Break v Link between Thermodynamics and Kinetics Kinetics
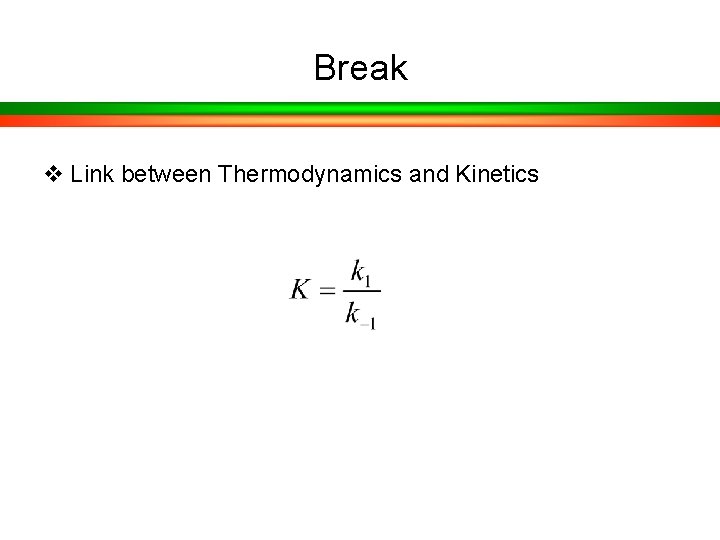
Break v Link between Thermodynamics and Kinetics

Kinetics Modern Methods in Heterogeneous Catalysis F. C. Jentoft, November 1, 2002

Outline v Motivation and Strategy v Some Important Concepts v Rate Equations v Mechanisms and Kinetics v Temperature Dependence of Rate Constant v Compensation Effect

What Kinetics Will (Not) Deliver… v Reaction rates v Rate equation / reaction order v Rate constant v Apparent activation energies v Will not deliver a mechanism…. . v But any mechanism we think of should be consistent with the kinetic data….
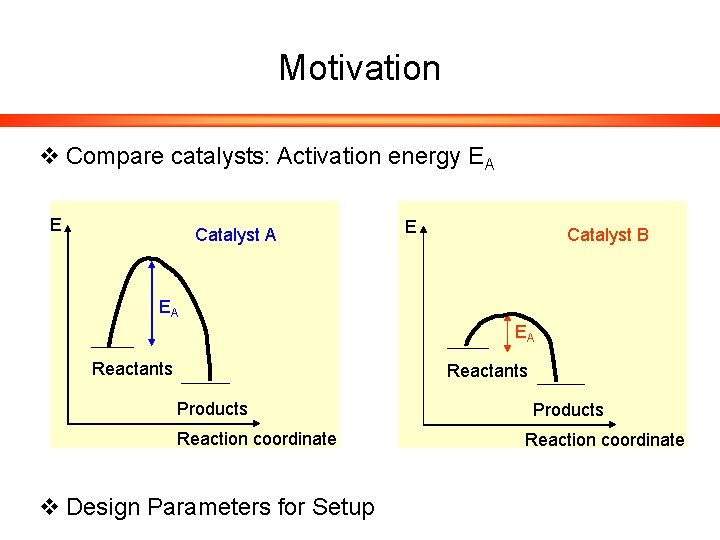
Motivation v Compare catalysts: Activation energy EA E Catalyst B EA EA Reactants Products Reaction coordinate v Design Parameters for Setup Products Reaction coordinate

Microscopic Reversibility k 1 A+B AB k-1 k 2 k 3 k-2 k-3 A* + B v Equilibrium conditions v Unidirectional reaction with identical rates is not an option

Steady State Approximation A k 1 B k 1* C v Bodenstein’s approximation for consecutive reactions If k 1*>>k 1, then v Simplifies Rate Equations

Rate Equations I v Typical rate equation: v With a, b, c, the individual reaction order with respect to a particular reactant and the total reaction order n the sum of the exponents v With r the reaction rate in units of mol/l per time

Rate Equations II v Typical rate equation: v With k the rate constant in units of min-1 for a first order reaction, for higher orders in inverse units of concentration in different powers

Catalysis in Solution: Specific Acid / Base Catalysis v Proton donor: H 3 O+ (solvated protons) Proton acceptor: OHv Rate equation (analogous for base catalysis) pseudo 1 st order v Rate constant a linear function of p. H

Specific Acid Catalysis v Dependence of the observed rate constant for oximation of acetone on p. H at 25°C. The rate equation is r = kobs * Cacetone
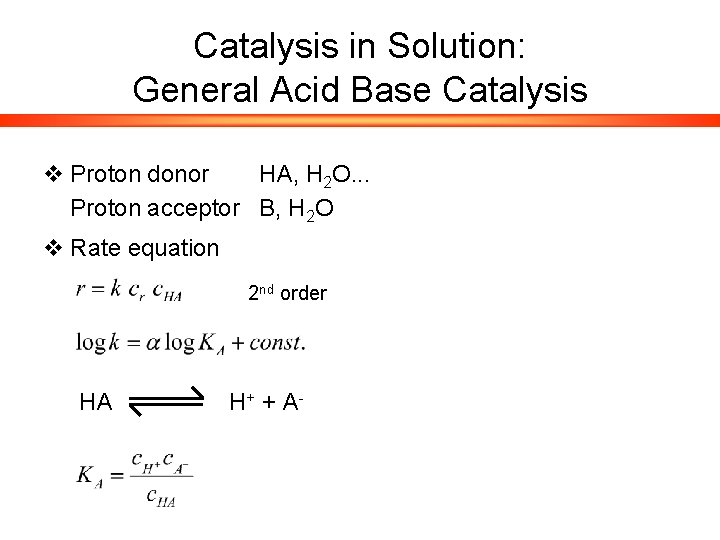
Catalysis in Solution: General Acid Base Catalysis v Proton donor HA, H 2 O. . . Proton acceptor B, H 2 O v Rate equation 2 nd order HA H+ + A -

General Acid Catalysis
Rates in Heterogeneous Catalysis v Rate with respect to mass or surface area
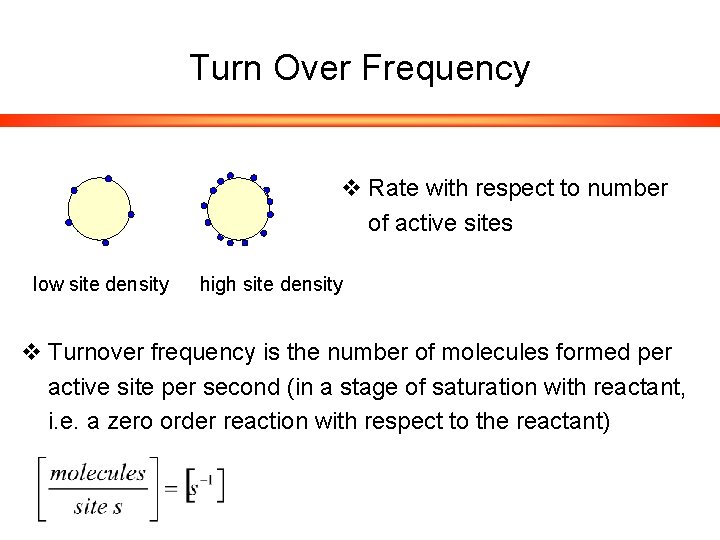
Turn Over Frequency v Rate with respect to number of active sites low site density high site density v Turnover frequency is the number of molecules formed per active site per second (in a stage of saturation with reactant, i. e. a zero order reaction with respect to the reactant)

TOF, TON, Catalysis v TON Total number of product formed molecules per active site TON= TOF*catalyst life time v TON = 1 TON 102 TON = 106 -107 stoichiometric reaction catalytic reaction industrial application v TON origins from enzyme kinetics, definitions vary

Examples for TOFs

Reaction Steps in Heterogeneous Catalysis v Diffusion of reactant to catalyst v Adsorption of reactant on catalyst surface v Reaction v Desorption of products from catalyst surface v Diffusion of products away from catalyst We want to know the reaction kinetics. Diffusion should thus not be a rate limiting step.

Interfacial Gradient Effects v Mass transfer bulk of fluid to surface v Case 1: reaction at surface instantaneous global rate controlled through mass transfer “diffusion control”, favored at high T v Case 2: reactant concentration at surface same as in bulk fluid global rate controlled through reaction rate “reaction controlling”, favored at low T and high turbulence

Intraparticle Gradient Effects v Mass transfer within the pores of a catalyst v Vary particle size!

Langmuir Hinshelwood Mechanism A B v Both species are adsorbed, adsorption follows Langmuir isotherm (see class next week)

Eley Rideal Mechanism B A v Only one species is adsorbed, adsorption follows Langmuir isotherm

How to Derive a Rate Equation I 2 C 2 H 5 OH H+ C 2 H 5 -O-C 2 H 5 + H 2 O
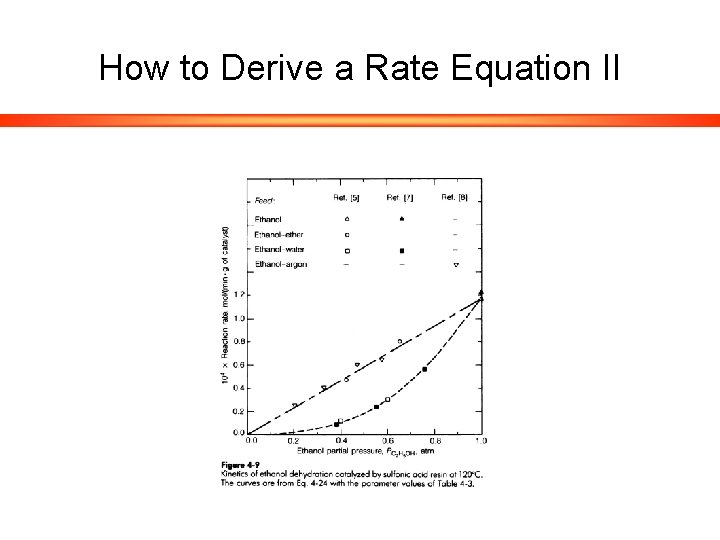
How to Derive a Rate Equation II
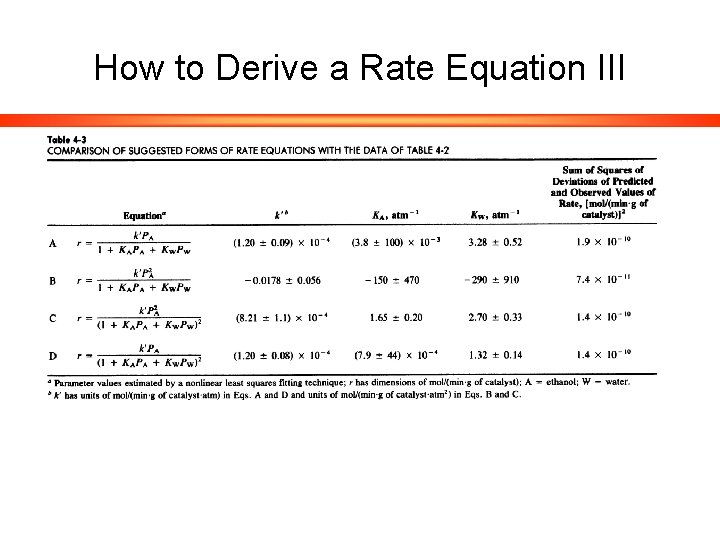
How to Derive a Rate Equation III
Structure Insensitivity v rate per exposed metal surface area is NOT a function of the metal particle size v active site 1 -2 atoms v Example: the hydrogenation of cyclohexene + H 2

Structure Insensitivity

Structure Sensitivity v rate per exposed metal surface area is a function of the metal particle size / the exposed facet plane v active site an ensemble of atoms v Example: the hydrogenolysis of ethane C 2 H 6 + H 2 2 CH 4 v also: ammonia synthesis (reactions involving C-C, N-N bond breaking)

Structure Sensitivity

Temperature Dependence of Rate Constant v Once a rate equation has been established, a rate constant can be calculated v The rate constant is temperature dependent v There are three different ways to derive this relation: Arrhenius Theory Collision Theory Transition State Theory (Eyring)
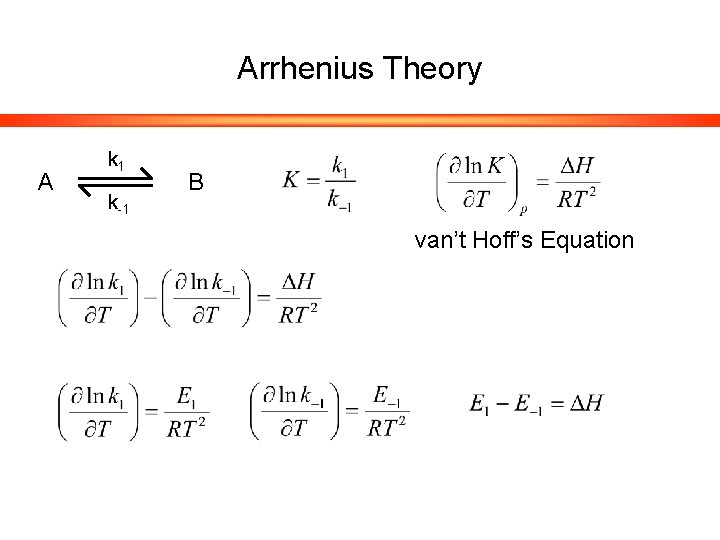
Arrhenius Theory A k 1 k-1 B van’t Hoff’s Equation

Arrhenius Theory v With E the apparent activation energy in k. J mol-1 A the frequency factor v Plot of ln k vs. 1/T gives a slope of -EA/R which allows the calculation of the activation energy v A rule of thumb: the rate doubles for 10 K rise in temperature

Collision Theory v According to the simple collision theory, the preexponential factor is dependent on T 1/2 v with NA Avogadro’s number, σ cross section, μ reduced mass, k Boltzmann’s constant

Activated Complex Theory A + BC A B C v Evans/Polanyi, Eyring v based on statistical thermodynamics AB + C

Results of Activated Complex Theory v Rate constant (based on number of moles) v Function of T v From the equilibrium constant for the activated complex, a standard free enthalpy of activation can be calculated
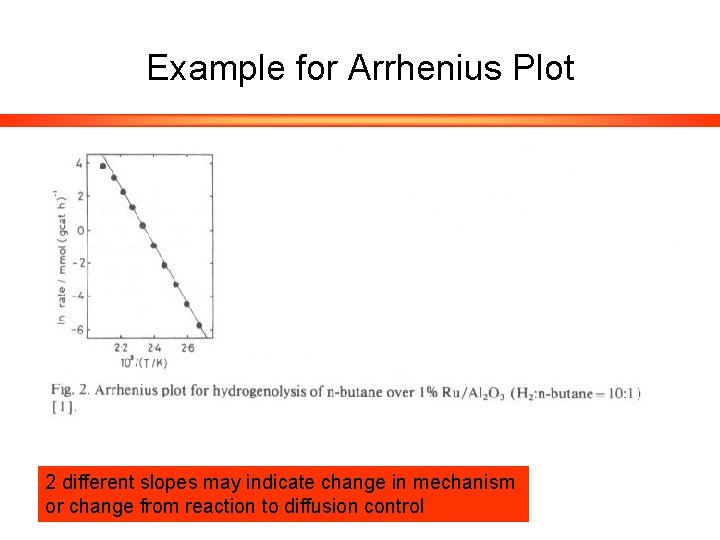
Example for Arrhenius Plot 2 different slopes may indicate change in mechanism or change from reaction to diffusion control

Compensation Effect v A “sympathetic variation of the activation energy with the ln of the pre-exponential factor” v ln A and EA/RT have the same order of magnitude but different signs v Change in EA may b compensated by change in A
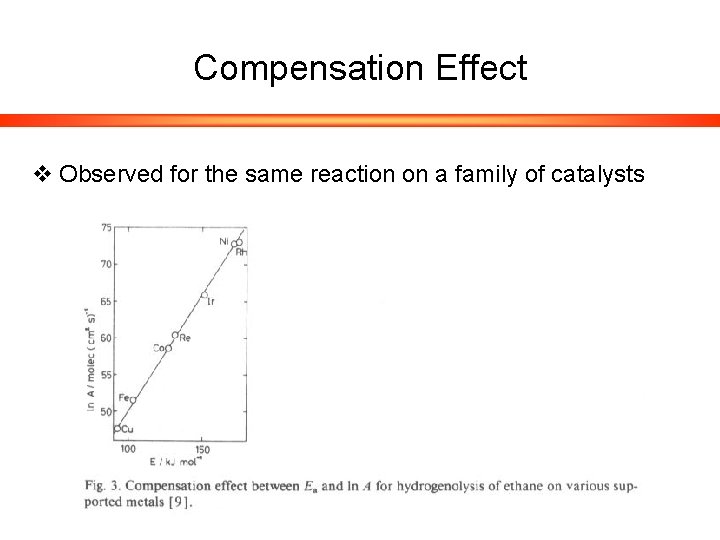
Compensation Effect v Observed for the same reaction on a family of catalysts
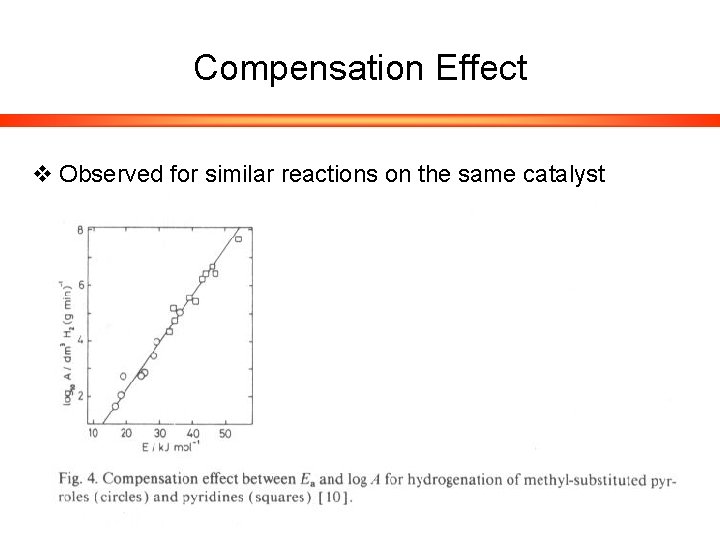
Compensation Effect v Observed for similar reactions on the same catalyst

Compensation Effect: Explanations v “Apparent” activation energy EA, app derived from measured rate and rate equation v With increasing temperature, the “true” reaction rate will increase v With increasing temperature the coverage decreases (exothermic adsorption), leading to a smaller measured rate v EA, app is a weighted sum of the EA, true and the enthalpy of adsorption

Literature v Gabor A. Somorjai, Introduction to Surface Chemistry and Catalysis, John Wiley, New York, 1994 v Bruce C. Gates, Catalytic Chemistry, John Wiley, New York, 1992 v G Ertl, H. Knözinger, J. Weitkamp, Handbook of Heterogeneous Catalysis, Wiley-VCH, Weinheim 1997 v G. Wedler, Physikalische Chemie, Verlag Chemie Weinheim v G. F. Froment, K. B. Bischoff, Chemical Reactor Analysis and Design, Wiley 1990 v Compensation effect: G. C. Bond, Catal. Today 1993, J. Catal. 1996
- Slides: 41