CHAPTER 3 CHROMATOGRAPHIC METHODS OF SEPARATIONS Prepared By

CHAPTER -3 ----CHROMATOGRAPHIC METHODS OF SEPARATIONS Prepared By Dr. Khalid Ahmad Shadid Chemistry Department – Islamic University in Madinah 5/19/2021 1

GAS CHROMATOGRAPHY • The Separation Process in Gas Chromatography • In gas chromatography gaseous analyte is transported through the column by a gaseous mobile phase, called the carrier gas. • In gas-liquid chromatography the stationary phase is a nonvolatile liquid bonded to the inside of the column or to a fine solid support. • In gas-solid chromatography, analyte is adsorbed directly on solid particles of stationary phase. • In a schematic gas chromatograph (see the figure), volatile liquids or gaseous sample is injected through a septum into a heated port, in which it rapidly evaporates. Vapor is swept through the column by He, N 2 or H 2 carrier gas and separated analytes flow through a detector, whose response is displayed through a computer. The column must be hot enough to provide sufficient vapor pressure for analytes to be eluted in a reasonable time. The detector is maintained at a higher temperature 5/19/2021 than the 2
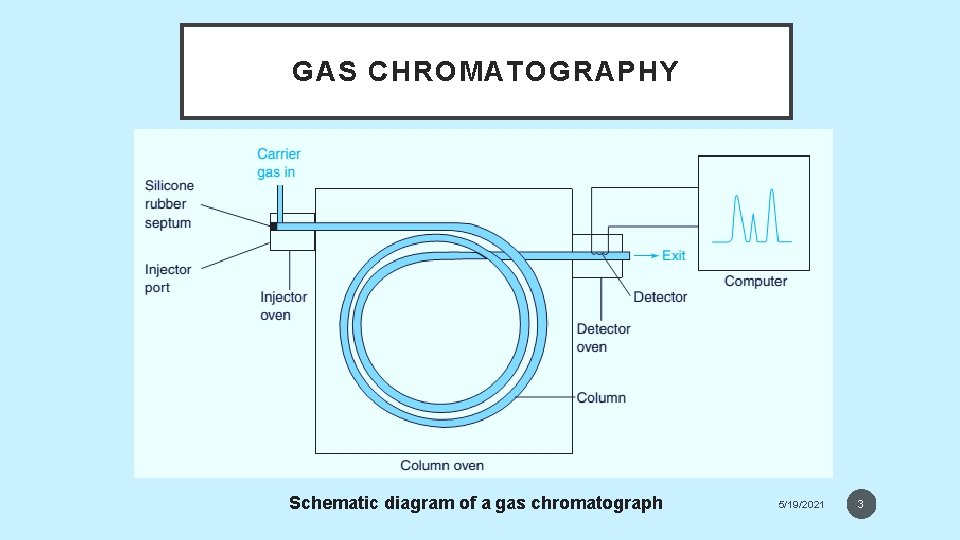
GAS CHROMATOGRAPHY Schematic diagram of a gas chromatograph 5/19/2021 3
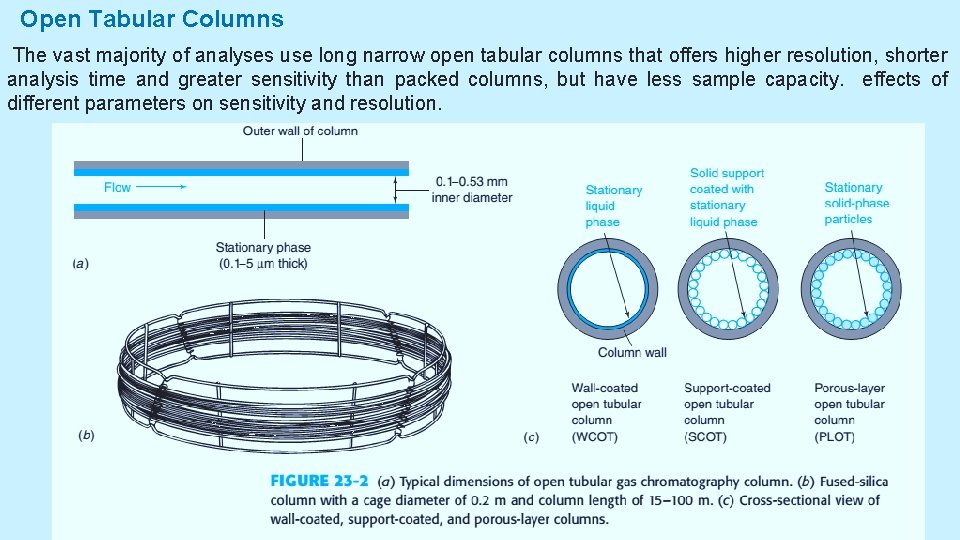
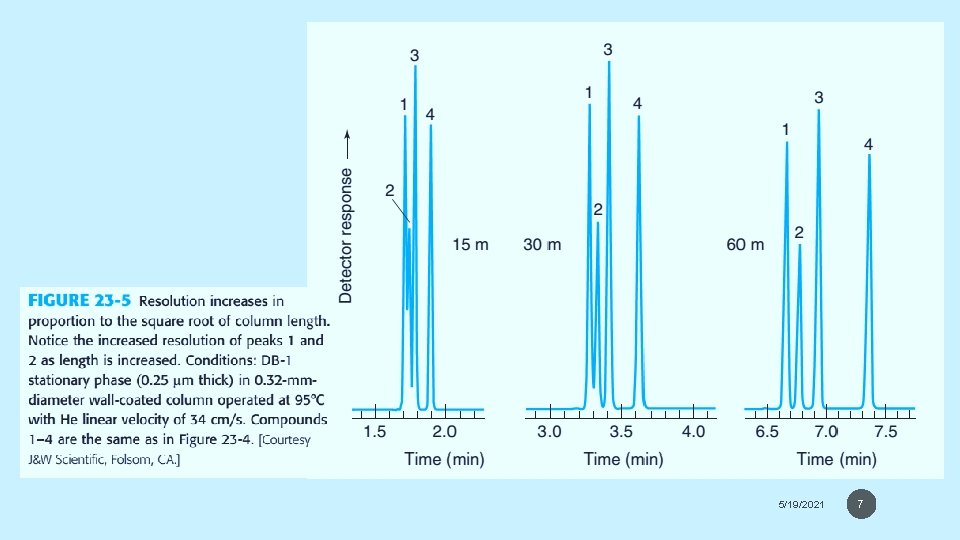
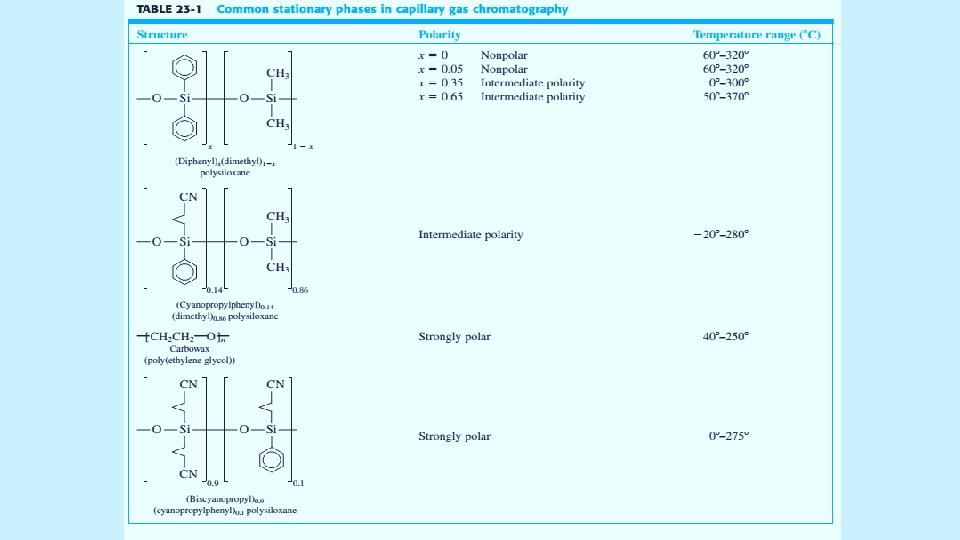
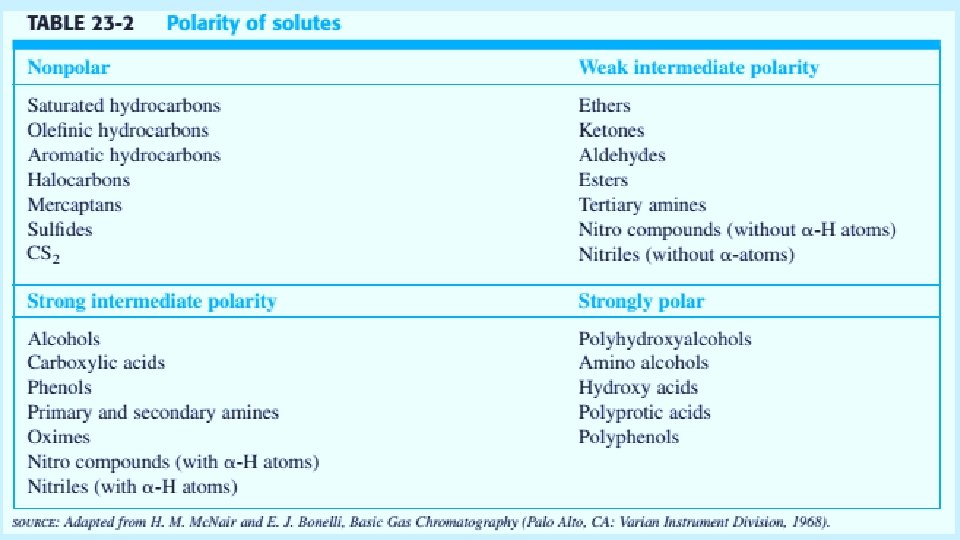
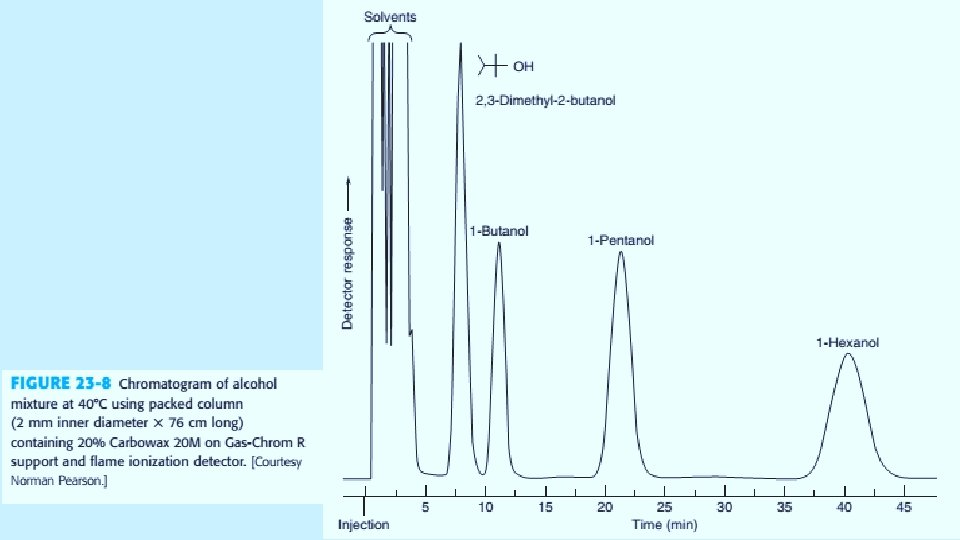
Open Tabular Columns The vast majority of analyses use long narrow open tabular columns that offers higher resolution, shorter analysis time and greater sensitivity than packed columns, but have less sample capacity. effects of different parameters on sensitivity and resolution. 5/19/2021 4
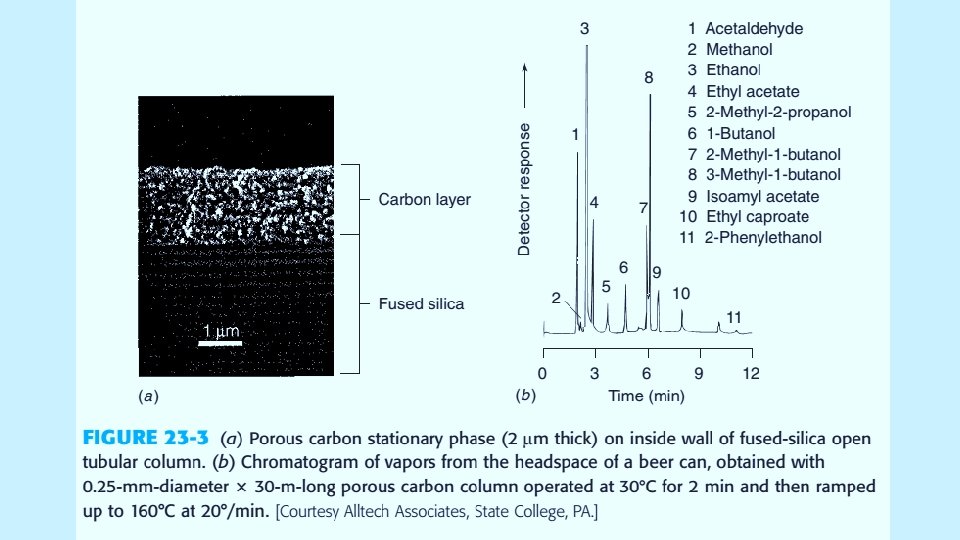
5/19/2021 5
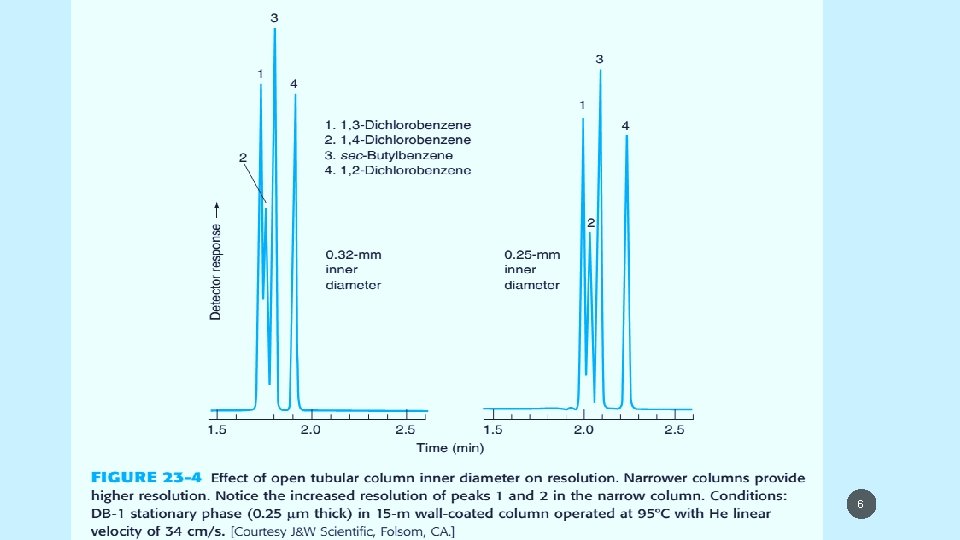
5/19/2021 6
5/19/2021 7

Packed Columns Packed columns contain fine particles of solid support coated with nonvolatile liquid stationary phase, or the solid itself may be the stationary phase. Compared with open tabular columns, packed columns provide greater sample capacity, but provide broader peaks, longer retention times and less resolution. Despite their resolution, packed columns are used for preparative separations or to separate gases that are poorly retained. Packed column Open tubular column (capillary column) 5/19/2021 10
5/19/2021 11

• Temperature and Pressure Programming • In temperature programming the temperature is raised during the separation to increase solute vapor pressure and decrease retention times of late components. At a constant temperature of 150 o. C, the more volatile compounds in Figure emerge close together, and less volatile compounds may not be eluted from the column. If the temperature is increased from 50 o. C, to 250 o. C, at a rate of 8 o. C, /min, all components are eluted and the separation of peaks is fairly uniform. Temperature should not be higher that stationary phase may be decomposed. • Many chromatograms may work with pressure control, but not common. Programmed pressure is useful for analytes that are thermally unstable 5/19/2021 12


Carrier Gas - Helium is most common carrier gas and is compatible with most detectors. All He, N 2 and H 2 gases have approximately the same plate height (0. 3 mm). - Fastest separation can be achieved with H 2 as carrier gas and H 2 can be run at higher flow rates. Figure shows the effect of carrier gas on separation of two compounds. H 2 is not commonly used due to high reactivity with metals and saturated compounds. - Impurities in the carrier gas degrade the stationary phase. Highly quality gas should be used, and they should pass through purifiers to remove O 2, H 2 O and trace organics. 5/19/2021 14
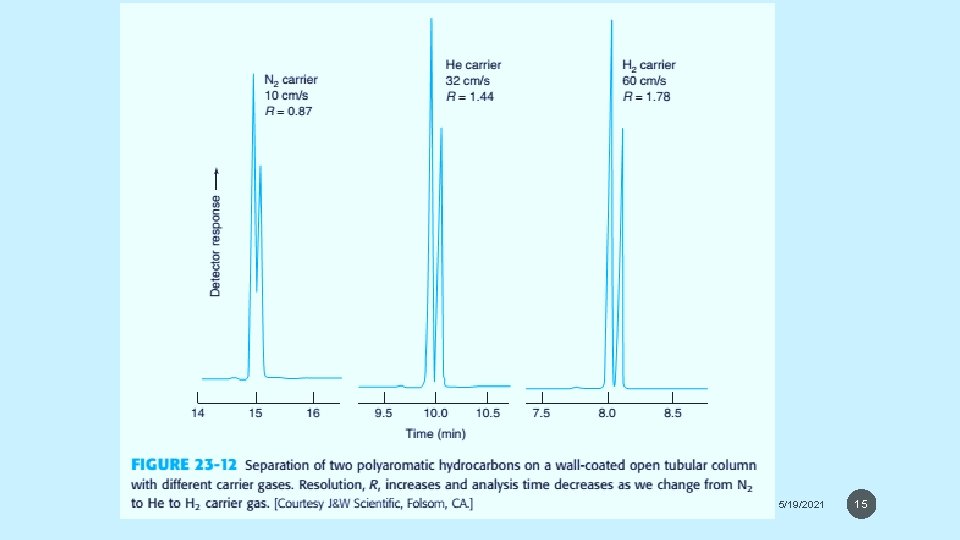
5/19/2021 15

SAMPLE INJECTION - Functions 1. An inlet for the sample. 2. To vaporize and mix the sample with the carrier gas before the sample enters the head of the column. - Temperature is set about 50°C higher than boiling point of the least volatile component of the sample. - Modes of injection and characteristics of injectors vary depending on type of column used whether split/splitless.
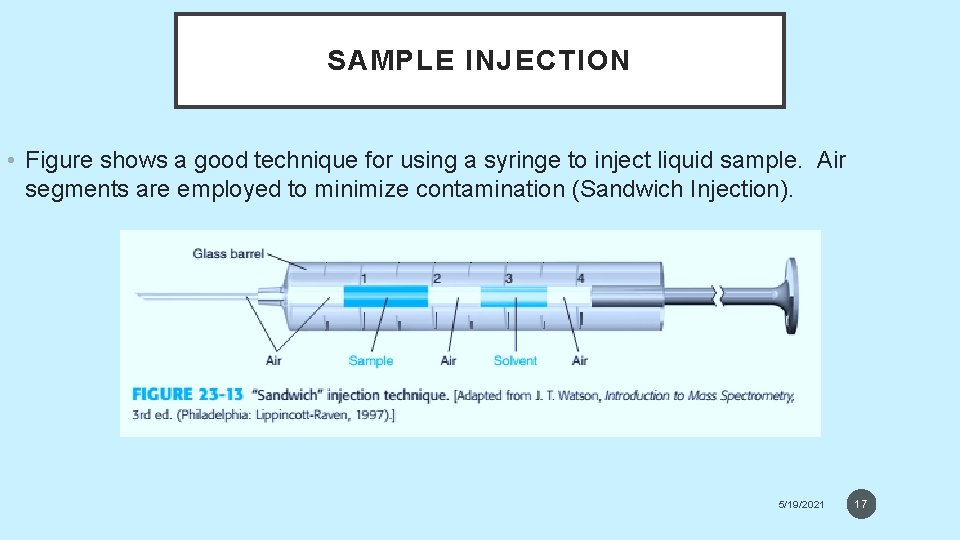
SAMPLE INJECTION • Figure shows a good technique for using a syringe to inject liquid sample. Air segments are employed to minimize contamination (Sandwich Injection). 5/19/2021 17

SAMPLE INJECTION- MODE OF INJECTIONS 5/19/2021 18

SAMPLE INJECTION SYSTEM - Column efficiency requires sample to be a suitable size and to introduced as a plug of vapor. - Band broadening and poor resolution are caused by slow injection and oversized sample. - Sample introduction usually In the form of neat liquid or solution. - Introduced in a small volumes: a. 1 μL - 20 μL for packed column. b. 1 x 10 -3 μL for capillary column. 5/19/2021 19
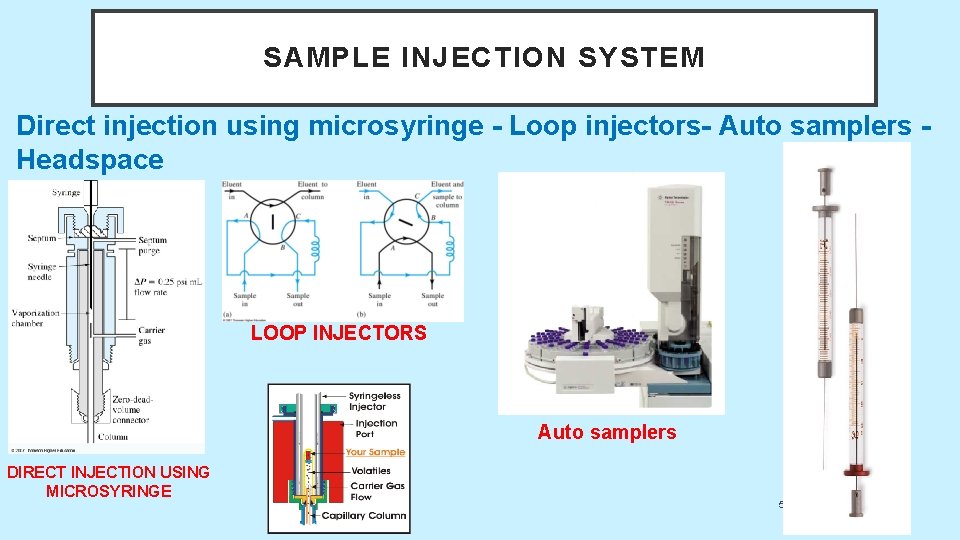
SAMPLE INJECTION SYSTEM Direct injection using microsyringe - Loop injectors- Auto samplers Headspace LOOP INJECTORS Auto samplers DIRECT INJECTION USING MICROSYRINGE 5/19/2021 20

SAMPLE INJECTION SYSTEM Headspace: - A headspace sample is normally prepared in a vial containing the sample, the dilution solvent, a matrix modifier and the headspace. - Volatile components from complex sample mixtures can be extracted from non-volatile sample components and isolated in the headspace or gas portion of a sample vial. - A sample of the gas in the headspace is injected into a GC system for separation of all of the volatile components. q q G = the gas phase (headspace) The gas phase is commonly referred to as the headspace and lies above the condensed sample phase. S = the sample phase The sample phase contains the compound(s) of interest. It is usually in the form of a liquid or solid in combination with a dilution solvent or a matrix modifier. Once the sample phase is introduced into the vial and the vial is sealed, volatile components diffuse into the gas phase until the headspace has reached a state of equilibrium as depicted by the arrows. The sample is then taken from the headspace.

DETECTORS The requirements of an ideal detector are: § Applicability to wide range of samples § Rapidity § High sensitivity § Linear response to solutes that extends over several orders of magnitude. § Response should be unaffected by temperature, flow rate… § Detector should be nondestructive. § Temperature range (from room temperature to at least 400 C) § Simple & inexpensive 5/19/2021 22
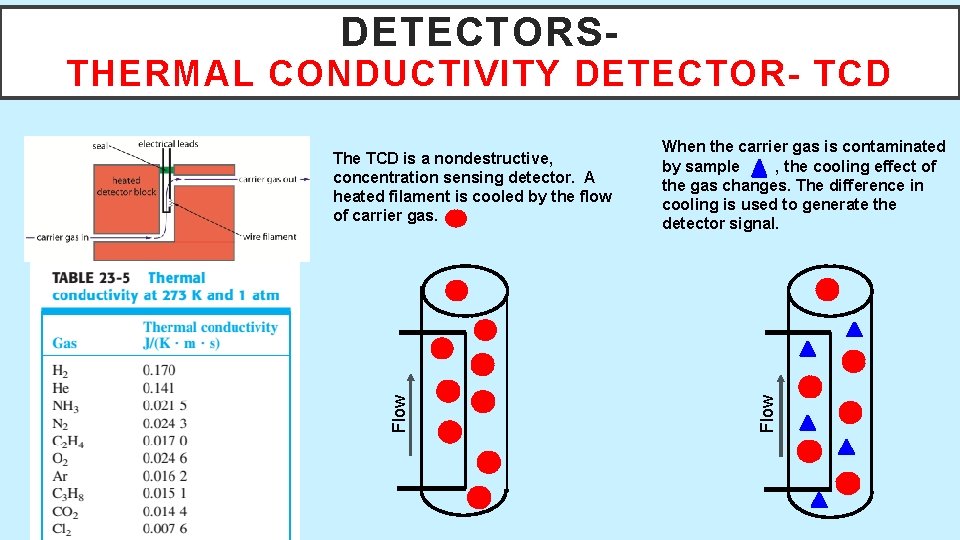
DETECTORS- THERMAL CONDUCTIVITY DETECTOR- TCD When the carrier gas is contaminated by sample , the cooling effect of the gas changes. The difference in cooling is used to generate the detector signal. Flow The TCD is a nondestructive, concentration sensing detector. A heated filament is cooled by the flow of carrier gas.

DETECTORS- THERMAL CONDUCTIVITY DETECTOR- TCD -When a separated compound elutes from the column , thermal conductivity of the mixture of carrier gas and compound gas is lowered. The filament in the sample column becomes hotter than the control column. -The imbalance between control and sample filament temperature is measured by a simple gadget and a signal is recorded -Measures heat loss from a hot filament – -Filament heated to constant Temperature • when only carrier gas flows heat loss to metal block is constant, filament T remains constant. • when an analyte species flows past the filament generally thermal conductivity goes down, T of filament will rise. (resistance of the filament will rise). 5/19/2021 24

DETECTORS- THERMAL CONDUCTIVITY DETECTOR- TCD Advantages Linearity is good Applicable to most organic and inorganic Nondestructive; permits collection of solutes after detection Simple& inexpensive Disadvantages Low sensitivity Affected by fluctuations in temperature and flow rate Biological samples cannot be analyzed 5/19/2021 25
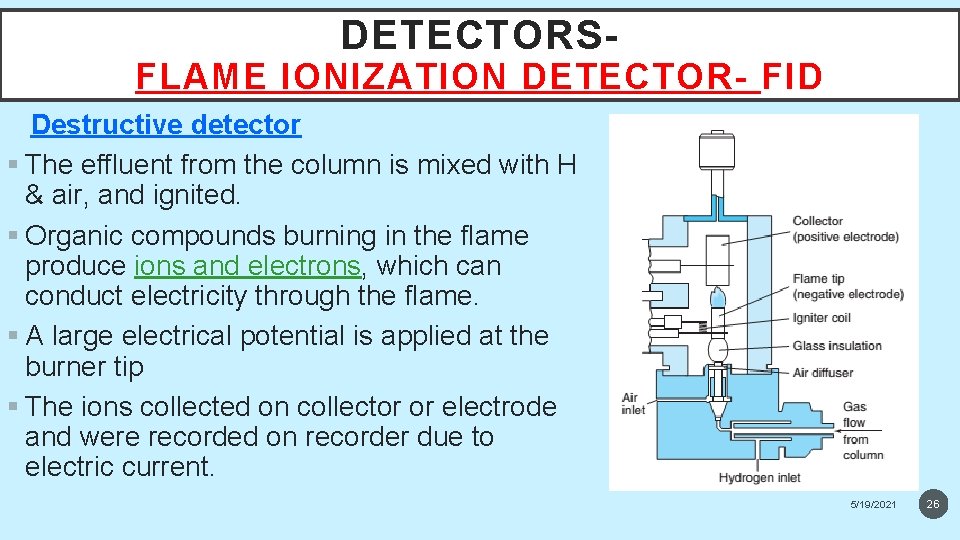
DETECTORS- FLAME IONIZATION DETECTOR- FID Destructive detector § The effluent from the column is mixed with H & air, and ignited. § Organic compounds burning in the flame produce ions and electrons, which can conduct electricity through the flame. § A large electrical potential is applied at the burner tip § The ions collected on collector or electrode and were recorded on recorder due to electric current. 5/19/2021 26
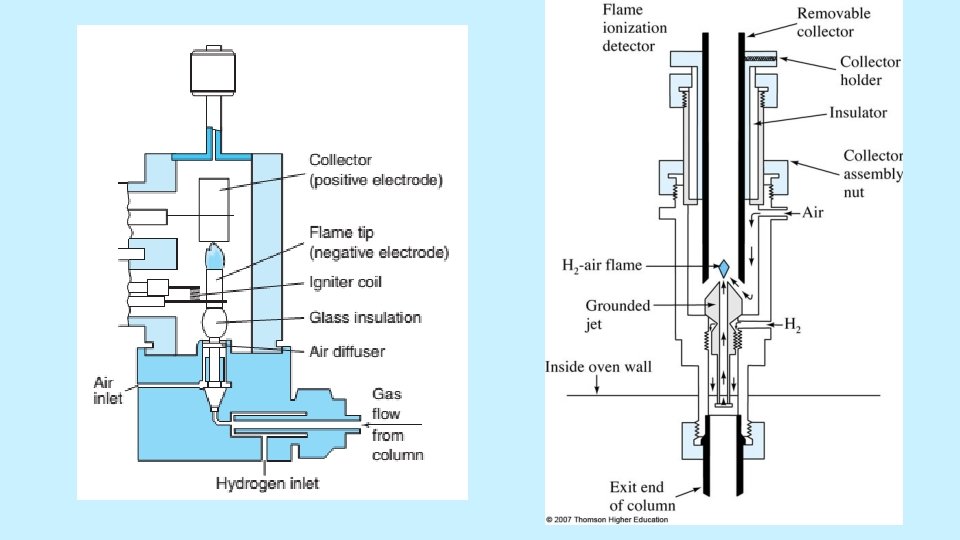

FIDs are mass sensitive rather than conc. sensitive • Advantages: µg quantities of the solute can be detected, Stable, Responds to most of the organic compounds, Linearity is excellent • Disadvantages: destroy the sample 5/19/2021 28

HIGH PERFORMANCE LIQUID CHROMATOGRAPHY - HPLC • Liquid chromatography is important because most compounds are not sufficiently volatile for gas chromatography. High performance liquid chromatography (HPLC) uses high pressure to force solvent through closed column, containing the stationary phase that gives high resolution separations. • The HPLC system consists of a solvent delivery system, a sample injection valve, a high pressure column, a detector and a data system. Many systems include an oven for temperature control of the column. 5/19/2021 29

THE CHROMATOGRAPHIC PROCESS • Increasing the rate at which solute equilibrates between stationary and mobile phases increase the efficiency of chromatography. • For gas chromatography with an open tabular column, rapid equilibration is established by decreasing the diameter of the column, so that molecules can diffuse quickly. Diffusion in liquids is 100 times slower than gases. • Therefore, in liquid chromatography it is not feasible to use open tabular columns. Liquid chromatography is conducted with packed columns. 5/19/2021 31
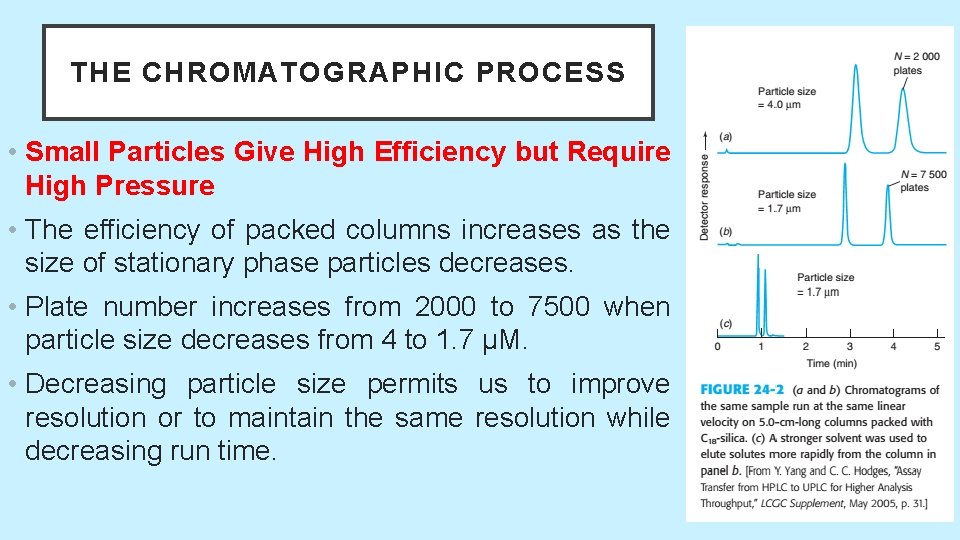
THE CHROMATOGRAPHIC PROCESS • Small Particles Give High Efficiency but Require High Pressure • The efficiency of packed columns increases as the size of stationary phase particles decreases. • Plate number increases from 2000 to 7500 when particle size decreases from 4 to 1. 7 μM. • Decreasing particle size permits us to improve resolution or to maintain the same resolution while decreasing run time. 5/19/2021 32
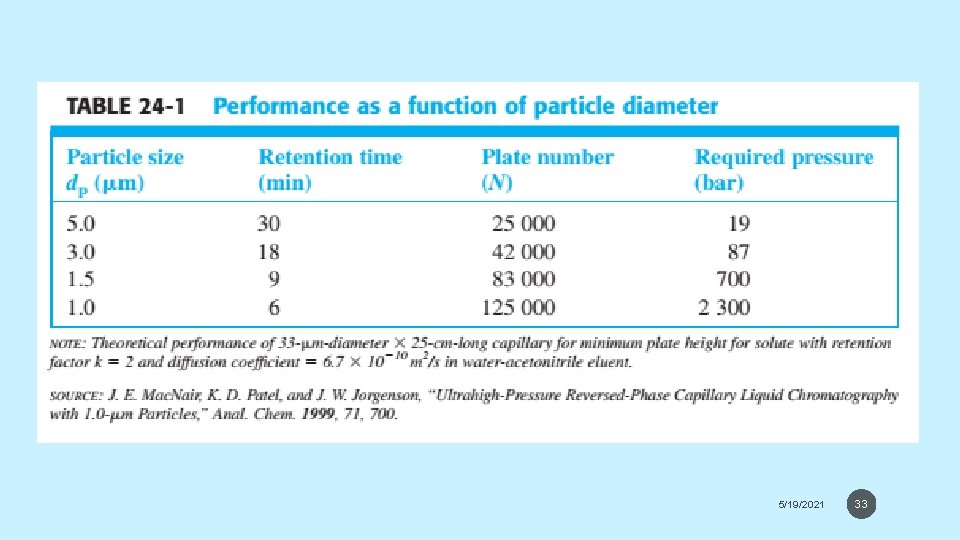
5/19/2021 33

THE COLUMN • Made of Stainless steel tubing for high pressure. • Analytical column: straight, L(5 ~ 25 cm), d (3 ~ 5 mm), dp(3~5 μm). N (40 k ~ 70 k plates/m) • Microcolumn: L (3 ~ 7. 5 cm), d (1 ~ 5 mm), dp: (3 ~ 5 μm) , N: ~100 k plates/m, high speed and minimum solvent consumption • Guard column: protects analytical column, similar packing, remove particulate matter and contamination. • Temperature control: 0. 1 °C - 150 °C. • Column packing: silica, alumina, a polystyrene-divinylbenzene, synthetic or an ion-exchange resin • Pellicular particle: original, spherical, nonporous beads, proteins and large biomolecules separation (dp: 5 μm) • Porous particle: commonly used, dp: 3 ~ 10 μm. Narrow size distribution, porous microparticle coated with thin organic films.

THE STATIONARY PHASE • The most common support is highly pure, spherical, microporous particles of silica as permeable to solvent and have a surface area of hundreds of square meters per gram. • Most silica should not be used at p. H>8, because it dissolves in base. • Bare silica can be used as stationary phase for adsorption chromatography. Most commonly liquid partition chromatography is conducted with a bonded stationary phase covalently attached to the silica surface. 5/19/2021 35
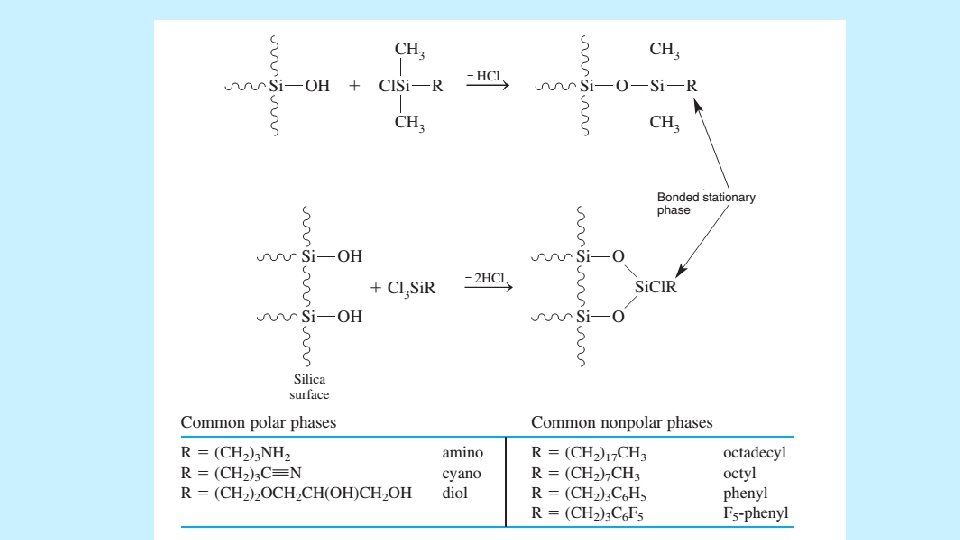
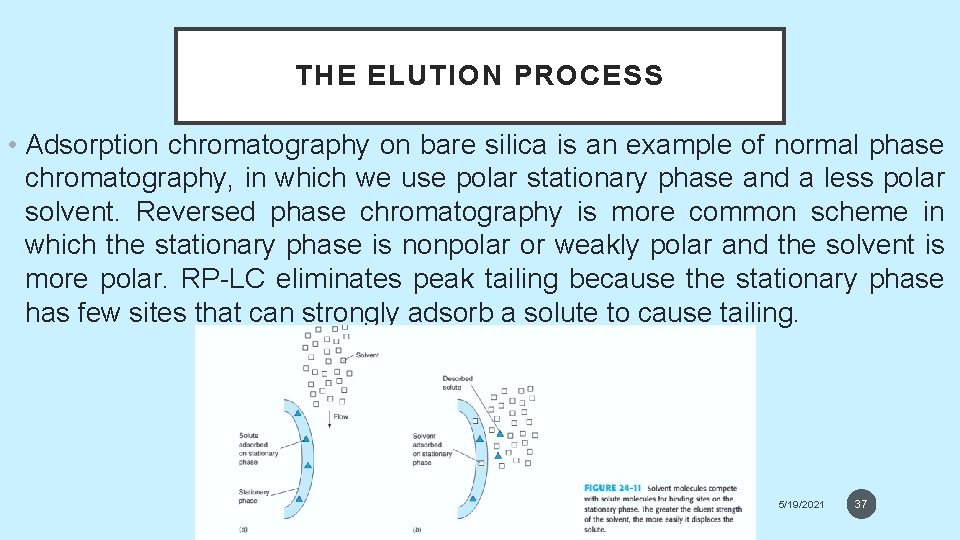
THE ELUTION PROCESS • Adsorption chromatography on bare silica is an example of normal phase chromatography, in which we use polar stationary phase and a less polar solvent. Reversed phase chromatography is more common scheme in which the stationary phase is nonpolar or weakly polar and the solvent is more polar. RP-LC eliminates peak tailing because the stationary phase has few sites that can strongly adsorb a solute to cause tailing. 5/19/2021 37
ISOCRATIC AND GRADIENT ELUTION • Isocratic elution is performed with a single solvent (or constant solvent mixture). • If one solvent does not provide sufficiently rapid elution for all components, then gradient elution can be used. In this case, increasing amounts of solvent B are added to solvent A to cause continuous gradient • Selecting the Separation Mode • There can be many ways to separate components of a given mixture. • Look into a scheme: a decision tree for choosing a stationary phase and Look into Table ranks solvent polarities with the most polar solvents at the bottom. 5/19/2021 38

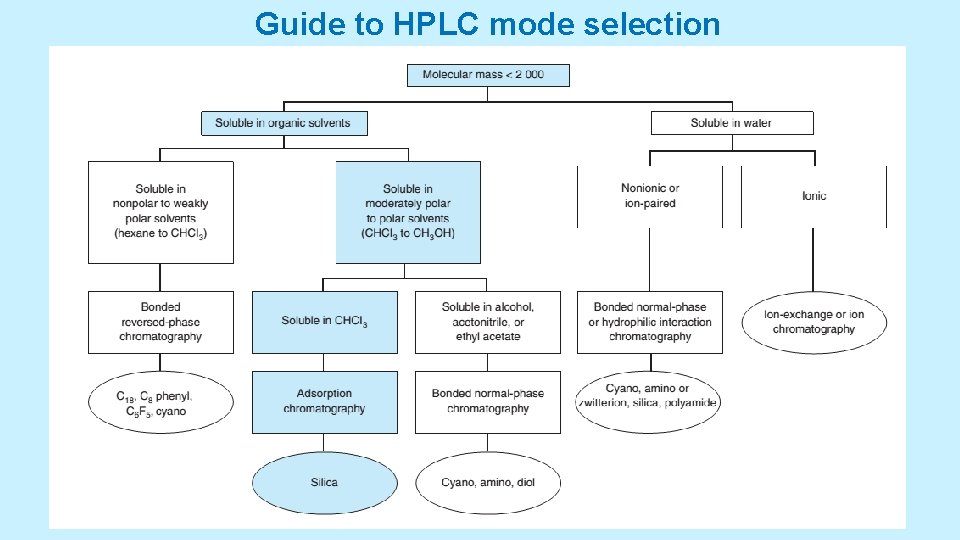
Guide to HPLC mode selection
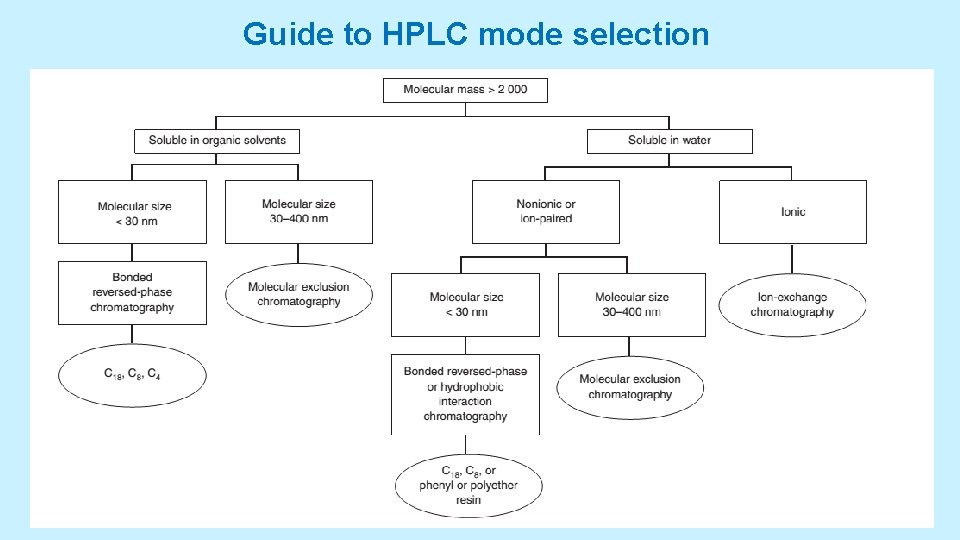
Guide to HPLC mode selection

INJECTION AND DETECTION IN HPLC Pumps and Injection Valves • • The quality of a pump for HPLC is measured by steady and reproducible a flow it can produce. A fluctuating flow rate can create detector noise that obscures weak signals. A typical pump is shown in Figure in next slide. • Gradients made from up to four solvents are constructed by proportioning the liquids through a four-way valve at low pressure and then pumping the mixture at high pressure into the column. The gradient is electronically controlled and programmable in 0. 1 vol% increments. 5/19/2021 42
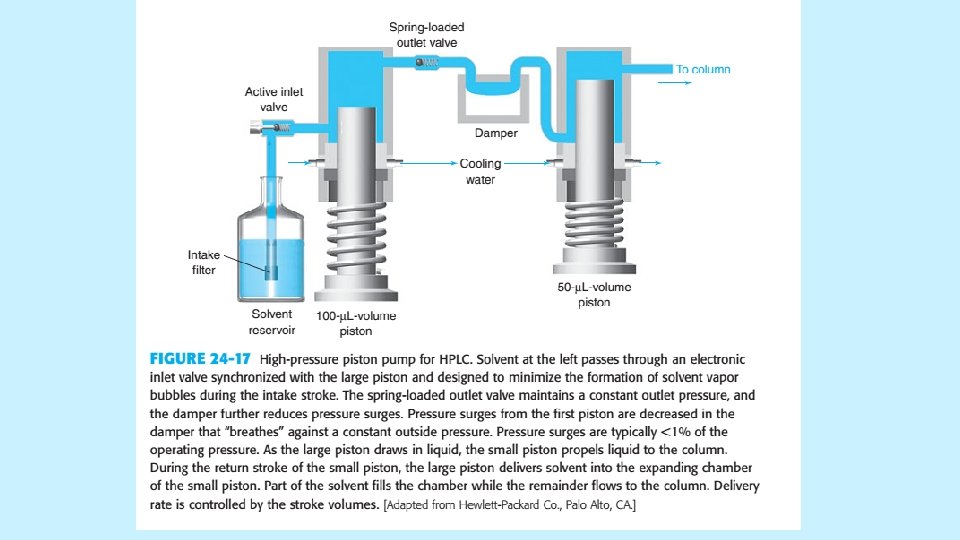

INJECTION AND DETECTION IN HPLC • The injector has interchangeable sample loops, each of which has a fixed volume. In the load position, a syringe is used to wash and load the loop with fresh sample at atmospheric pressure. High-pressure flow from the pump to the column passes through the segment of valve at lower left. When valve is rotated 60 o counterclockwise, the content of the sample loop is injected at high pressure. 5/19/2021 44
DETECTORS • An ideal detector of any type is sensitive to low concentration of every analyte, provide linear response, and does not broaden the eluted peaks. It is also insensitive to changes in temperature and solvent composition. To prevent band broadening, the detector volume should be less than 20% of the volume of chromatographic band. Gas bubbles in the detector creates noise. 5/19/2021 45

SPECTROPHOTOMETRIC DETECTORS • An Ultraviolet detector using a flow cell is the most common HPLC detector, because many solutes absorb UV light. • Simple systems employ the intense 254 -nm emission of a mercury lamp. • More versatile detectors has deuterium, xenon or tungeston lamps and a monochromator so you can choose the optimum UV or visible wavelength for your analytes. • The system uses a photodiode array to record the spectrum of each solute as it eluted. • UV detectors are good for gradient elution of non absorbing solvents 5/19/2021 46
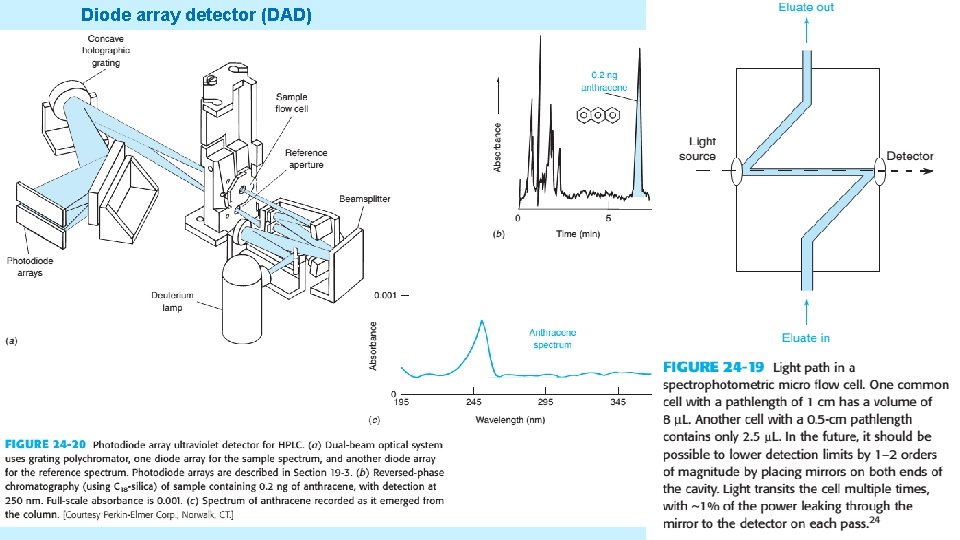
Diode array detector (DAD) 5/19/2021 47

FLUORESCENCE DETECTORS • Fluorescence detectors excite the eluate with laser or powerful UV light and measure the fluorescence. These detectors are very sensitive but respond only to the few analytes that fluoresce. • To increase the utility of fluorescence, fluprescent groups can be attached to the analyte. This process is called derivetization, which it could be pre-column or postcolumn. 5/19/2021 48
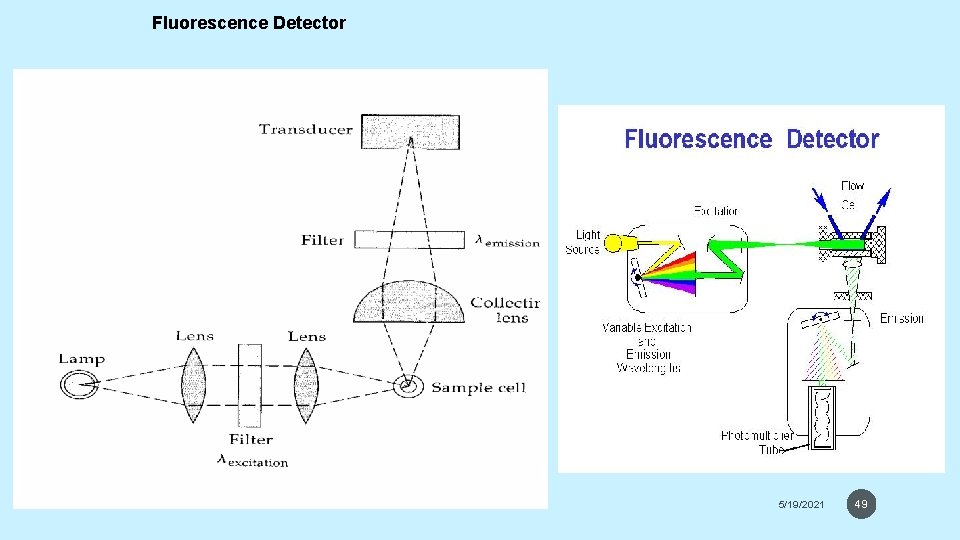
Fluorescence Detector 5/19/2021 49

REFRACTIVE INDEX DETECTORS • Refractive index detectors respond almost to every solute, but its detection limit is about 1000 times less than UV detector. Figure in next slide represents a refractive index detector. • When a solute with a different refractive index enters the cell, the beam is deflected and different pixels of the array are irradiated. RIDs are useless in gradient elution and sensitive to changes in temperature and pressure. Also they have lower sensitivities and dynamic range. They are universal and useful for compounds with little UV absorption. 5/19/2021 50
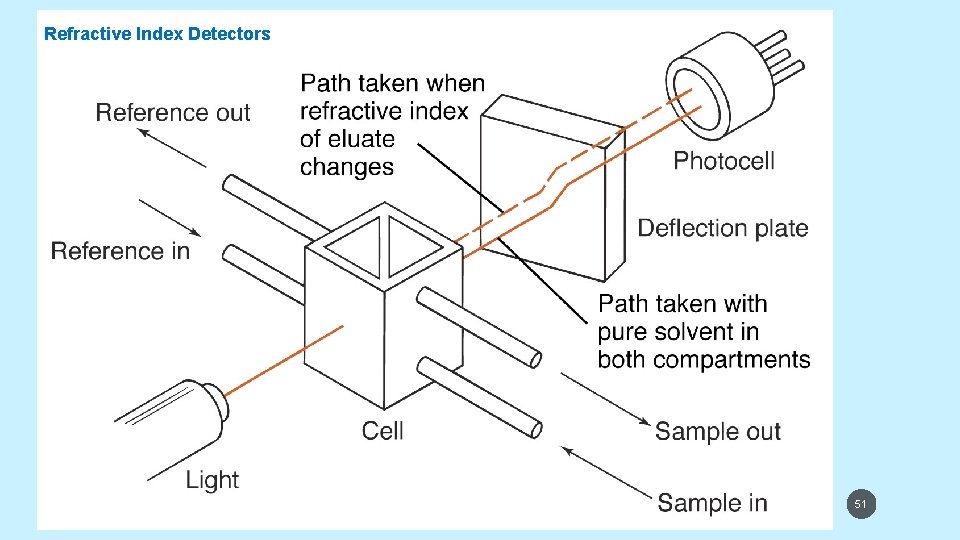
Refractive Index Detectors 5/19/2021 51

ELECTROCHEMICAL DETECTORS • An electrochemical detector responds to analytes that could be oxidized or reduced such as phenols aromatic amines, peroxides, nitrogenphosphorous and halogen containing compounds. • The potential is maintained at constant value between the working and reference electrodes and current is measured between working and auxiliary electrode. • For oxidizable solutes, copper or glassy carbon electrodes are common. • For reducible solutes, dropping mercuric electrodes are common. • Current is proportional to solute concentration over six orders of magnitude. Dissolved oxygen must be removed. • The detector is sensitive to temperature and flow rate and requires frequent cleaning. 5/19/2021 52
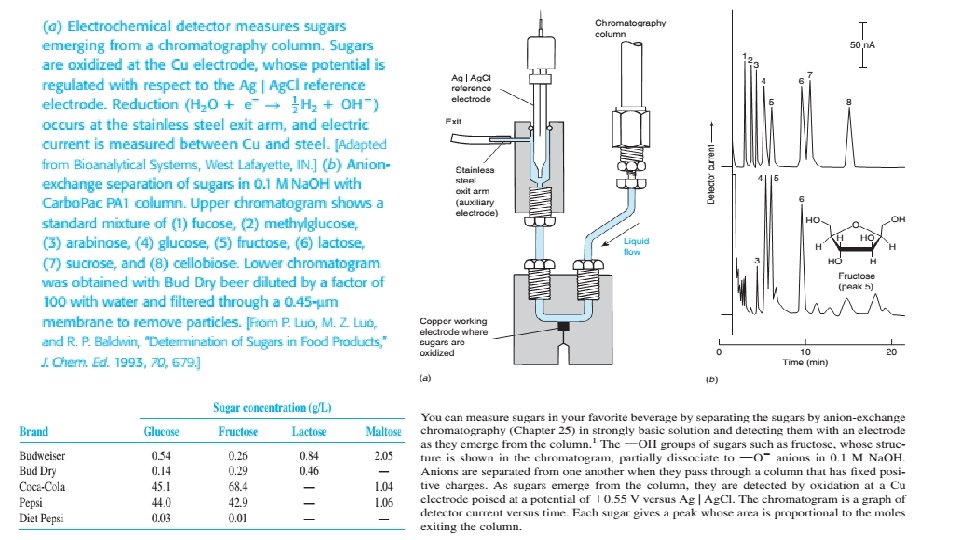
5/19/2021 53

METHOD DEVELOPMENT FOR REVERSE-PHASE SEPARATIONS (ISOCRATIC) • Developed method should be rugged, which means the separation will not be seriously degraded by gradient deterioration of the column, small variations in solvent composition, p. H, and temperature or the use of columns from different manufacturers. • As in gas chromatography the goals of analysis to choice of detector will be followed. Additionally the followings should be optimized. • Criteria for an Adequate Separation • Keep capacity ratios (K’) in for all peaks in the range 0. 5 to 20. For quantitation a minimum resolution of 1. 50 should be maintained and for ruggedness 2. 0 is preferable. Operating pressure should be maintained 54 5/19/2021

METHOD DEVELOPMENT FOR REVERSE-PHASE SEPARATIONS (ISOCRATIC) • Optimization with One Organic Solvent • Combination of acetonitrile, methanol and tetrahydrofuran (THF) with water or aqueous buffer provide a sufficient range of bipolar and hydrogen-bonding interactions with solutes to separate a vast number of components. Starting conditions are listed in Table at next slide. 5/19/2021 55

5/19/2021 56
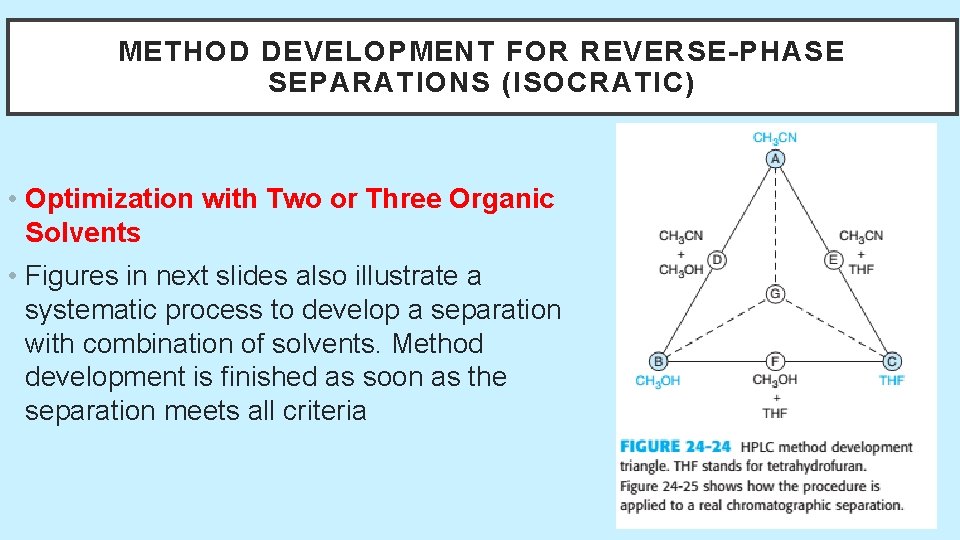
METHOD DEVELOPMENT FOR REVERSE-PHASE SEPARATIONS (ISOCRATIC) • Optimization with Two or Three Organic Solvents • Figures in next slides also illustrate a systematic process to develop a separation with combination of solvents. Method development is finished as soon as the separation meets all criteria 5/19/2021 57
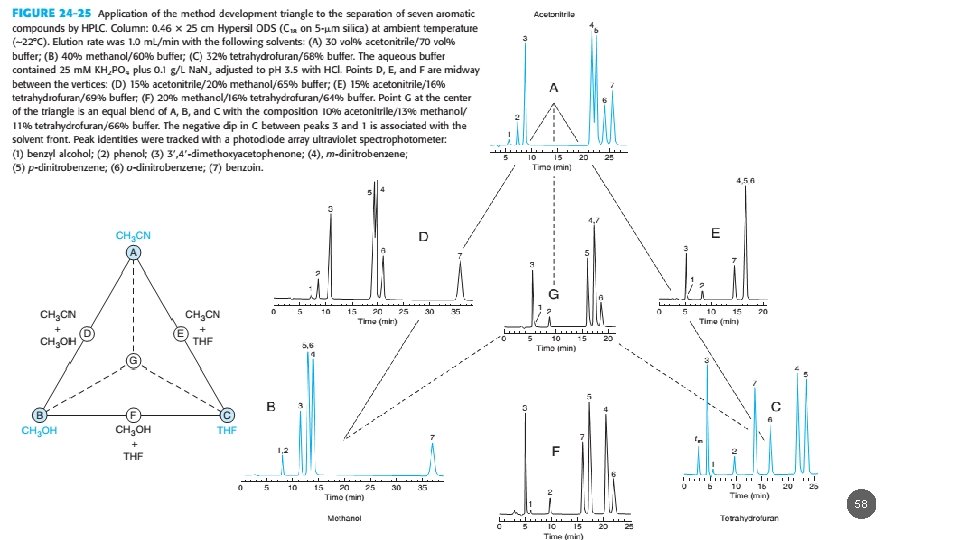
5/19/2021 58

METHOD DEVELOPMENT FOR REVERSE-PHASE SEPARATIONS (ISOCRATIC) • Temperature as a Variable • Column temperature affects relative retention of different compounds and elevated temperatures permits high speed chromatography to be conducted. For elevated temperature operation, p. H should be below 6 to retard dissolution of silica. • Choosing a Stationary Phase • C 18 silica is the most common stationary phase and it separates a wide range of mixtures when solvents is chosen carefully. • Table in next slide is a guide to selecting other bonded phases. The following example suggests an approach for resolving six compounds in shorter time. 5/19/2021 59

METHOD DEVELOPMENT FOR REVERSE-PHASE SEPARATIONS (ISOCRATIC) 5/19/2021 60
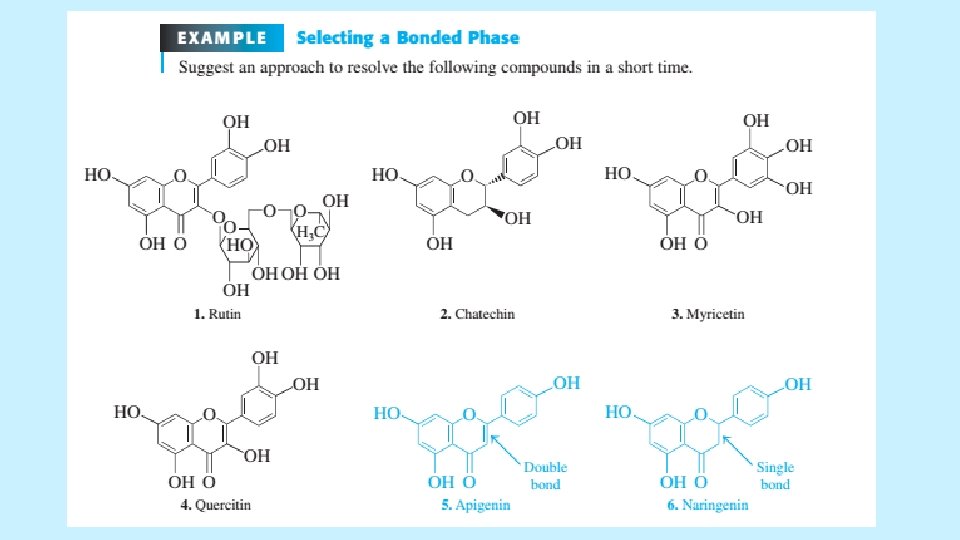
5/19/2021 61
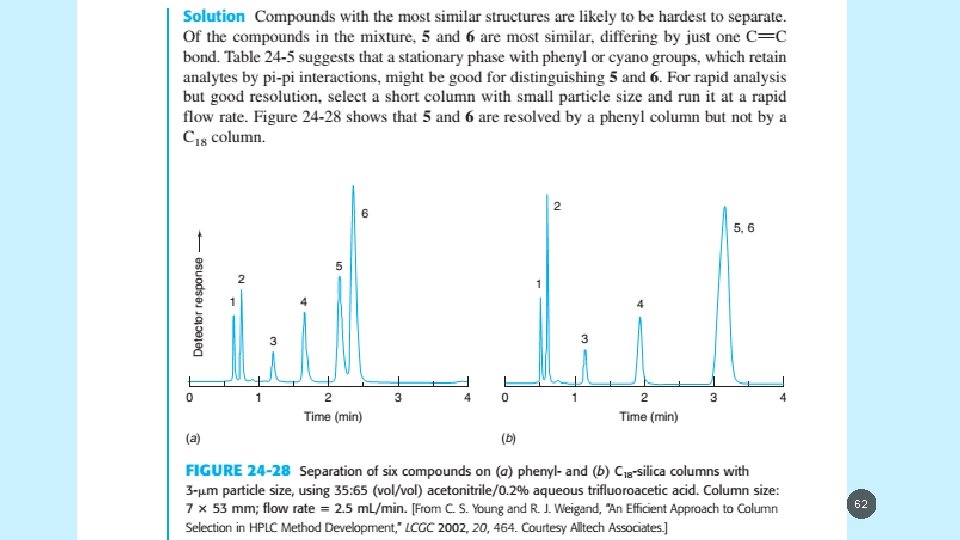
5/19/2021 62
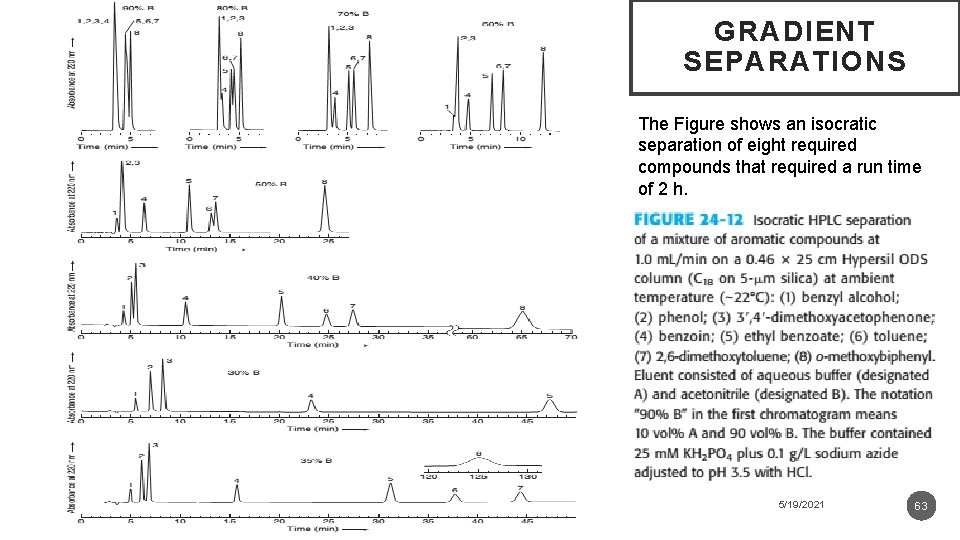
GRADIENT SEPARATIONS The Figure shows an isocratic separation of eight required compounds that required a run time of 2 h. 5/19/2021 63
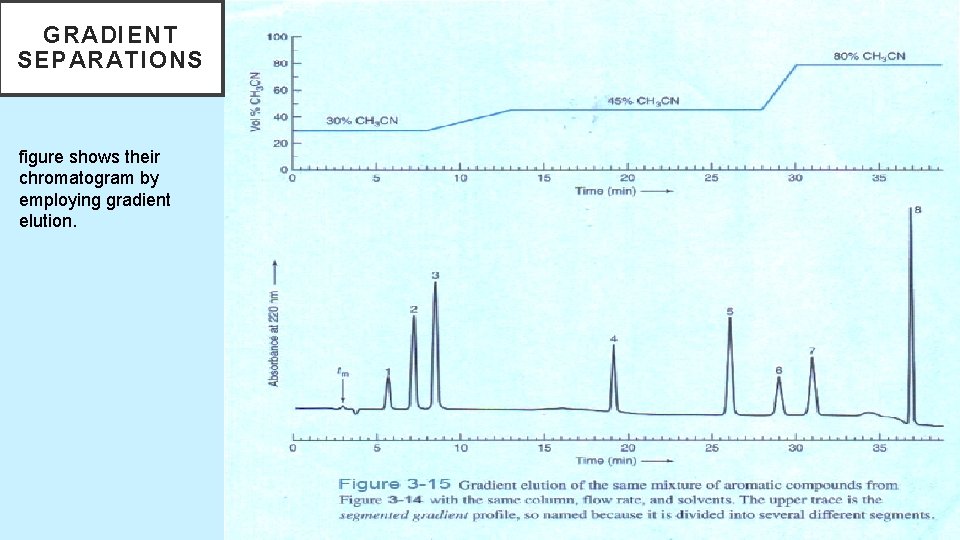
GRADIENT SEPARATIONS figure shows their chromatogram by employing gradient elution. 5/19/2021 64

P 9 RP- HPLC SALIENT FEATURES. • Reverse phase HPLC. • 15 min method, RT- Between 4 to 6 min. • Mobile phase : A - Water + TFA B - ACN + TFA • Column: Brand Name - Discovery • Type of Column: Reverse phase C 18 column • Specification of Column. Diameter: 4. 6 mm Length: 50 mm Porosity: 300 • A, Particle size: 5 Micron • Guard column Brand Name - Zorbex 300 SB (Agilent) • Type of Column: Reverse phase Zorbax 300 SB-C 18 • Specification of Column. Diameter: 4. 6 mm, Length: 35 mm. Porosity: 300 • A, Particle size: 5 Micron
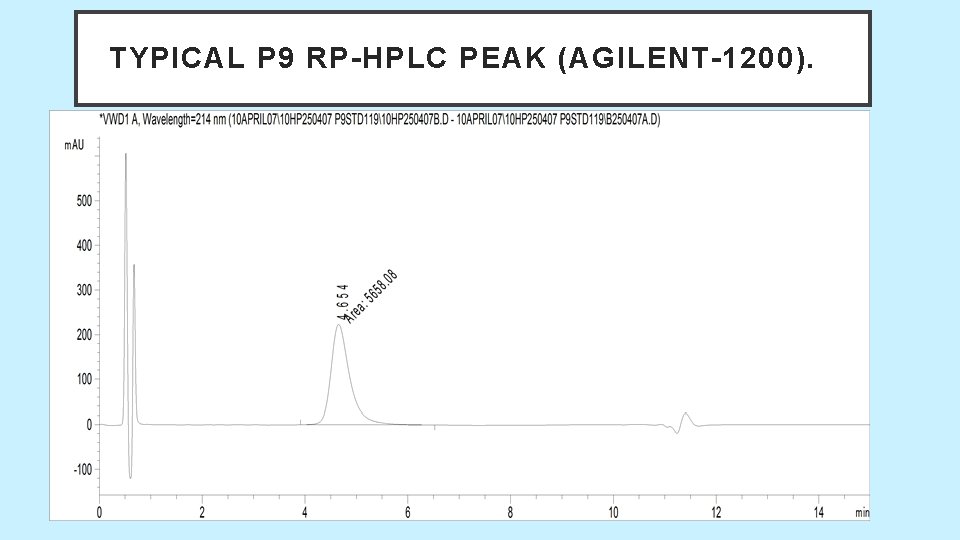
TYPICAL P 9 RP-HPLC PEAK (AGILENT-1200).

VALIDATION OF CHROMATOGRAPHIC METHODS • Most of chromatographic methods are concerned of traces and ultra-traces of organic analytes, that could be organic pollutants or toxic chemicals or drugs. • Matrices vary between drinking water to food to biological fluids. • The nature of analyte (polarity, volatility and molecular mass) determines the separation method of being based on GC or HPLC. • Within the separation method proper mobile phases, stationary phases and detectors should be carefully optimized. • Almost, standaed methods are available for every common analyte with various separation and detection methods. • Remember, derivatization of analyte enhances separation and/or detection. 67

VALIDATION OF CHROMATOGRAPHIC METHODS • GC methods don’t accept direct aqueous injection, while LC methods require injection of clean samples, otherwise columns will be destructed. Thus, a suitable extraction method must be followed. • Extraction methods usually transfer and concentrate analytes into more friendly matrix that could be compatible with the chromatographic instrument. Selected method should satisfy highest recoveries of analytes and elimination of most endogenous compounds that may interfere with analyte and shorten the life of the column. 5/19/2021 68

VALIDATION OF CHROMATOGRAPHIC METHODS • Two common extraction methods are available. The liquid-liquid extraction (LLE) and the solid phase extraction (SPE). The most disadvantage of LLE is the limited number of immiscible liquids with aqueous and for SPE the most compatible stationary phase. • Detection methods are another issue, MS methods are emerging as more convenient detectors with highest sensitivities and capabilities of identification. However, large budget is required to obtain, run and maintain these instruments. 5/19/2021 69

VALIDATION OF CHROMATOGRAPHIC METHODS • Most laboratories follow standard methods in their fields. If they are lucky the method will satisfy 80% of their needs. Thus, modification and development are continuous process in analytical laboratories. • Development could include sample preparation, separation and detection. Developed methods should be validated. • Validation methods include, accuracy, precision, linearity, stability, specificity, recovery and ruggedness 5/19/2021 70

VALIDATION OF CHROMATOGRAPHIC METHODS • Validation involves documenting, through the use of specific laboratory investigation, that the performance characteristics of the methods are suitable and reliable for intended applications, The acceptability of analytical data corresponds directly to the criteria used to validate the method. In our discussion we assume that our method is concerned with determination of drug in serum. 5/19/2021 71

VALIDATION OF CHROMATOGRAPHIC METHODS • Reference Standard • Analysis of drugs and their metabolites in biological matrix is carried out using samples spiked with calibration (reference) standards and using quality control samples (QC), which are used to monitor the performance of the method and to assess the validity of the results of unknown samples analyzed. • Method Development • Typical method development and establishment of a bioanalytical method include determination of selectivity, sensitivity, accuracy, precision. Recovery, calibration curve and stability of analyte in spiked samples. 72

VALIDATION OF CHROMATOGRAPHIC METHODS • Selectivity is the ability of an analytical method to differentiate and quantify the analyte in the presence of other components in the samples. • For selectivity, analyte of blank samples of the appropriate biological matrix should be obtained from at least six sources. Each blank should be tested for interference, and selectivity should be ensured at the lower limit of quantitation (LLOQ). • Potential interfering substances in a biological matrix include endogenous matrix components, metabolites, decomposition products, and in the actual study, concomitant medication and other xenobiotics. If the method is intended for more than one analyte, each should be tested to insure that there is no interference. 73

VALIDATION OF CHROMATOGRAPHIC METHODS Accuracy • The accuracy of an analytical method describes the closeness of mean test results obtained by the method to true value of analyte. • Accuracy is determined by replicate analysis of samples containing known amounts of analyte. • Accuracy should be measured using a minimum of five determinations per concentration. A minimum of three concentrations in the linear range are recommended. The mean should be within 15% of the actual value except at LLOQ, where should not deviate by more than 20%. The deviation of the mean from the true value (Bias) serves as a measure of accuracy. 5/19/2021 74

VALIDATION OF CHROMATOGRAPHIC METHODS Precision • The precision of analytical method describes the closeness of individual measures of an analyte when the procedure is applied repeatedly to multiple aliquots of a single homogeneous volume of biological matrix. • Precision should be measured using a minimum of five determinations per concentrations. A minimum of three concentrations in the linear range are recommended. The precision determined at each concentration level should not exceed 15% except that at LLOQ (20%) of the coefficient of 5/19/2021 75

VALIDATION OF CHROMATOGRAPHIC METHODS Recovery • The recovery of the analyte in an assay is the detector response obtained from an amount of analyte added to and extracted from the biological matrix, compared to the detector response obtained for the true concentration of the pure authentic standard. Recovery pertains the extraction efficiency of an analytical method within the limits of variability. • Recovery experiments should be performed by comparing the analytical results for extracted samples at three concentrations with unextracted standards that represent 100% recovery. 5/19/2021 76

VALIDATION OF CHROMATOGRAPHIC METHODS Calibration Standard Curve • A calibration curve is the relationship between instrument response and concentration of analyte. • A calibration curve should be generated for each analyte. • A sufficient number of standards should be used adequately to define the relationship between concentration and response. • A calibration curve should be prepared in the same biological matrix as the sample in the intended study by spiking the matrix with known concentration of analytes. • A calibration curve should include a blank sample processed without internal standard, a zero sample with internal standard and six to eight non -zero samples covering the expected range including (LLOQ). 77

VALIDATION OF CHROMATOGRAPHIC METHODS • Internal Standard • The use of internal standard is preferred by many analytes for quantitative analysis. Its purpose is to minimize system and procedure variations, thus eliminating variations of precision as a function of sample size. • Selected internal standard should be completely resolved, not elute over another component, similar chemical properties, high purity and chemically inert and stable. • The addition of internal standard makes the analysis less influenced by errors in transferred and injected volumes. 5/19/2021 78

VALIDATION OF CHROMATOGRAPHIC METHODS Stability • Drug stability in a biological fluid is a function of the storage conditions; the chemical properties of the drug, the matrix and the container system. • The stability of the analyte in a particular matrix and container system is relevant only to the matrix and container system and should not be extended to other matrices and container systems. • Stability procedures should evaluate the stability of analytes during sample collection and handling after long term (frozen at intended storage temperature) and short term (bench top, room temperature) storage, and after going through freez and thaw cycles and the analytical process. 5/19/2021 79

VALIDATION OF CHROMATOGRAPHIC METHODS Stability • Conditions used in stability experiments should reflect situations likely to be encountered during actual sample handling and analysis. • The procedure should also include an evaluation of analyte stability in stock solutions. Specificity • Specificity is related to common drugs the patient may take with studied drug. • These drugs include caffeine, salicyclic acid, acetaminophen, metronidazole, ibuprofen, nicotine and sulfamethoxazole. These compounds should not be detected by the method or elute at different time 5/19/2021 80

THANK YOU 5/19/2021 81
- Slides: 81