Review 1 Describe van Deemter equation and van

Review: (1) Describe van Deemter equation and van Deemter plot. (2) Define retention factor, selectivity factor, and resolution. How to enhance resolution of a chromatographic separation? (3) What is the general elution problem? How to solve this problem?

Principles of Instrumental Analysis Chapter 27 Gas Chromatography

Gas Chromatography (GC): vaporized analytes (solutes) are partitioned between a mobile gaseous phase and a liquid or a solid stationary phase held in a column. The mobile phase does not interact with molecules of the analytes (solutes). Gas-Liquid Chromatography (GLC): partition between mobile phase and liquid phase immobilized (固定化) on the surface of an inert solid packing or on the walls of a capillary tubing. Gas-Solid Chromatography (GSC): physical adsorption 27 A Principles of GLC Due to compressibility of gaseous mobile phases, the mathematical relationships obtained in Chapter 26 need minor modification. Retention volume (Vg) instead of retention time (t. R): Vg = (K/r. S) x (273/Tc) (27 -7) Tc : column temperature r. S : density of liquid stationary phase

VR = t. RF, VM = t. MF F: average volumetric flow rate within the column F = Fm x (Tc/T) x (P - PH 2 O)/P Tc: column temperature (T: ambient) P: gas pressure at column end (ambient) Fm: measure flow rate PH 2 O: vapor pressure of water Corrected retention volume (at average column pressure): VR 0 = jt. RF, VM 0 = jt. MF j: pressure drop correction factor (compressibility factor) …………. . (27 -4) Specific retention volume: Vg = (VR 0 - VM 0)/m. S x (273/Tc) = [j. F(t. R – t. M)/m. S] x 273/Tc m S: mass of the stationary phase Vg = j. Ft. Mk/m. S x 273/Tc = VM 0 k/m. S x 273/Tc = KVS/m. S x 273/Tc r. S = m. S/VS Vg = K/r. S x 273/Tc ∵ retention factor k = (t. R - t. M)/t. M ∵ k = KVS/VM (K: distribution constant) ---------------(27 -7) At a given temperature, Vg depends only on the distribution constant (K) and density (r. S) of stationary phase (liquid).
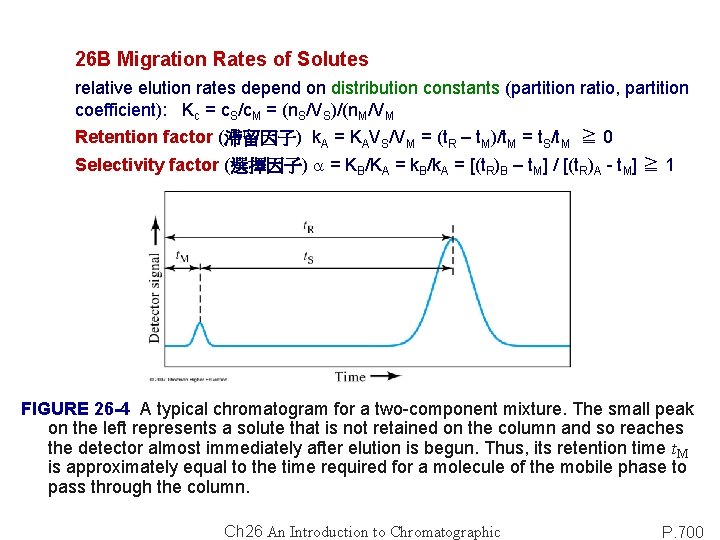
26 B Migration Rates of Solutes relative elution rates depend on distribution constants (partition ratio, partition coefficient): Kc = c. S/c. M = (n. S/VS)/(n. M/VM Retention factor (滯留因子) k. A = KAVS/VM = (t. R – t. M)/t. M = t. S/t. M ≧ 0 Selectivity factor (選擇因子) a = KB/KA = k. B/k. A = [(t. R)B – t. M] / [(t. R)A - t. M] ≧ 1 FIGURE 26 -4 A typical chromatogram for a two-component mixture. The small peak on the left represents a solute that is not retained on the column and so reaches the detector almost immediately after elution is begun. Thus, its retention time t. M is approximately equal to the time required for a molecule of the mobile phase to pass through the column. Ch 26 An Introduction to Chromatographic P. 700

Theory of Band Broadening: van Deemter equation: H = A + (B/u) + CSu + Cmu……. (26 -23) A: multiple path effects (eddy diffusion) B: longitudinal diffusion coefficient CS: mass-transfer coefficient for stationary phase CM: mass-transfer coefficient for mobile phase TABLE 26 -3 Processes That Contribute to Band Broadening Large diffusion rate in gases: 104 - 105 greater than liquids.
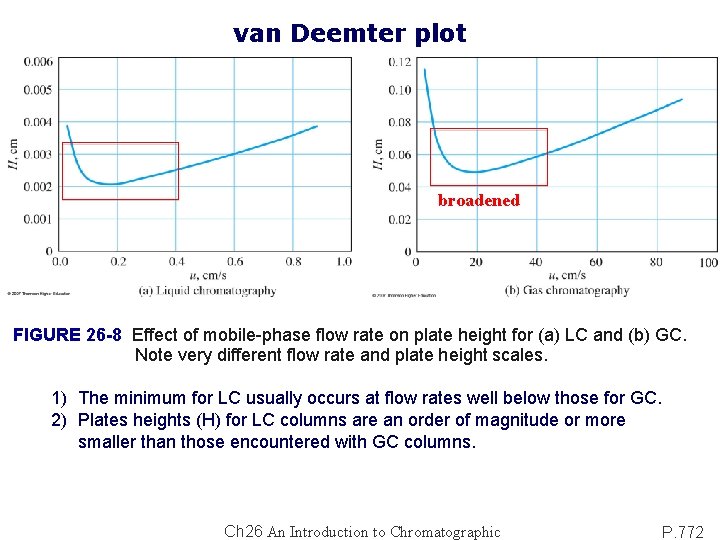
van Deemter plot broadened FIGURE 26 -8 Effect of mobile-phase flow rate on plate height for (a) LC and (b) GC. Note very different flow rate and plate height scales. 1) The minimum for LC usually occurs at flow rates well below those for GC. 2) Plates heights (H) for LC columns are an order of magnitude or more smaller than those encountered with GC columns. Ch 26 An Introduction to Chromatographic P. 772
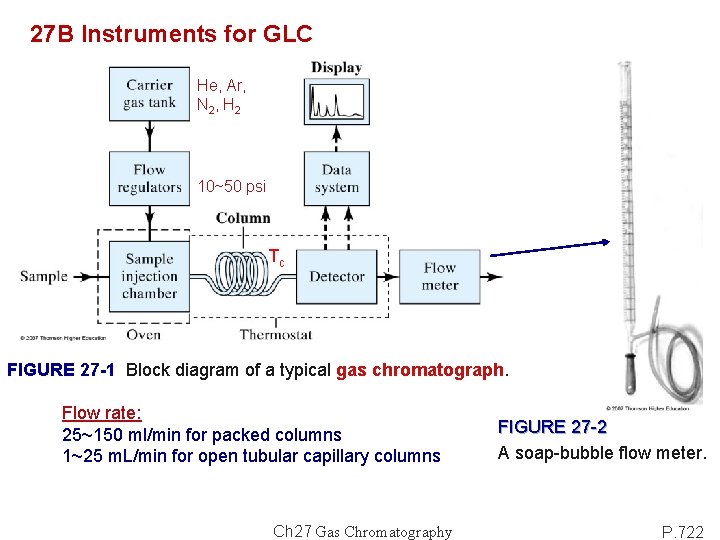
27 B Instruments for GLC He, Ar, N 2 , H 2 10~50 psi Tc FIGURE 27 -1 Block diagram of a typical gas chromatograph. Flow rate: 25~150 ml/min for packed columns 1~25 m. L/min for open tubular capillary columns Ch 27 Gas Chromatography FIGURE 27 -2 A soap-bubble flow meter. P. 722
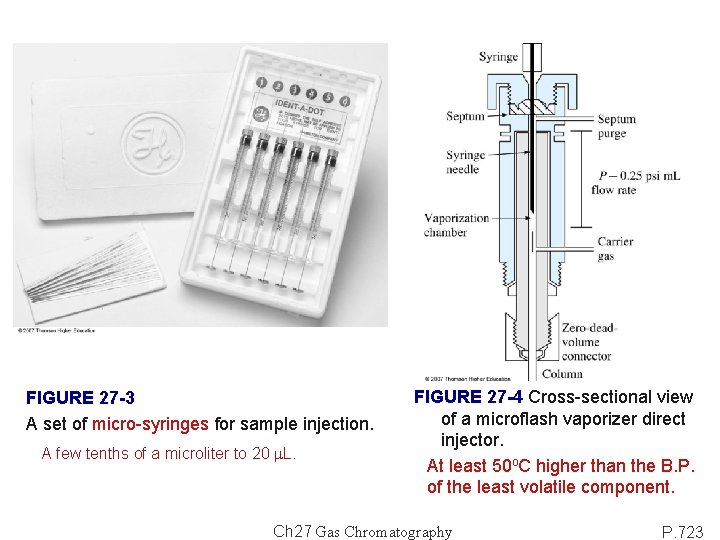
FIGURE 27 -3 A set of micro-syringes for sample injection. A few tenths of a microliter to 20 m. L. FIGURE 27 -4 Cross-sectional view of a microflash vaporizer direct injector. At least 50 o. C higher than the B. P. of the least volatile component. Ch 27 Gas Chromatography P. 723
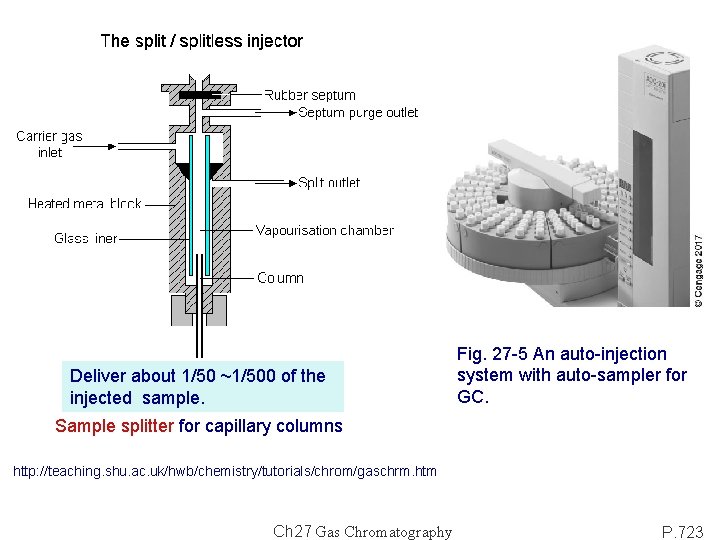
Deliver about 1/50 ~1/500 of the injected sample. Fig. 27 -5 An auto-injection system with auto-sampler for GC. Sample splitter for capillary columns http: //teaching. shu. ac. uk/hwb/chemistry/tutorials/chrom/gaschrm. htm Ch 27 Gas Chromatography P. 723
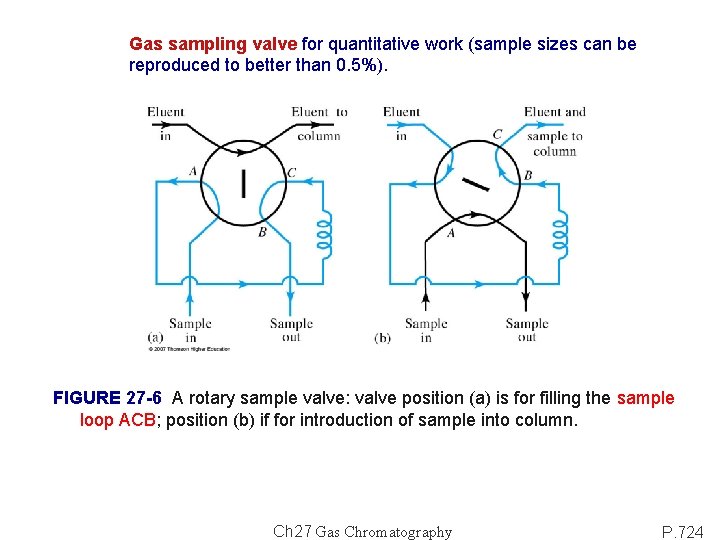
Gas sampling valve for quantitative work (sample sizes can be reproduced to better than 0. 5%). FIGURE 27 -6 A rotary sample valve: valve position (a) is for filling the sample loop ACB; position (b) if for introduction of sample into column. Ch 27 Gas Chromatography P. 724

Column configuration: constructed of fused silica, stainless steel or Teflon; formed as coils (diameter: 10~30 cm); housed in a thermostatted oven. Packed column: 1~5 m. Open tubular (capillary) column: ~100 m. Column temperature is an important variable: equal or slightly above the average boiling point of a sample → reasonable elution time (2 -30 min). For sample with a broad boiling range Temperature programming. In general, optimal resolution is associated with minimal temperature; however, the cost of lowered temperature is an increase in elution time [Figure 27 -8(a)-(b)]. FIGURE 27 -7 Fused-silica capillary columns. Ch 27 Gas Chromatography P. 792
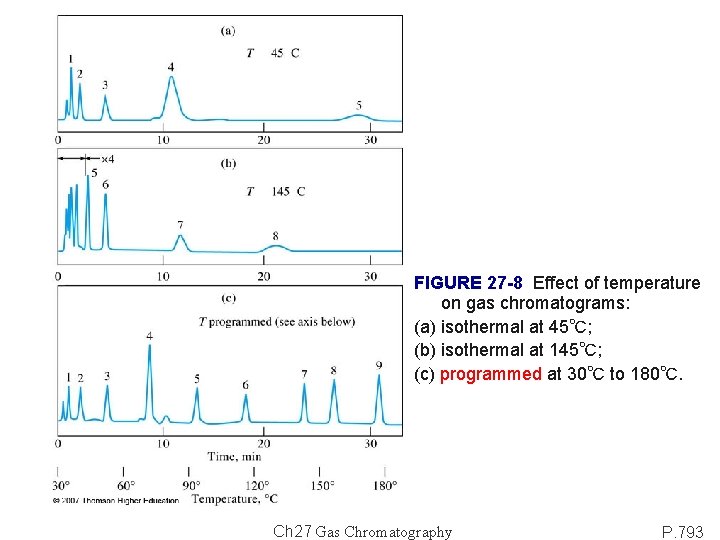
FIGURE 27 -8 Effect of temperature on gas chromatograms: (a) isothermal at 45℃; (b) isothermal at 145℃; (c) programmed at 30℃ to 180℃. Ch 27 Gas Chromatography P. 793

27 B-4 Detection Systems Characteristics of the ideal detectors: 1) Adequate sensitivity. (10 -8 ~ 10 -15 g solute/sec) 2) Good stability and reproducibility. 3) A linear responses to solutes (several orders of magnitude). 4) A temperature range from r. t. to at least 400 o. C. 5) A short response time independent of flow rate. 6) High reliability and ease of use. 7) Similarity in response toward all solutes. 8) The detector should be non-destructive. TABLE 27 -1 Typical Gas Chromatographic Detectors P. 725
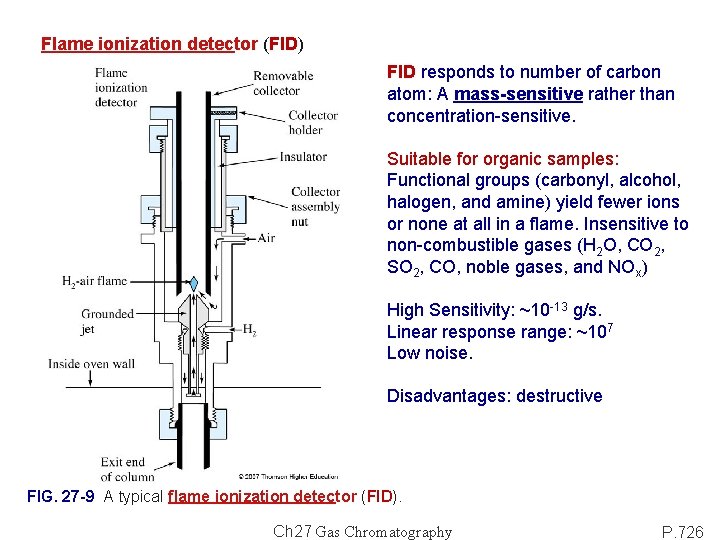
Flame ionization detector (FID) FID responds to number of carbon atom: A mass-sensitive rather than concentration-sensitive. Suitable for organic samples: Functional groups (carbonyl, alcohol, halogen, and amine) yield fewer ions or none at all in a flame. Insensitive to non-combustible gases (H 2 O, CO 2, SO 2, CO, noble gases, and NOx) High Sensitivity: ~10 -13 g/s. Linear response range: ~107 Low noise. Disadvantages: destructive FIG. 27 -9 A typical flame ionization detector (FID). Ch 27 Gas Chromatography P. 726
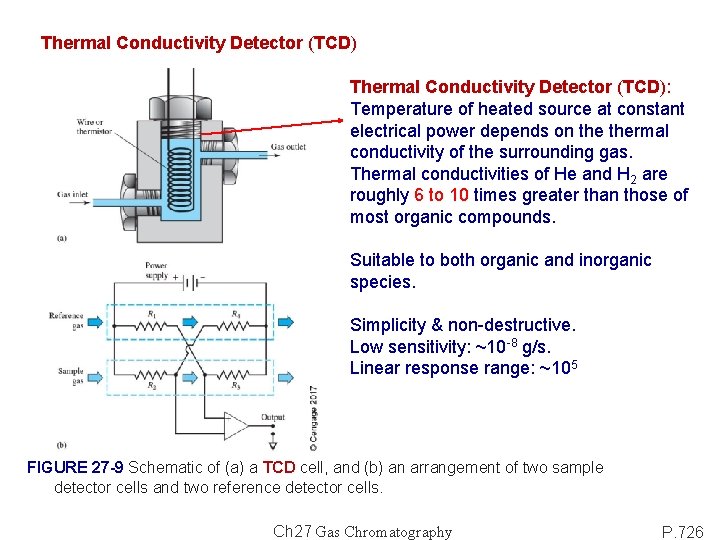
Thermal Conductivity Detector (TCD): Temperature of heated source at constant electrical power depends on thermal conductivity of the surrounding gas. Thermal conductivities of He and H 2 are roughly 6 to 10 times greater than those of most organic compounds. Suitable to both organic and inorganic species. Simplicity & non-destructive. Low sensitivity: ~10 -8 g/s. Linear response range: ~105 FIGURE 27 -9 Schematic of (a) a TCD cell, and (b) an arrangement of two sample detector cells and two reference detector cells. Ch 27 Gas Chromatography P. 726
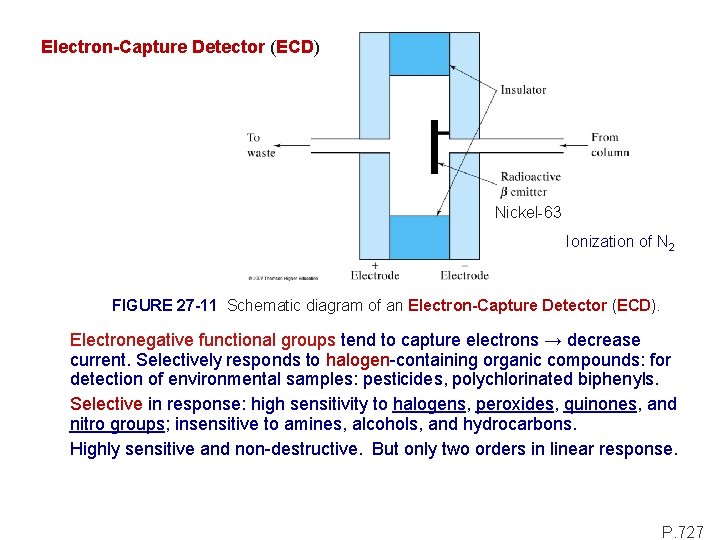
Electron-Capture Detector (ECD) Nickel-63 Ionization of N 2 FIGURE 27 -11 Schematic diagram of an Electron-Capture Detector (ECD). Electronegative functional groups tend to capture electrons → decrease current. Selectively responds to halogen-containing organic compounds: for detection of environmental samples: pesticides, polychlorinated biphenyls. Selective in response: high sensitivity to halogens, peroxides, quinones, and nitro groups; insensitive to amines, alcohols, and hydrocarbons. Highly sensitive and non-destructive. But only two orders in linear response. P. 727
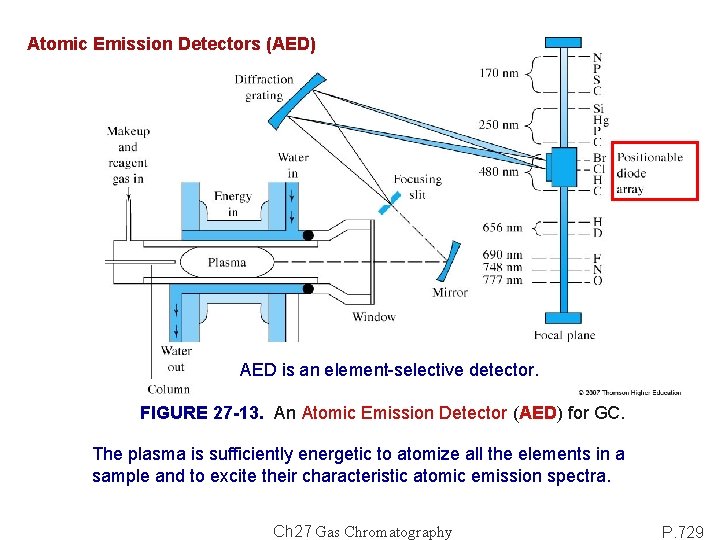
Atomic Emission Detectors (AED) AED is an element-selective detector. FIGURE 27 -13. An Atomic Emission Detector (AED) for GC. The plasma is sufficiently energetic to atomize all the elements in a sample and to excite their characteristic atomic emission spectra. Ch 27 Gas Chromatography P. 729
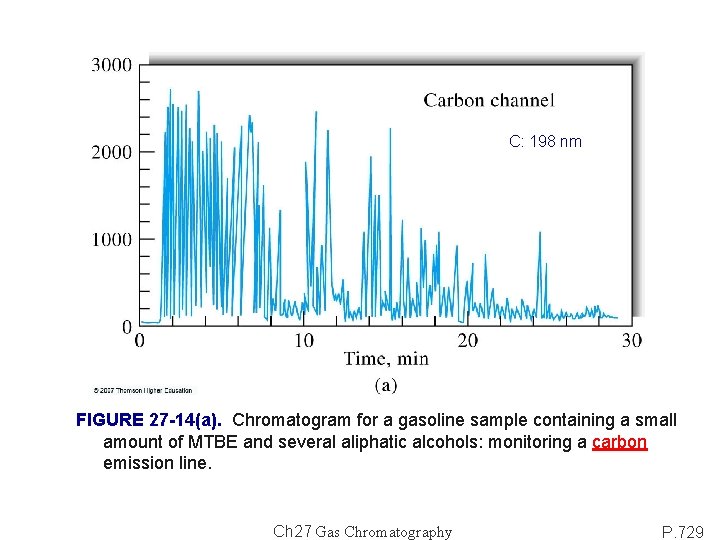
C: 198 nm FIGURE 27 -14(a). Chromatogram for a gasoline sample containing a small amount of MTBE and several aliphatic alcohols: monitoring a carbon emission line. Ch 27 Gas Chromatography P. 729
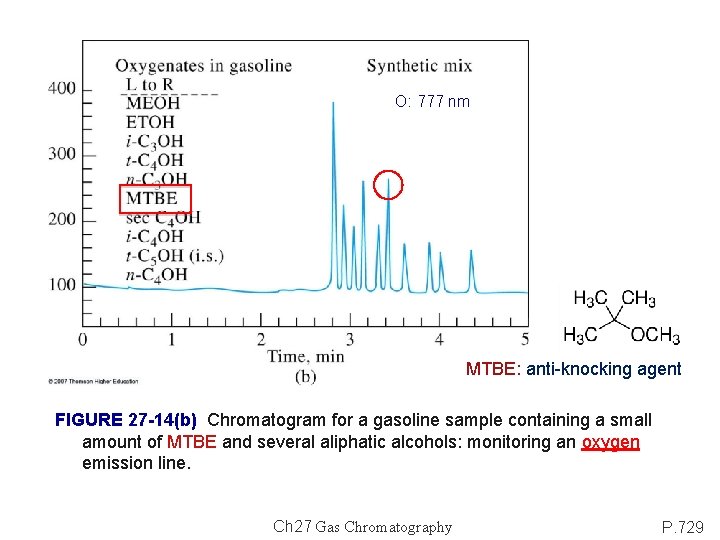
O: 777 nm MTBE: anti-knocking agent FIGURE 27 -14(b) Chromatogram for a gasoline sample containing a small amount of MTBE and several aliphatic alcohols: monitoring an oxygen emission line. Ch 27 Gas Chromatography P. 729
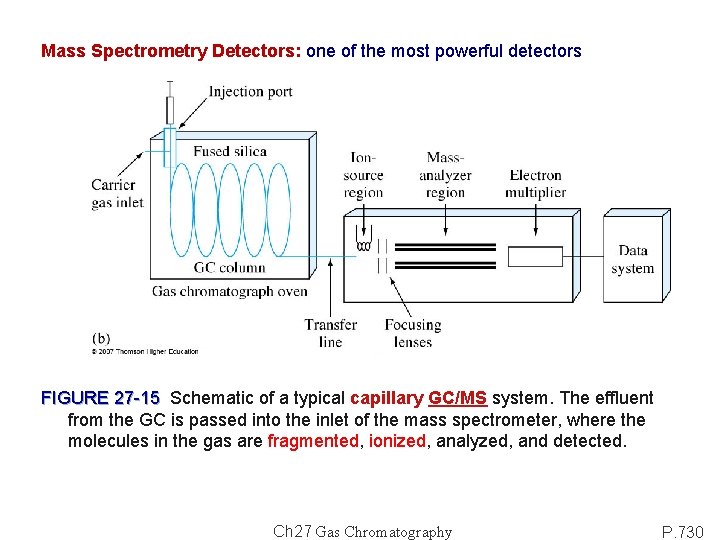
Mass Spectrometry Detectors: one of the most powerful detectors FIGURE 27 -15 Schematic of a typical capillary GC/MS system. The effluent from the GC is passed into the inlet of the mass spectrometer, where the molecules in the gas are fragmented, ionized, analyzed, and detected. Ch 27 Gas Chromatography P. 730
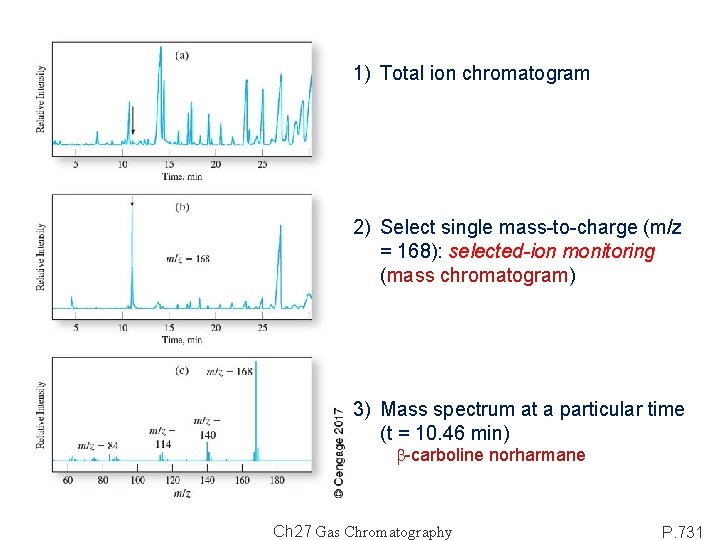
1) Total ion chromatogram 2) Select single mass-to-charge (m/z = 168): selected-ion monitoring (mass chromatogram) 3) Mass spectrum at a particular time (t = 10. 46 min) b-carboline norharmane Ch 27 Gas Chromatography P. 731

27 C GC Columns and Stationary Phases TABLE 27 -2 Properties and Characteristics of Typical GC Columns Greater sample capacity P. 733

60~80 mesh (250 -170 mm) or 80~100 mesh (170 -149 mm) FIGURE 27 -17 A photomicrograph of a diatomaceous earth (矽藻土). Magnification 5000 X. (in support-coated open tubular: SCOT) P. 733
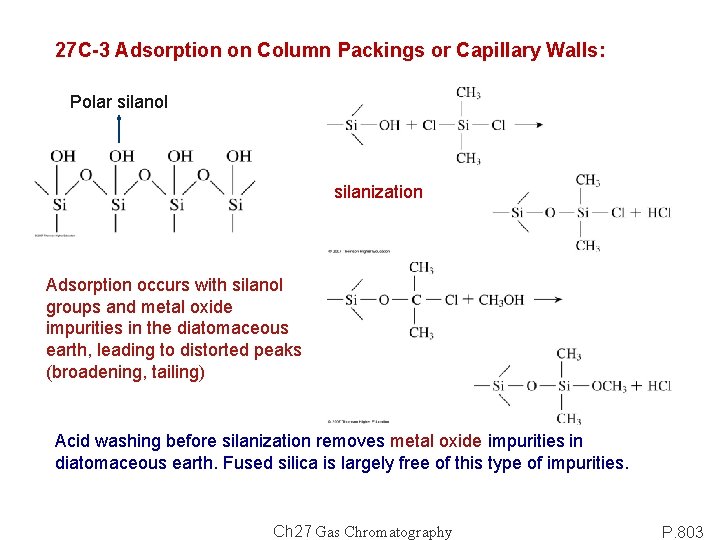
27 C-3 Adsorption on Column Packings or Capillary Walls: Polar silanol silanization Adsorption occurs with silanol groups and metal oxide impurities in the diatomaceous earth, leading to distorted peaks (broadening, tailing) Acid washing before silanization removes metal oxide impurities in diatomaceous earth. Fused silica is largely free of this type of impurities. Ch 27 Gas Chromatography P. 803

27 C-4 Stationary phase (immobilized liquid) requires: (1) low volatility, (2) thermal stability, (3) chemical inertness, and (4) solvent characteristics (k and a). Compatibility (solubility) of an analyte with stationary phase leads to reasonable residence time (k). 溶劑之溶解參數(d) “Like dissolves like”. Like refers to the “polarities” of the analyte and the immobilized liquid (stationary phase). In GC the polarity of stationary phase should match with that of the sample components. The order of elution is determined by the boiling point of the analytes. Ecoh = DHvap – RT (Cohesive energy) CED = Ecoh/V (Cohesive Energy Density) d = (CED)1/2 (Solubility Parameter) 26 (cal/ml)1/2
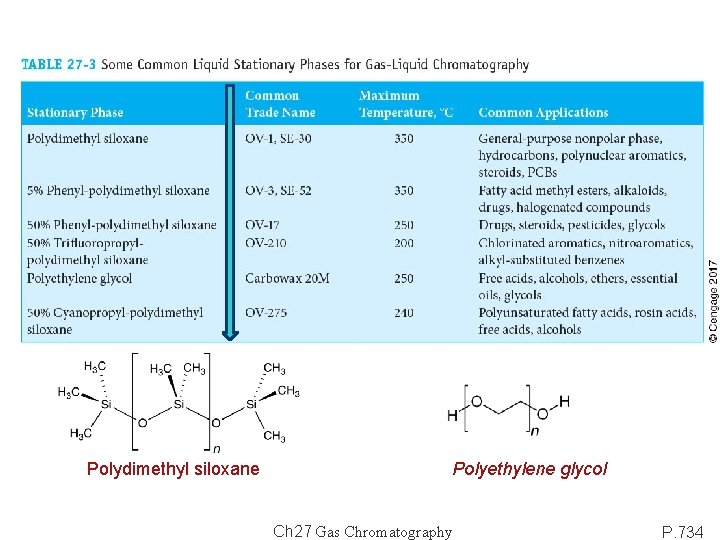
Polydimethyl siloxane Polyethylene glycol Ch 27 Gas Chromatography P. 734

Liquid Stationary Phase fro GLC Polarity Hydrocarbon Polysiloxane R= methyl: Polydimethyl siloxane bonded or cross-linked R= methyl, phenyl: x% Phenyl-polydimethyl siloxane R= methyl, trifluoropropyl: x% Trifluoropropyl-polydimethyl siloxane R= methyl, cyanopropyl: x% Cyanopropyl-polydimethyl siloxane Polyethylene glycol: H-(OCH 2)n-OH Polyester: H-(O-RO-CO-R’-CO)n-OH Analytes: Polar: alcohols, acids, amines. Medium polar: ethers, ketones, aldehydes. Non-polar: saturated hydrocarbons. The polarity of the stationary phase should match that of the analytes. When the match is good, the elution order is determined by the boiling point of the analytes Ch 27 Gas Chromatography P. 804
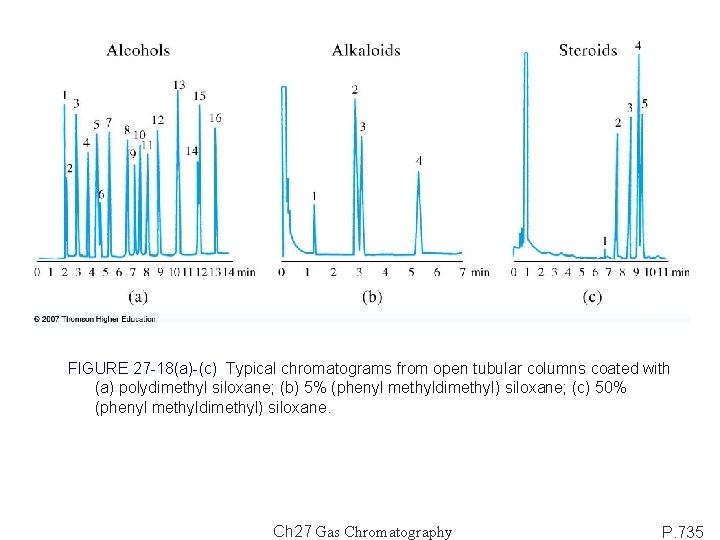
FIGURE 27 -18(a)-(c) Typical chromatograms from open tubular columns coated with (a) polydimethyl siloxane; (b) 5% (phenyl methyldimethyl) siloxane; (c) 50% (phenyl methyldimethyl) siloxane. Ch 27 Gas Chromatography P. 735
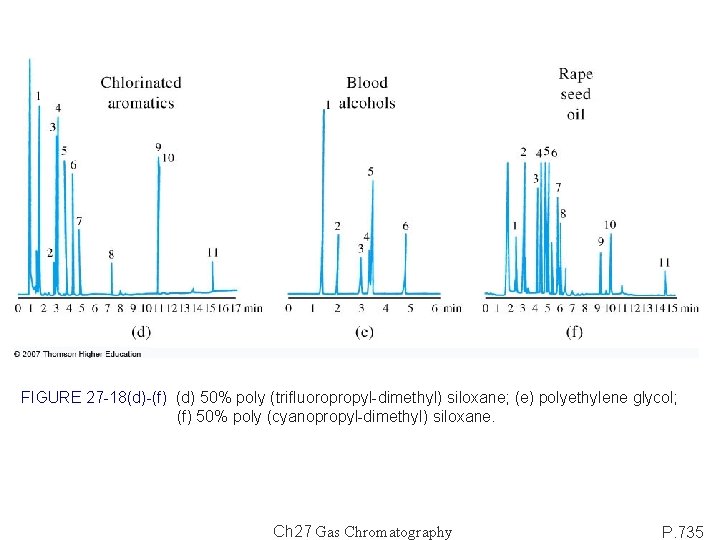
FIGURE 27 -18(d)-(f) (d) 50% poly (trifluoropropyl-dimethyl) siloxane; (e) polyethylene glycol; (f) 50% poly (cyanopropyl-dimethyl) siloxane. Ch 27 Gas Chromatography P. 735
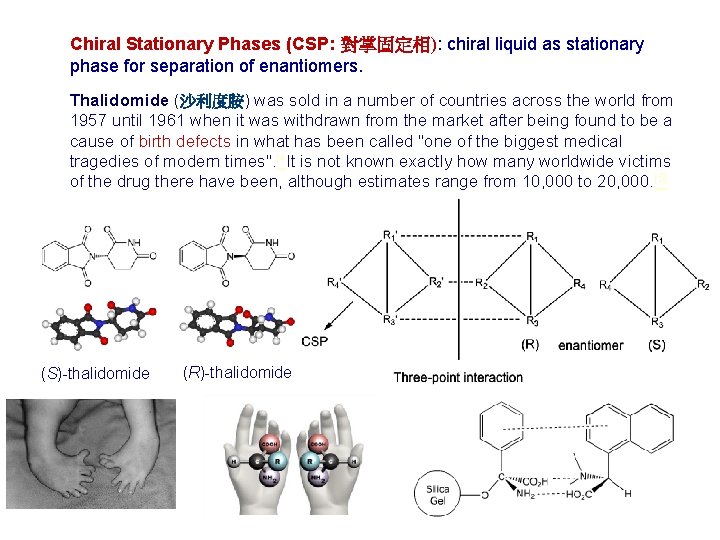
Chiral Stationary Phases (CSP: 對掌固定相): chiral liquid as stationary phase for separation of enantiomers. Thalidomide (沙利度胺) was sold in a number of countries across the world from 1957 until 1961 when it was withdrawn from the market after being found to be a cause of birth defects in what has been called "one of the biggest medical tragedies of modern times". [4 It is not known exactly how many worldwide victims of the drug there have been, although estimates range from 10, 000 to 20, 000. [5] (S)-thalidomide (R)-thalidomide

27 D Applications of GC 1) Perform separations 2) Completion of analysis: a) Qualitative analysis: retention times or volumes. (detector response factor) * Selectivity factors: a = KB/KA = (t. R’)B/(t. R’)A, B as common standard. 1) * Retention Index (I): The retention index scale is based on normal alkanes: equal to 100 x no. of carbon, regardless of the column packing, the temperature, or other chromatographic conditions. b) Quantitative analysis: peak heights or areas.
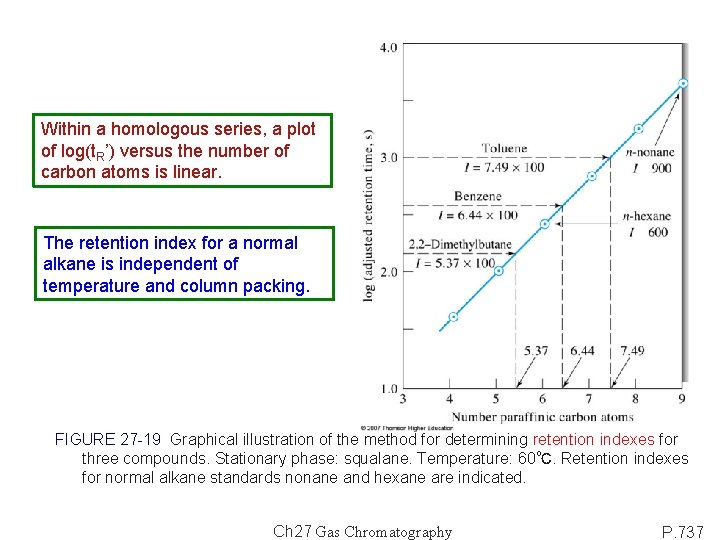
Within a homologous series, a plot of log(t. R’) versus the number of carbon atoms is linear. The retention index for a normal alkane is independent of temperature and column packing. FIGURE 27 -19 Graphical illustration of the method for determining retention indexes for three compounds. Stationary phase: squalane. Temperature: 60℃. Retention indexes for normal alkane standards nonane and hexane are indicated. Ch 27 Gas Chromatography P. 737
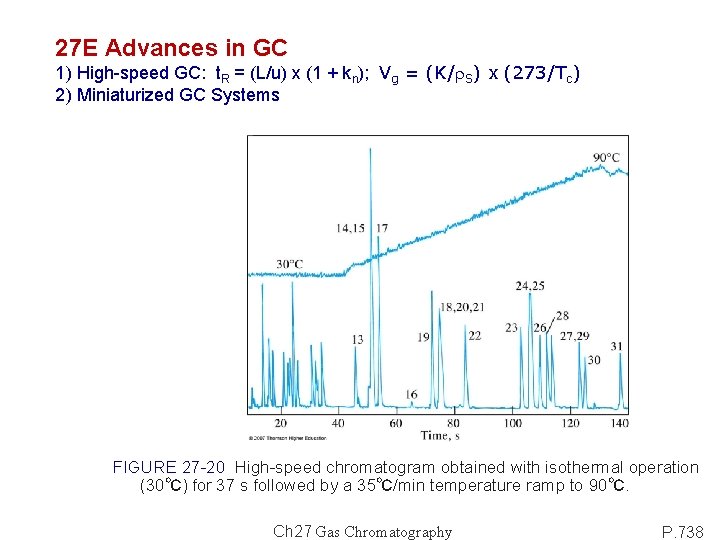
27 E Advances in GC 1) High-speed GC: t. R = (L/u) x (1 + kn); Vg = (K/r. S) x (273/Tc) 2) Miniaturized GC Systems FIGURE 27 -20 High-speed chromatogram obtained with isothermal operation (30℃) for 37 s followed by a 35℃/min temperature ramp to 90℃. Ch 27 Gas Chromatography P. 738
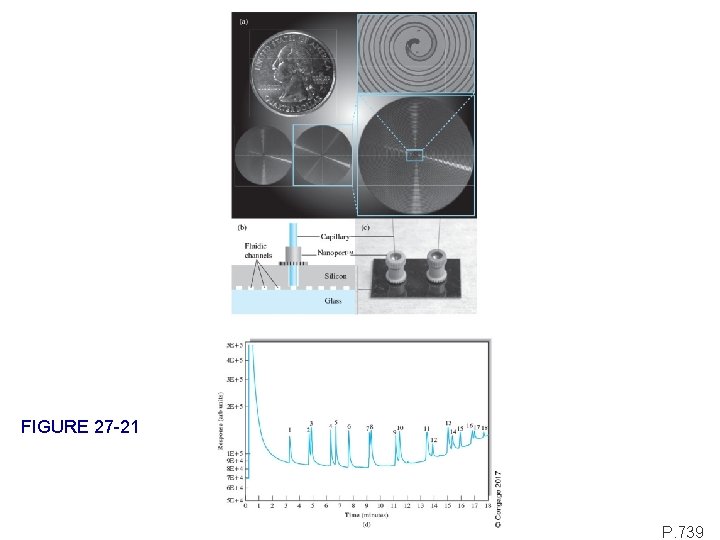
FIGURE 27 -21 P. 739
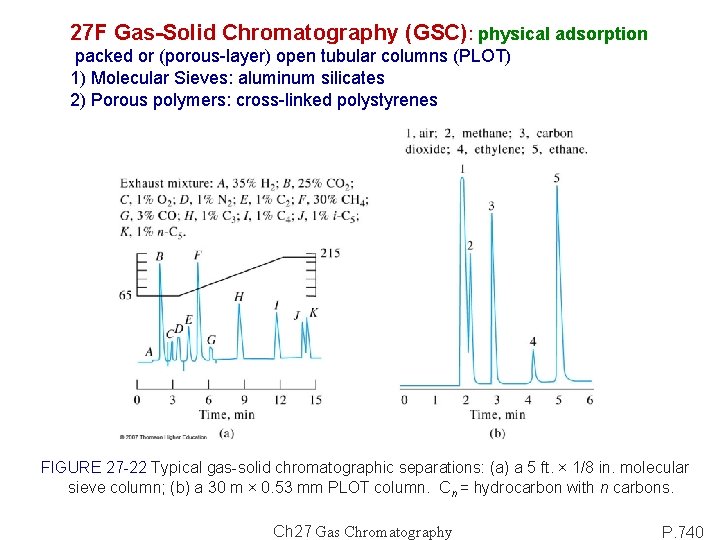
27 F Gas-Solid Chromatography (GSC): physical adsorption packed or (porous-layer) open tubular columns (PLOT) 1) Molecular Sieves: aluminum silicates 2) Porous polymers: cross-linked polystyrenes FIGURE 27 -22 Typical gas-solid chromatographic separations: (a) a 5 ft. × 1/8 in. molecular sieve column; (b) a 30 m × 0. 53 mm PLOT column. Cn = hydrocarbon with n carbons. Ch 27 Gas Chromatography P. 740

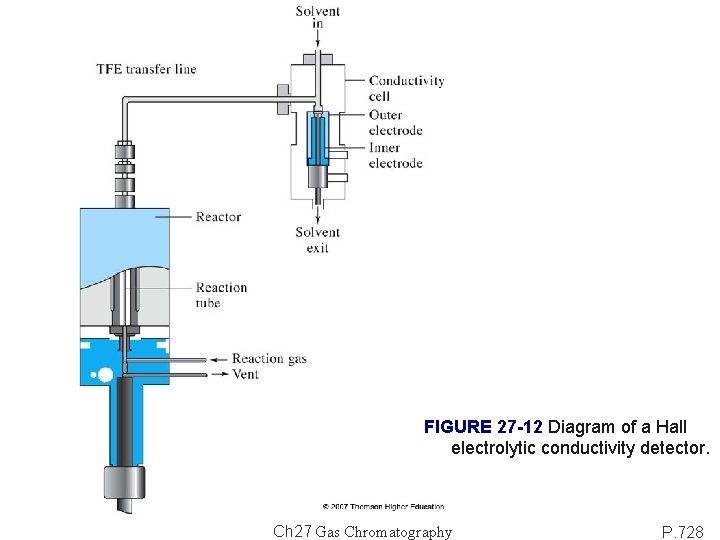
FIGURE 27 -12 Diagram of a Hall electrolytic conductivity detector. Ch 27 Gas Chromatography P. 728
- Slides: 38