Adsorption Theory of Heterogenous catalysis Explains mechanism Old









- Slides: 15

Adsorption Theory of Heterogenous catalysis

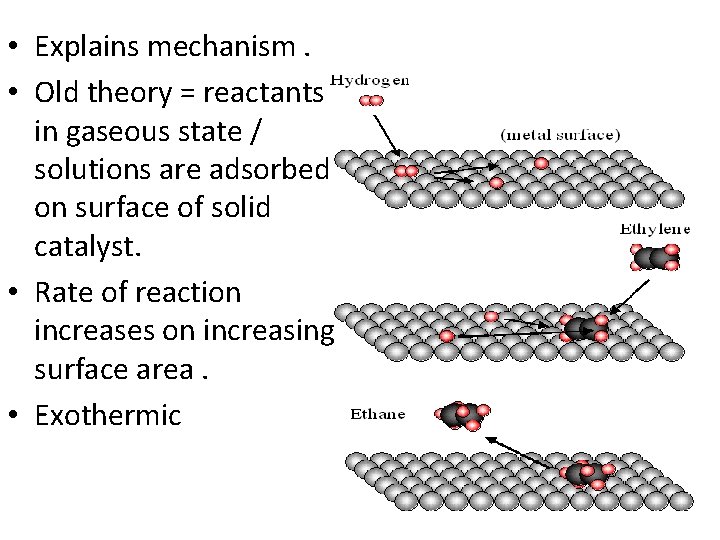
• Explains mechanism. • Old theory = reactants in gaseous state / solutions are adsorbed on surface of solid catalyst. • Rate of reaction increases on increasing surface area. • Exothermic

• Modern Theory-combination of intermediate compound theory and & old adsorbtion theory. • Steps – • 1. Diffusion • 2. Adsorption • 3. Formation of intermediate. • 4. Desorption • 5. Diffusion

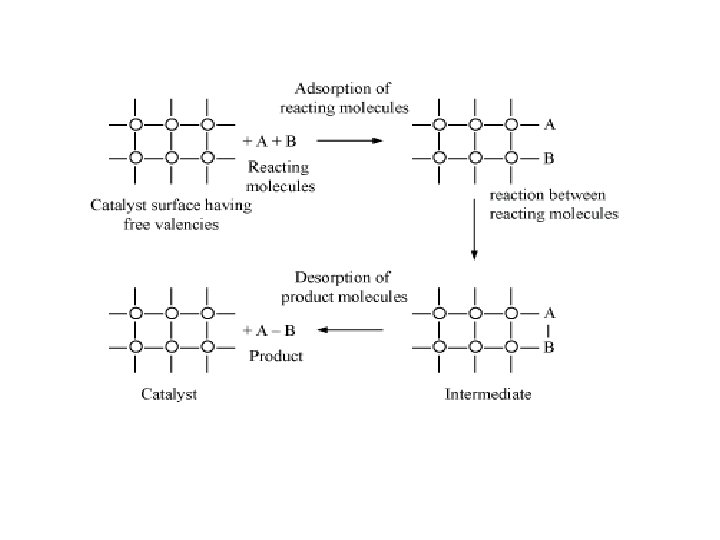
• Free valencies present on surface catalysts. • Provides seats for chemical raction. • When gas molecules comes in contact , they held up due to loose combination. • • Side by side adsorbtion results in formation of new molecule.


Important features of solid catalysts • Activity –. Depends upon the strength of chemisorption to a large extent. Reactant must get adsorbed on catalyst to become active. Catalytic activity –maximum in Gp. 7 to Gp 9 metals

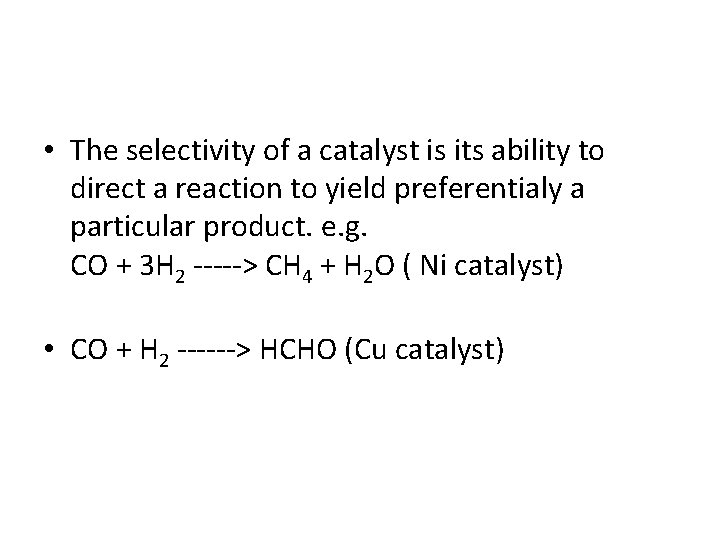
• The selectivity of a catalyst is its ability to direct a reaction to yield preferentialy a particular product. e. g. CO + 3 H 2 -----> CH 4 + H 2 O ( Ni catalyst) • CO + H 2 ------> HCHO (Cu catalyst)

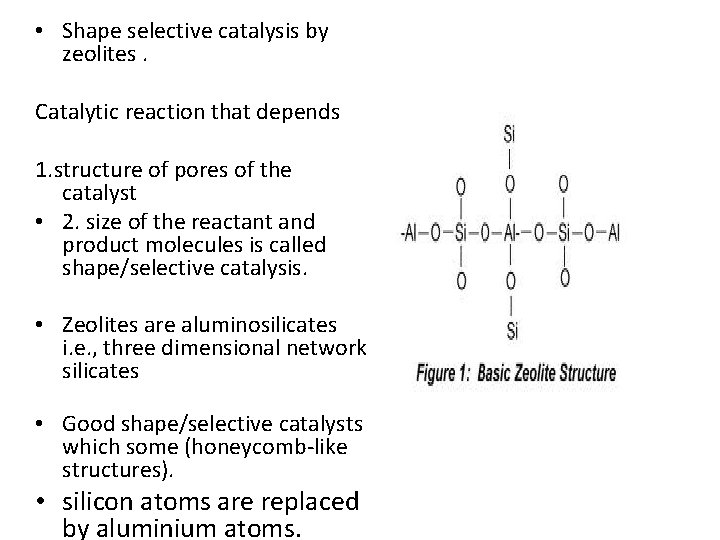
• Shape selective catalysis by zeolites. Catalytic reaction that depends 1. structure of pores of the catalyst • 2. size of the reactant and product molecules is called shape/selective catalysis. • Zeolites are aluminosilicates i. e. , three dimensional network silicates • Good shape/selective catalysts which some (honeycomb-like structures). • silicon atoms are replaced by aluminium atoms.

• Found in nature as well a synthesized • Zeolites, before as catalysts, are heated in vacuum so that the water of hydration is lost. • As a result, zeolite becomes porous i. e. , the cavities in the cage-like structure’ • size of the pores - 260 pm and 740 pm. Petroleum industry –ZSM-5.

Enzyme catalysis • • Complex nitrogeneous organic compounds. Sources –Plants & animals. Protein molecules of high molecular mass. Forms colloidal solutions in water. Effective catalysts. Biological catalysts. First enzyme-synthesised in 1969.

• Examples • Inversion of cane sugar. • Conversion of glucose into ethyl alcohol.

Characteristics of enzyme catalysis 1. Hihly efficient 2. Highly specific nature 3. Highly active under optimum T(298 -310) 4. Highly active under optimum p. H(5 -7) 5. Increasing activity in presence of activators & coenzymes –(non protein +enzymes) • 6. Influence of inhibitors &poisons. • • •

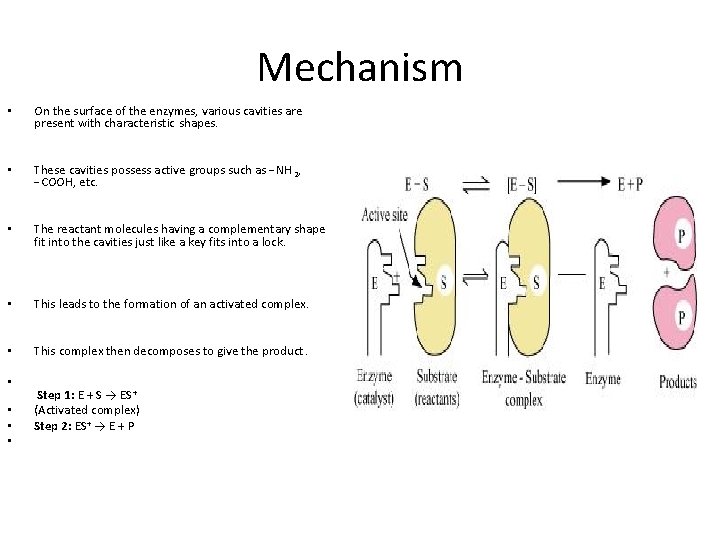
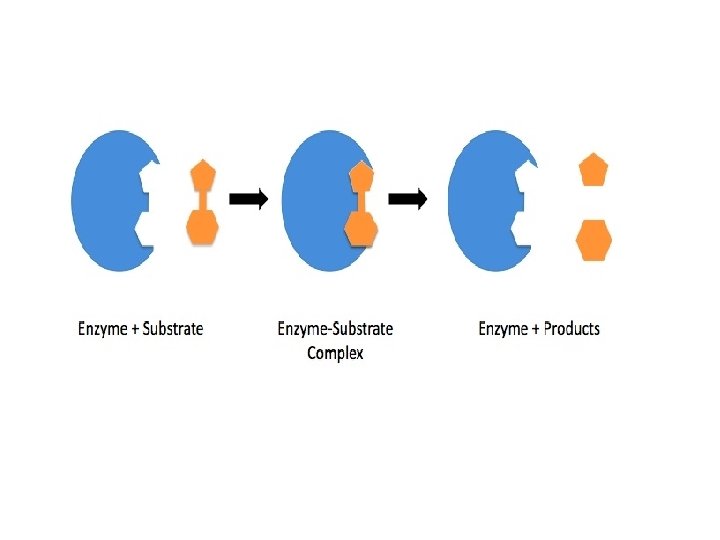
Mechanism • On the surface of the enzymes, various cavities are present with characteristic shapes. • These cavities possess active groups such as −NH 2, −COOH, etc. • The reactant molecules having a complementary shape fit into the cavities just like a key fits into a lock. • This leads to the formation of an activated complex. • This complex then decomposes to give the product. • • Step 1: E + S → ES + (Activated complex) Step 2: ES+ → E + P


Catalysts in industry • Haber’s process –Ammonia • Conditions –Finely divided Fe, Mo/200 bar/723773 K • Ostwalds process –Nitric acid • Conditions-Platinised aesbestos /573 K • Contact process • Condition-V 2 O 5/pt aesbestos /673 -723