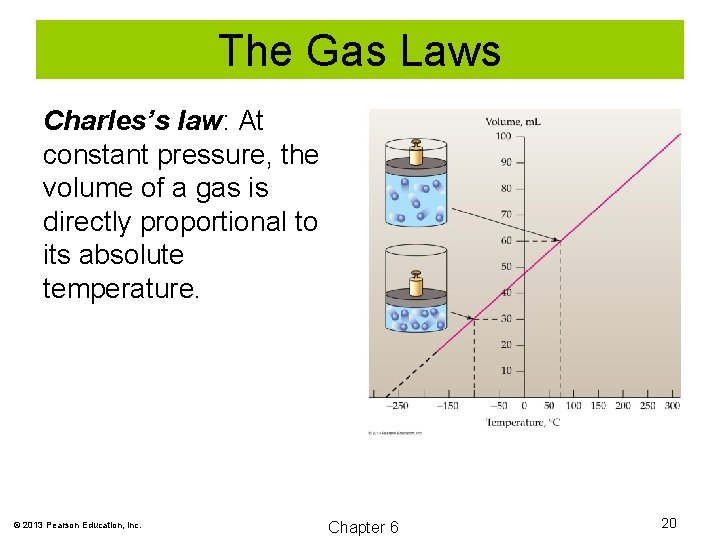
The Gas Laws Charles’s law: At constant pressure, the volume of a gas is directly proportional to its absolute temperature. VαT V = b. T V/T = b V 1/T 1 = V 2/T 2 © 2013 Pearson Education, Inc. Chapter 6 19

The Gas Laws Charles’s law: At constant pressure, the volume of a gas is directly proportional to its absolute temperature. © 2013 Pearson Education, Inc. Chapter 6 20

The Gas Laws Charles’s Law © 2013 Pearson Education, Inc. Chapter 6 21

The Gas Laws Avogadro’s law: At fixed temperature and pressure, the volume of a gas is directly proportional to the amount of gas. Vαn V = cn V/n = c V 1/n 1 = V 2/n 2 © 2013 Pearson Education, Inc. Chapter 6 22

The Gas Laws Standard temperature and pressure: Standard temperature = 0 o. C Standard pressure = 1 atm A mole of any gas at STP occupies 22. 4 L © 2013 Pearson Education, Inc. Chapter 6 23

The Gas Laws Combined gas law: P 1 V 1 T 1 © 2013 Pearson Education, Inc. = Chapter 6 P 2 V 2 T 2 24

The Gas Laws Ideal gas law: PV = n. RT R = 0. 0821 © 2013 Pearson Education, Inc. Chapter 6 25