Water Properties of Water Bill Nye Surface Tension

Water

Properties of Water • Bill Nye – Surface Tension • http: //www. youtube. com/watch? v=Hm 52 rkh 68 JA &safety_mode=true&persist_safety_mode=1&safe =active

Universal Solvent
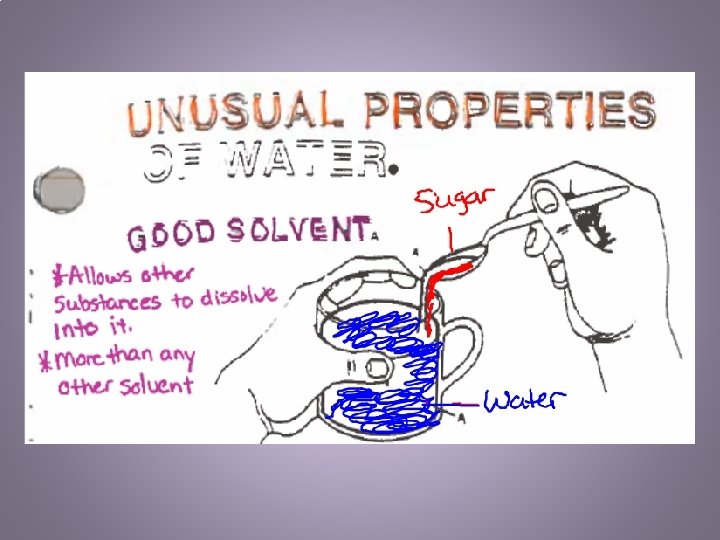

“Universal” Solvent • What happens when you make a fruit drink from a powdered mix? • homogeneous = solution. • In an aqueous solution, water is the solvent.

Capillary Action • Water defies gravity to travel up. • Water molecules are attracted to each other and to other molecules as well
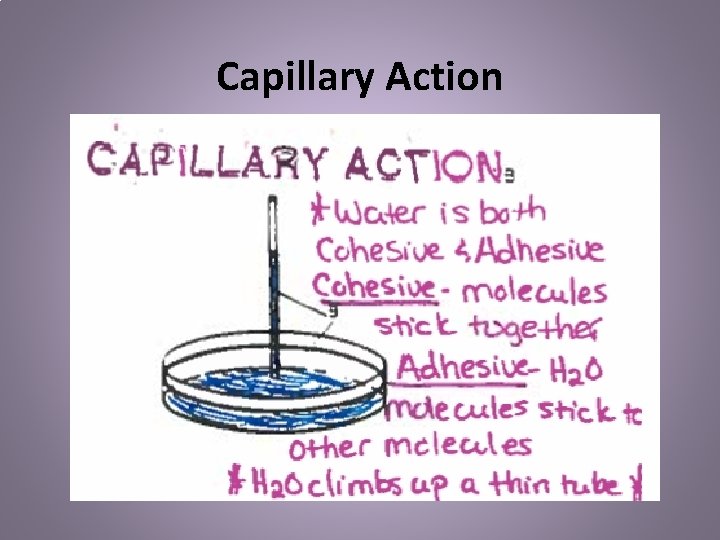
Capillary Action

Trees have specialized structures to transport water: - xylem and phloem “plumbing” * Water molecules are “dragged” from the roots to the top of the tree by capillary action and cohesion!
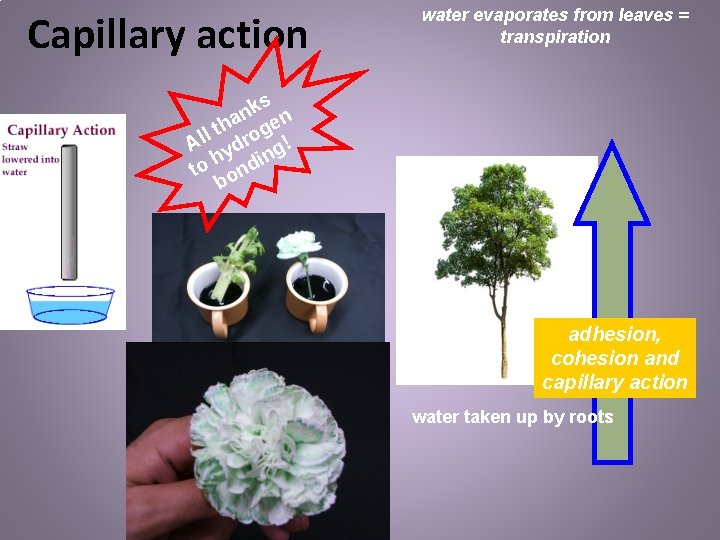
Capillary action water evaporates from leaves = transpiration ks n a en h t g l Al ydro g! h din o t on b adhesion, cohesion and capillary action water taken up by roots

Hydrogen bonds hold water together, a phenomenon called …. Cohesion and Adhesion Launchpad: Cohesion and Adhesion On -Board the International Space Station http: //www. youtube. com/watch? v=o. AY 3 y. ISf 24&safety_mode=true&persist_safety_mode=1&safe=a ctive

Cohesion Co-hesion= 1. “Co-” = share 2. “-hesion”= bonded example -> (adhere) So… Cohesion refers to waters attraction to other water molecules.

Adhesion – Adhesion refers to attraction to other molecules. • Water is adhesive to any substance with which it can form hydrogen bonds.

High Specific Heat • It is a steamy summer day. The air is hot, the sidewalk is hot, and the sandy beach is hot. But when you jump into the ocean, the water is surprisingly cool! If you go for an evening swim, however, the water is warmer than the cool air.


Three-fourths of the earth is covered by water. The water serves as a large heat sink responsible for: 1. Prevention of temperature fluctuations that are outside the range suitable for life. 2. Coastal areas having a mild climate 3. A stable marine environment The Earth is over 75% water!
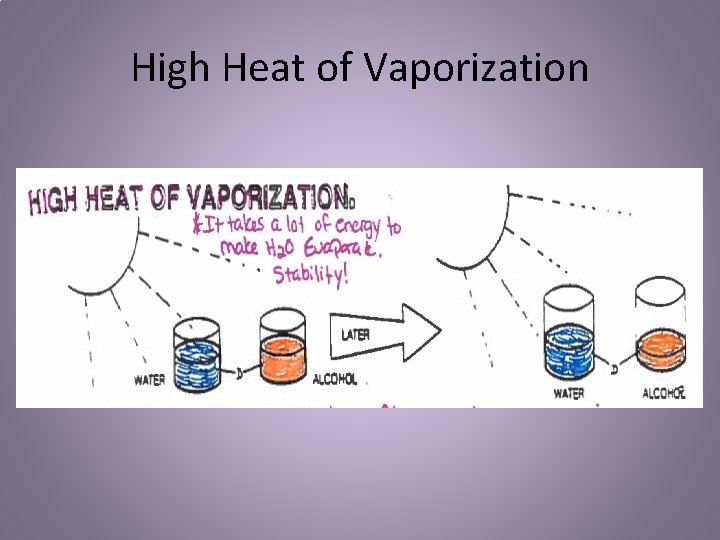
High Heat of Vaporization

High Heat of Fusion & High Surface Tension

Surface Tension • Have you ever watched water striders (spiders!!!) skate across the surface of a pond without sinking? • They are supported by the surface tension of the water….

• Surface tension is the tightness across the surface of water that is caused by the polar molecules pulling on one another. (its related to cohesion) Some animals can stand, walk, or run on water without breaking the surface.

Density of Water • • Most dense at 4 o. C Contracts until 4 o. C Expands from 4 o. C to 0 o. C Ice floats because it is less dense. The density of water: 1. Prevents water from freezing from the bottom up. 2. Ice forms on the surface first—the freezing of the water releases heat to the water below creating insulation. 3. Makes transition between season less abrupt.

Expansion on Freezing

Review of Water Properties With Tim and Moby • http: //www. brainpop. com/science/earthsystem/water/ • Complete Daily Water Planner
- Slides: 22