Bioanalyse commission SOFAQ elution plate extraction after Capillary

Bioanalyse commission SOFAQ µelution plate extraction after Capillary µsampling 16/03/2016 (CMS) p 1 S. Couffin
Introduction 1. Why evaluate micro-sampling? 2. What benefits brings micro-sampling? 3. What stage requires micro-sampling? 4. Comparison between conventional procedure and CMS 5. Pierre Fabre experiment 6. Which parameters will be evaluate? 7. Questions and Discussion p 2

Why evaluate CMS? • Ethics reason for rodent studies (early or development). Blood sampling volumes can challenging in the preclinical setting (Total blood volume of 15% in any 28 day period). Decrease animal numbers. • Performed the PK or TK evaluation in rodent studies on the same animal • Decrease volume of sampling for PK or TK evaluation in order to add other test (for example research of biomarkers, early metabolism…) • Decrease quantity of reference item used for administration. • Toxicological effects can be correlated to exposure data in the same animal • To replace DBS sampling (not accepted by FDA) Animal usage Scientific Quality p 3

What benefits brings micro-sampling? • Causes less distress to animals during sampling • Only one technician needed for blood sampling • Lower costs - Fewer animal required per study & less test item used • More TK sampling time-points feasible • TK samples taken from main group animals – improved data quality 4 p 4
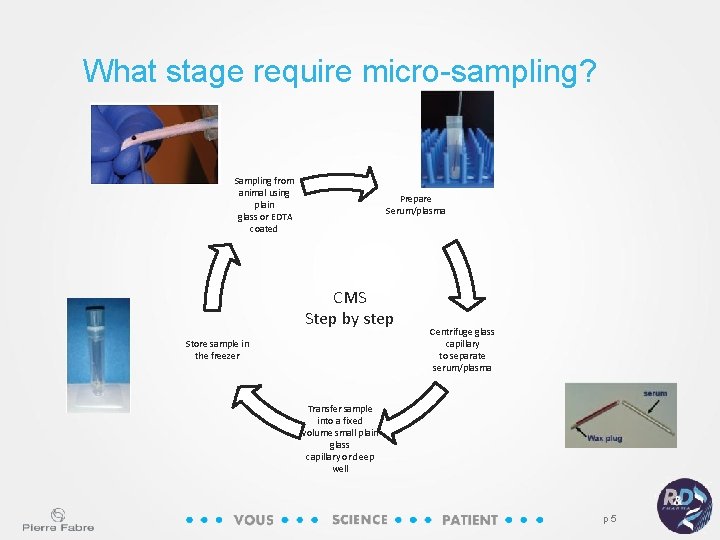
What stage require micro-sampling? Sampling from animal using plain glass or EDTA coated Prepare Serum/plasma CMS Step by step Store sample in the freezer Centrifuge glass capillary to separate serum/plasma Transfer sample into a fixed volume small plain glass capillary or deep well p 5
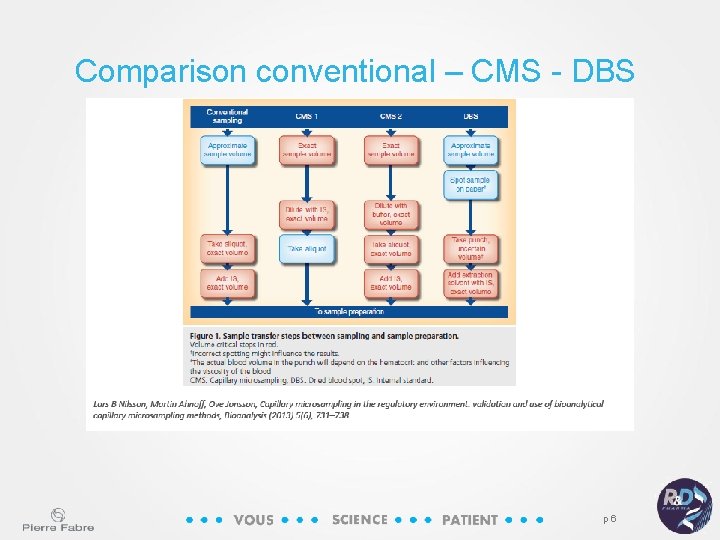
Comparison conventional – CMS - DBS p 6

Pierre Fabre Experiment 1. Comparison between conventional procedure and CMS for compound A 2. PK study in mouse plasma for compound A 3. PK study in mouse blood for Vinflunine and its metabolite 4. Development of method for different compound in rat plasma, CSF and brain p 7

Comparison between conventional procedure and CMS for compound A • Same point for the same rat • Three rat per time points • Mean results reported p 8

Extraction procedure Compound A Prepare sample 50 µL of plasma or 8 µL of plasma 2 µL of labelled internal standard at 10 ng/µL or at 1. 6 ng/µL 50 µL of H 3 PO 4 at 4% Shake plate around 2 min Condition and equilibrate Oasis µelution HLB 96 deep well 200 µL of methanol 200 µL of water Load Sample Wash 200 µL of a mixture of methanol /water 5/95 Elute 1: 50 µL of a mixture of ACN/Me. OH 40/60, 1% formic acid Elute 2: 50 µl water p 9

Mass spectrometer parameters Mode Positive Electro spray : 2 KV Source temperature: 150°C Desolvatation temperature: 500°C Cone gas flow: 150 L/Hr Desolvatation gas flow: 1000 L/Hr p 10

Chromatography parameters Pump UPLC Aquity Autosampler UPLC Aquity Column: Acquity UPLC BEH C 18 50 x 2. 1 mm, 1. 7 µm Aqueous mobile phase (A): Acetonitrile 0. 1% TFA Organic mobile phase (B): Formiate buffer 10 m. M 0. 1% TFA Flow rate: 0. 2 m. L/min Isocratic mode Strong wash solvent: Acetonitrile Weak wash solvent: Acetonitrile / Water 30/70, 0. 1% TFA Column temperature: 40°C Sample temperature: 10°C Injection volume: 8 µL p 11
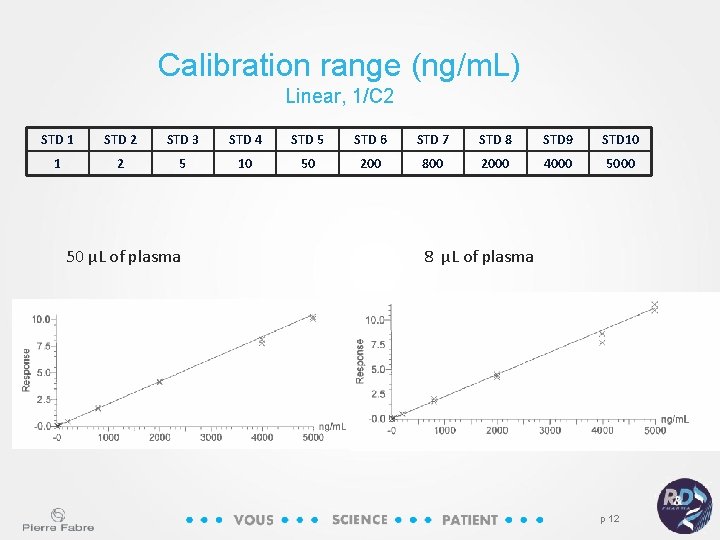
Calibration range (ng/m. L) Linear, 1/C 2 STD 1 STD 2 STD 3 STD 4 STD 5 STD 6 STD 7 STD 8 STD 9 STD 10 1 2 5 10 50 200 800 2000 4000 50 µL of plasma 8 µL of plasma p 12
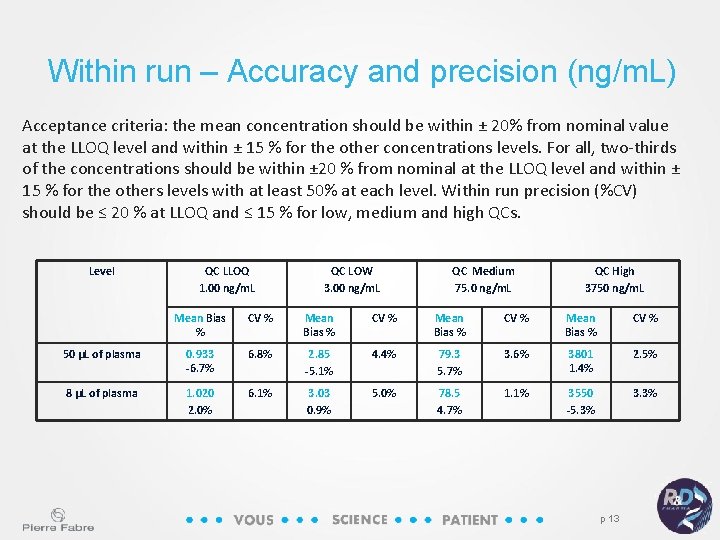
Within run – Accuracy and precision (ng/m. L) Acceptance criteria: the mean concentration should be within ± 20% from nominal value at the LLOQ level and within ± 15 % for the other concentrations levels. For all, two-thirds of the concentrations should be within ± 20 % from nominal at the LLOQ level and within ± 15 % for the others levels with at least 50% at each level. Within run precision (%CV) should be ≤ 20 % at LLOQ and ≤ 15 % for low, medium and high QCs. Level QC LLOQ 1. 00 ng/m. L QC LOW 3. 00 ng/m. L QC Medium 75. 0 ng/m. L QC High 3750 ng/m. L Mean Bias % CV % 50 µL of plasma 0. 933 -6. 7% 6. 8% 2. 85 -5. 1% 4. 4% 79. 3 5. 7% 3. 6% 3801 1. 4% 2. 5% 8 µL of plasma 1. 020 2. 0% 6. 1% 3. 03 0. 9% 5. 0% 78. 5 4. 7% 1. 1% 3550 -5. 3% 3. 3% p 13
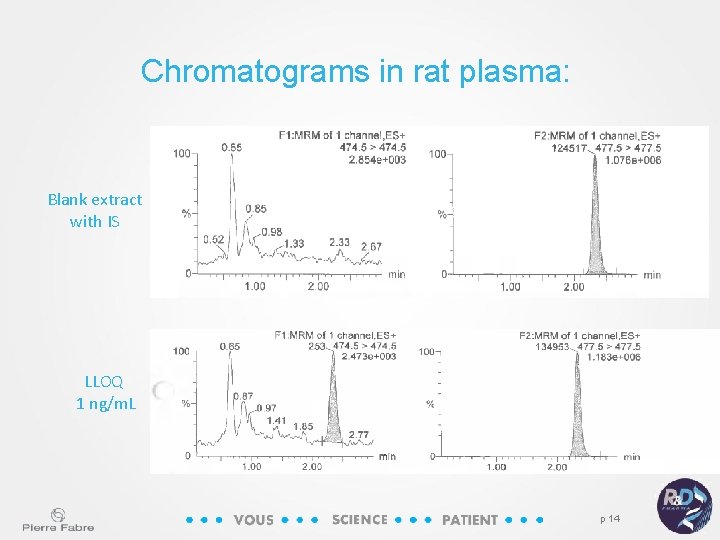
Chromatograms in rat plasma: Blank extract with IS LLOQ 1 ng/m. L p 14
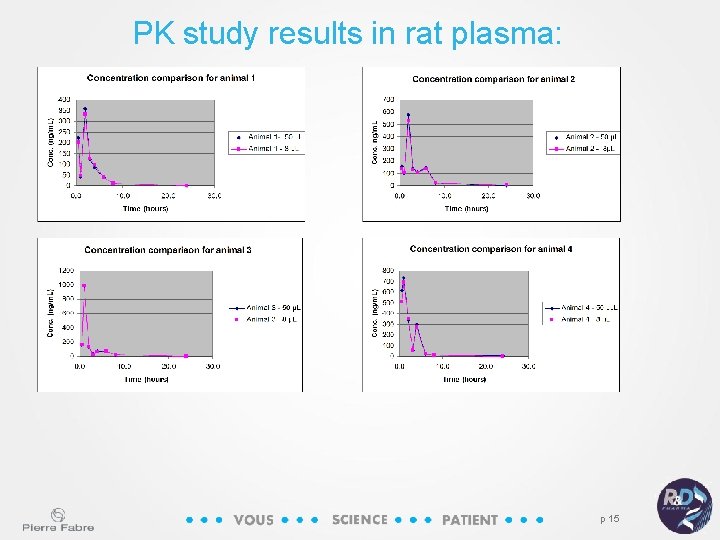
PK study results in rat plasma: p 15

Conclusion after comparison There was no results difference observed when we compared extraction with 50 µL of plasma versus 8 µL of plasma on µelution plate Oasis HLB. Therefore it is possible to use assay sampling from CMS with extraction on SPE. Results of ISR performed for compound A in different studies in rat with 4 µL of assay sample Bias FDA Bias EMEA (D 2 -D 1) D 1/MD 1 D 2 D 2/MD 1 D 2 (%) (%) Number of ISR out of acceptance criteria: 0 0 2 Total number of ISR: 75 75 75 % of ISR within acceptandce criteria: 100. 0 97. 3 p 16
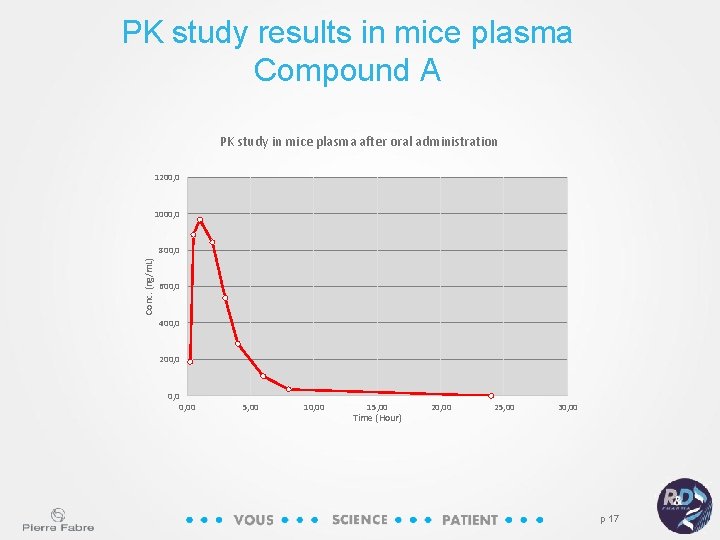
PK study results in mice plasma Compound A PK study in mice plasma after oral administration 1200, 0 1000, 0 Conc. (ng/m. L) 800, 0 600, 0 400, 0 200, 00 5, 00 10, 00 15, 00 Time (Hour) 20, 00 25, 00 30, 00 p 17

PK study in mouse blood for Vinflunine and its metabolite 4 -O-deacetyl vinflunine • The aim of this study is to decrease blood sampling. In the initial method, assay sampling was 200 µL of blood (1 mouse was used for 1 sample). • The objective is to have an bio analytical assay sampling at 25 µL and Blood sampling at 50 µL. • Three mouse for each time point – one mouse three samples • PK Mean results reported (PO: 5, 10 and 30 mg/kg) p 18

Extraction procedure vinflunine and its metabolite Prepare sample 25 µL blood 2 µL of internal standard 100 µL water with 2% of formic acid Shake and centrifuge at 14 000 rpm Condition and equilibrate Oasis µelution HLB 96 deep well 200 µL of methanol 200 µL of water Load Sample Wash 200 µL of water 200 µL of a mixture of methanol /water 5/95 Elute with 40 µL of acetonitrile Elute Dilute with 60 µL of 5 m. M ammonium acetate buffer 0. 1% HCOOH p 19

Mass spectrometer parameters Mode Positive Electro spray : 2. 5 KV Source temperature: 150°C Desolvatation temperature: 650°C Cone gas flow: 150 L/Hr Desolvatation gas flow: 1000 L/Hr p 20

Chromatography parameters Pump UPLC Aquity) Autosampler UPLC Aquity Class (FTN) Column: Xselect HSS CN Waters 50 x 2. 1 mm, 2. 5 µm Aqueous mobile phase (A): 5 m. M acetate ammonium buffer 0. 1% HCOOH Organic mobile phase (B): Acetonitrile Flow rate: 0. 3 m. L/min Isocratic mode: 60/40 (v/v) Column temperature: 30°C Sample temperature: 10°C Injection volume: 4 µL p 21
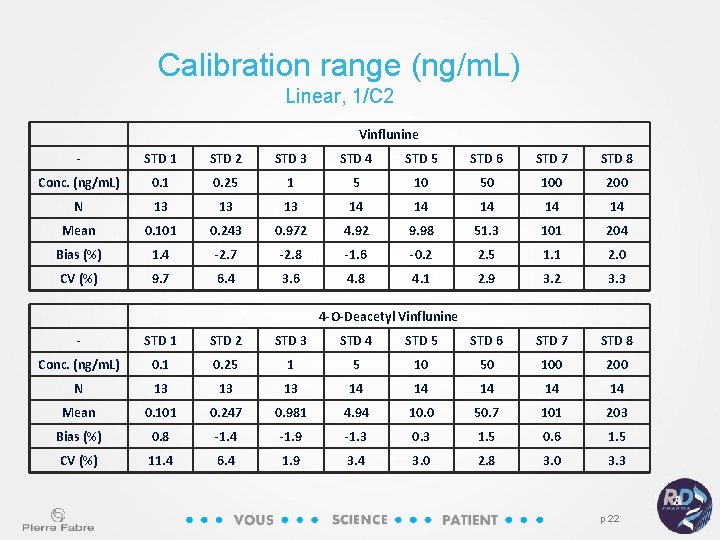
Calibration range (ng/m. L) Linear, 1/C 2 Vinflunine - STD 1 STD 2 STD 3 STD 4 STD 5 STD 6 STD 7 STD 8 Conc. (ng/m. L) 0. 1 0. 25 1 5 10 50 100 200 N 13 13 13 14 14 14 Mean 0. 101 0. 243 0. 972 4. 92 9. 98 51. 3 101 204 Bias (%) 1. 4 -2. 7 -2. 8 -1. 6 -0. 2 2. 5 1. 1 2. 0 CV (%) 9. 7 6. 4 3. 6 4. 8 4. 1 2. 9 3. 2 3. 3 4 -O-Deacetyl Vinflunine - STD 1 STD 2 STD 3 STD 4 STD 5 STD 6 STD 7 STD 8 Conc. (ng/m. L) 0. 1 0. 25 1 5 10 50 100 200 N 13 13 13 14 14 14 Mean 0. 101 0. 247 0. 981 4. 94 10. 0 50. 7 101 203 Bias (%) 0. 8 -1. 4 -1. 9 -1. 3 0. 3 1. 5 0. 6 1. 5 CV (%) 11. 4 6. 4 1. 9 3. 4 3. 0 2. 8 3. 0 3. 3 p 22
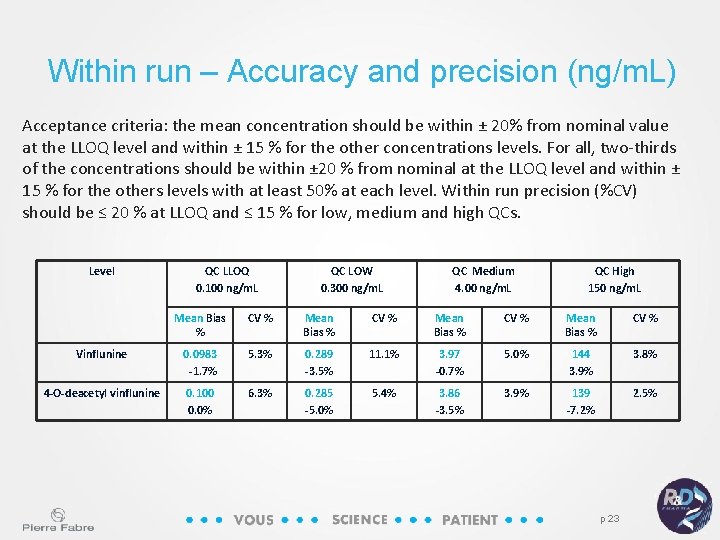
Within run – Accuracy and precision (ng/m. L) Acceptance criteria: the mean concentration should be within ± 20% from nominal value at the LLOQ level and within ± 15 % for the other concentrations levels. For all, two-thirds of the concentrations should be within ± 20 % from nominal at the LLOQ level and within ± 15 % for the others levels with at least 50% at each level. Within run precision (%CV) should be ≤ 20 % at LLOQ and ≤ 15 % for low, medium and high QCs. Level QC LLOQ 0. 100 ng/m. L QC LOW 0. 300 ng/m. L QC Medium 4. 00 ng/m. L QC High 150 ng/m. L Mean Bias % CV % Vinflunine 0. 0983 -1. 7% 5. 3% 0. 289 -3. 5% 11. 1% 3. 97 -0. 7% 5. 0% 144 3. 9% 3. 8% 4 -O-deacetyl vinflunine 0. 100 0. 0% 6. 3% 0. 285 -5. 0% 5. 4% 3. 86 -3. 5% 3. 9% 139 -7. 2% 2. 5% p 23
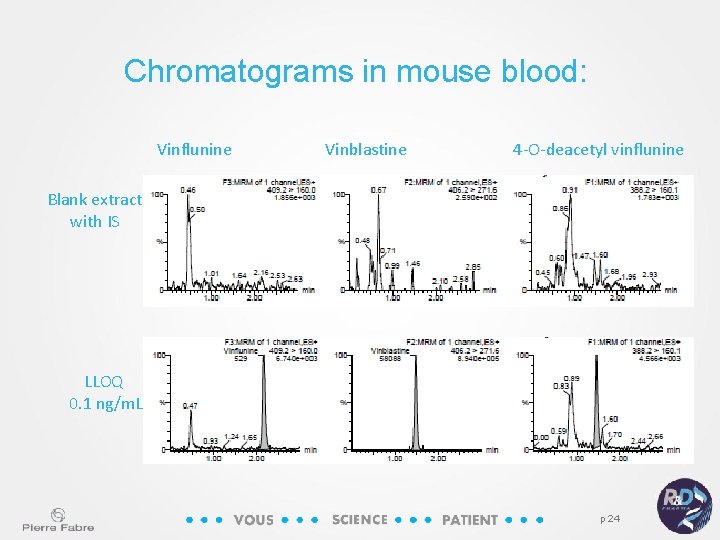
Chromatograms in mouse blood: Vinflunine Vinblastine 4 -O-deacetyl vinflunine Blank extract with IS LLOQ 0. 1 ng/m. L p 24
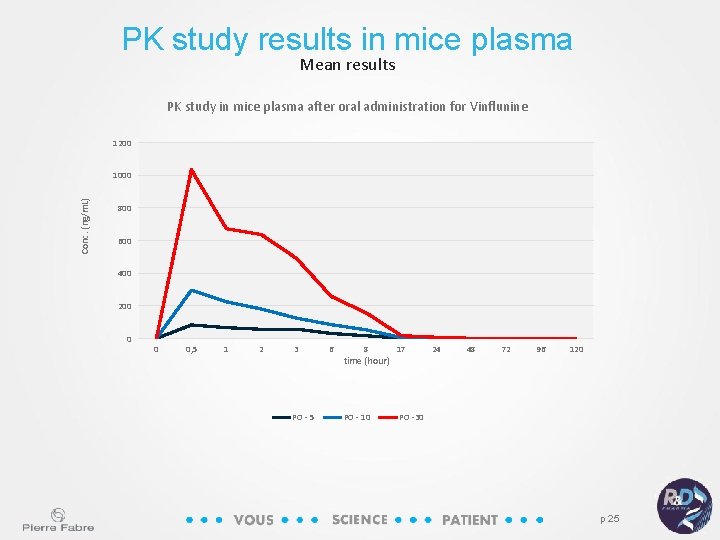
PK study results in mice plasma Mean results PK study in mice plasma after oral administration for Vinflunine 1200 Conc. (ng/m. L) 1000 800 600 400 200 0 0 0, 5 1 2 3 PO - 5 6 8 time (hour) PO - 10 17 24 48 72 96 120 PO -30 p 25
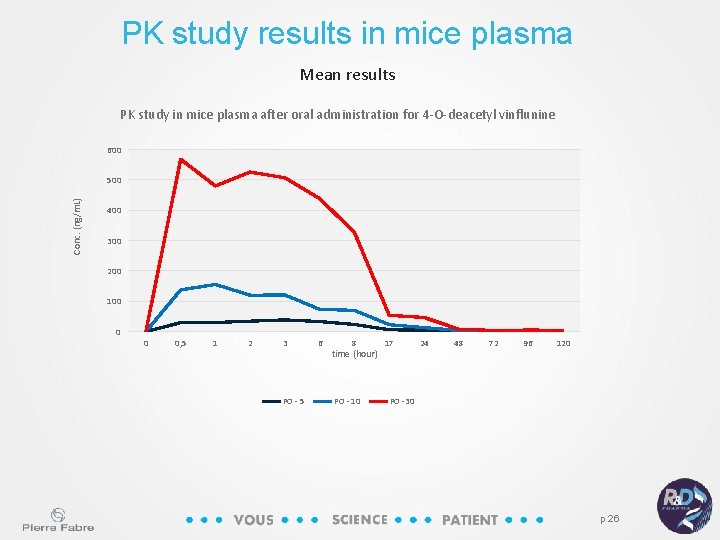
PK study results in mice plasma Mean results PK study in mice plasma after oral administration for 4 -O-deacetyl vinflunine 600 Conc. (ng/m. L) 500 400 300 200 100 0 0 0, 5 1 2 3 PO - 5 6 8 time (hour) PO - 10 17 24 48 72 96 120 PO -30 p 26

Development of method for different compounds in rat plasma, CSF and brain The aim of this development is to have a generic extraction method for similar compound using plasma as calibrator for dosing plasma, CSF and brain in early studies. QC is performed in all matrices. At this time 4 compounds on 15 is performed and samples are analysed. p 27

Extraction procedure Prepare sample 4 µL of sample 2 µL of internal standard 100 µL of 2% acid formic solution Shake plate Condition and equilibrate Oasis µelution HLB 96 deep well 200 µL of methanol 200 µL of water Load Sample Wash 200 µL of 2% ammonia solution 200 µL of a mixture of methanol /water 5/95 Elute 120 µL of a mixture of acetonitrile/ Water 0. 1% formic acid (50/20) p 28

Mass spectrometer parameters Mode Positive Electro spray : 3. 5 KV Capillary temperature: 300°C Sheath gas pressure: 35 Aux. gas pressure: 70 L/Hr Collision pressure: 1. 5 Skimmer Offset: 12 e. V Tube lens: auto Collision energy: Compound dependent p 29

Chromatography parameters Pump Surveyor Autosampler Surveyor Column: UPLC BEH phenyl Waters 50 x 2. 1 mm, 1. 7 µm Aqueous mobile phase (A): Water with 0. 1% formic acid Organic mobile phase %(B): Acetonitrile with 0. 1% formic acid Flow rate: 0. 4 m. L/min Isocratic mode: Compound dependent Column temperature: 30°C Sample temperature: 10°C Injection volume: 4 µL p 30
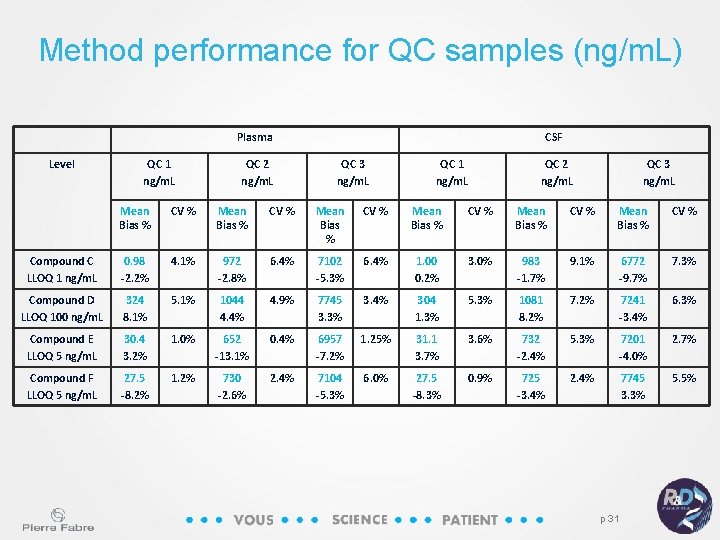
Method performance for QC samples (ng/m. L) Plasma Level QC 1 ng/m. L QC 2 ng/m. L CSF QC 3 ng/m. L QC 1 ng/m. L QC 2 ng/m. L QC 3 ng/m. L Mean Bias % CV % Mean Bias % CV % Compound C LLOQ 1 ng/m. L 0. 98 -2. 2% 4. 1% 972 -2. 8% 6. 4% 7102 -5. 3% 6. 4% 1. 00 0. 2% 3. 0% 983 -1. 7% 9. 1% 6772 -9. 7% 7. 3% Compound D LLOQ 100 ng/m. L 324 8. 1% 5. 1% 1044 4. 4% 4. 9% 7745 3. 3% 3. 4% 304 1. 3% 5. 3% 1081 8. 2% 7241 -3. 4% 6. 3% Compound E LLOQ 5 ng/m. L 30. 4 3. 2% 1. 0% 652 -13. 1% 0. 4% 6957 -7. 2% 1. 25% 31. 1 3. 7% 3. 6% 732 -2. 4% 5. 3% 7201 -4. 0% 2. 7% Compound F LLOQ 5 ng/m. L 27. 5 -8. 2% 1. 2% 730 -2. 6% 2. 4% 7104 -5. 3% 6. 0% 27. 5 -8. 3% 0. 9% 725 -3. 4% 2. 4% 7745 3. 3% 5. 5% p 31

Which parameters will be evaluate? The same as traditional validation as plasma is store frozen in adequate container and not in Capillary. Perhaps an attention point , the adsorption in container and freeze thaw cycle due to low volume p 32
Thanks to the team Eva Louise Caroline Coralie Nathalie p 33

Questions and discussion Go ahead p 34
- Slides: 34