7 7 Fischer Projections Purpose of Fischer projections












![CO 2 H HO H [a] = -9. 5° CO 2 H [a] = CO 2 H HO H [a] = -9. 5° CO 2 H [a] =](https://slidetodoc.com/presentation_image_h2/bd07b7a33d2b594bfa1e589bf3782ccd/image-27.jpg)

![CO 2 H HO H [a] = -9. 5° CO 2 H [a] = CO 2 H HO H [a] = -9. 5° CO 2 H [a] =](https://slidetodoc.com/presentation_image_h2/bd07b7a33d2b594bfa1e589bf3782ccd/image-30.jpg)



- Slides: 51

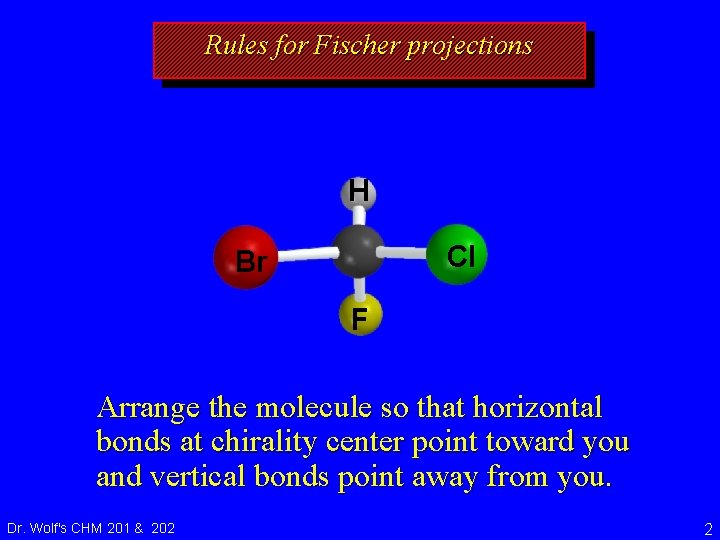
7. 7 Fischer Projections • Purpose of Fischer projections is to show configuration at chirality center without necessity of drawing wedges and dashes or using models. Dr. Wolf's CHM 201 & 202 1

Rules for Fischer projections H Cl Br F Arrange the molecule so that horizontal bonds at chirality center point toward you and vertical bonds point away from you. Dr. Wolf's CHM 201 & 202 2

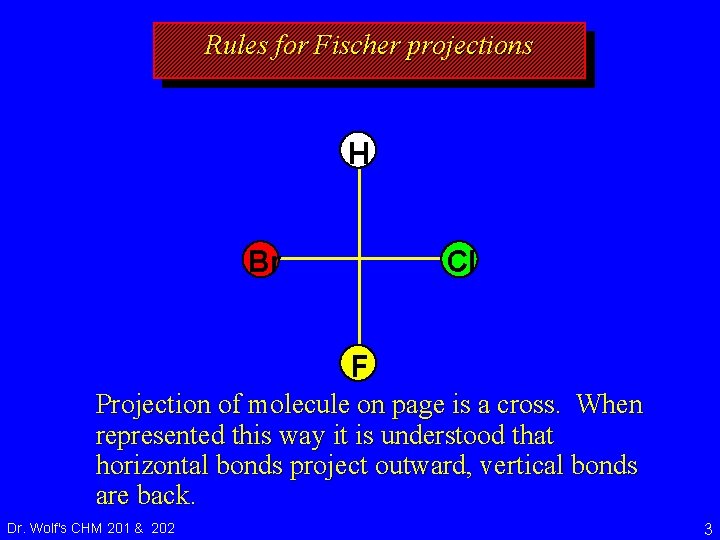
Rules for Fischer projections H Br Cl F Projection of molecule on page is a cross. When represented this way it is understood that horizontal bonds project outward, vertical bonds are back. Dr. Wolf's CHM 201 & 202 3

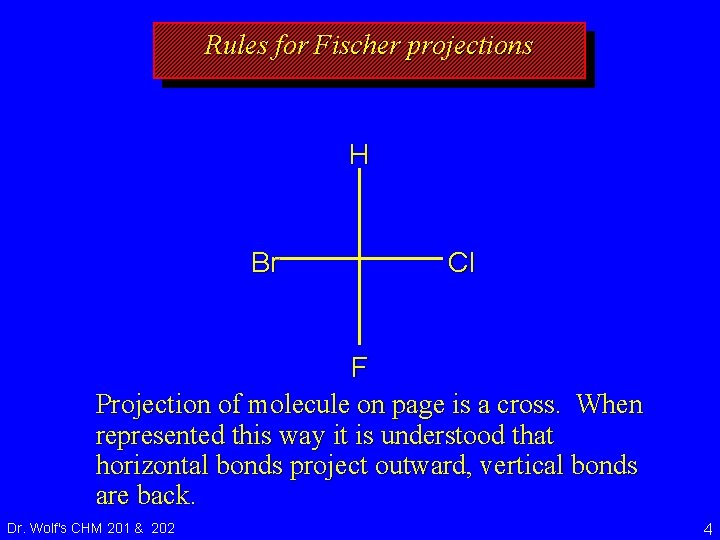
Rules for Fischer projections H Br Cl F Projection of molecule on page is a cross. When represented this way it is understood that horizontal bonds project outward, vertical bonds are back. Dr. Wolf's CHM 201 & 202 4

7. 8 Physical Properties of Enantiomers Dr. Wolf's CHM 201 & 202 5

Physical properties of enantiomers Same: melting point, boiling point, density, etc Different: properties that depend on shape of molecule (biological-physiological properties) can be different Dr. Wolf's CHM 201 & 202 6

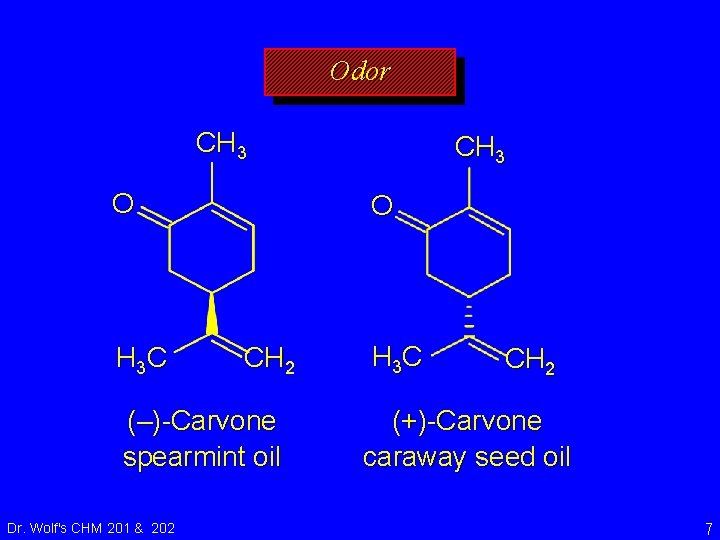
Odor CH 3 O H 3 C O CH 2 (–)-Carvone spearmint oil Dr. Wolf's CHM 201 & 202 CH 3 H 3 C CH 2 (+)-Carvone caraway seed oil 7

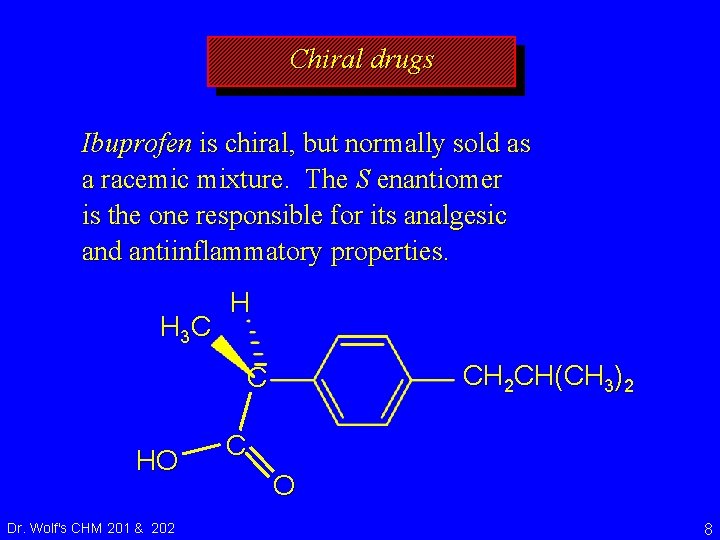
Chiral drugs Ibuprofen is chiral, but normally sold as a racemic mixture. The S enantiomer is the one responsible for its analgesic and antiinflammatory properties. H 3 C H CH 2 CH(CH 3)2 C HO Dr. Wolf's CHM 201 & 202 C O 8

7. 9 Reactions That Create A Chiral Center Dr. Wolf's CHM 201 & 202 9

Many reactions convert achiral reactants to chiral products. It is important to recognize, however, that if all of the components of the starting state (reactants, catalysts, solvents, etc. ) are achiral, any chiral product will be formed as a racemic mixture. This generalization can be more simply stated as "Optically inactive starting materials can't give optically active products. " (Remember: In order for a substance to be optically active, it must be chiral and one enantiomer must be present in greater amounts than the other. Dr. Wolf's CHM 201 & 202 10

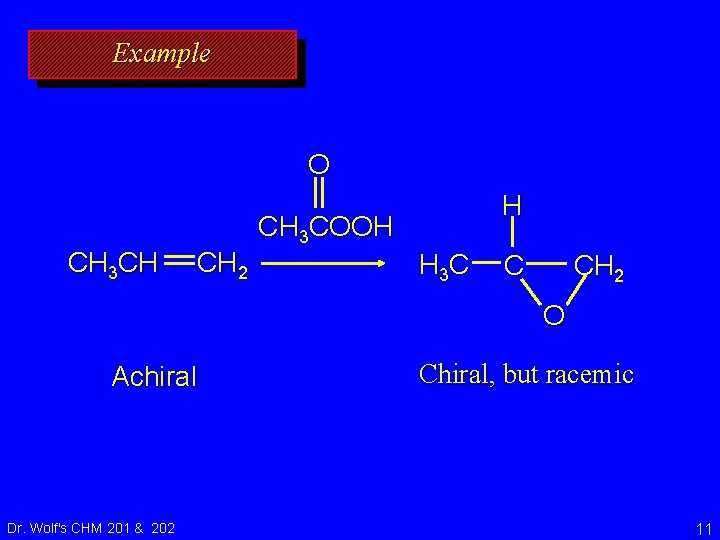
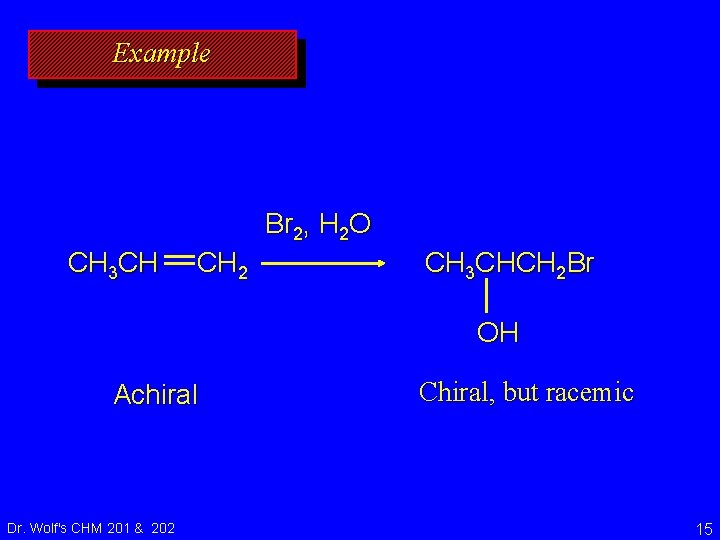
Example O CH 3 CH CH 2 H CH 3 COOH H 3 C C CH 2 O Achiral Dr. Wolf's CHM 201 & 202 Chiral, but racemic 11

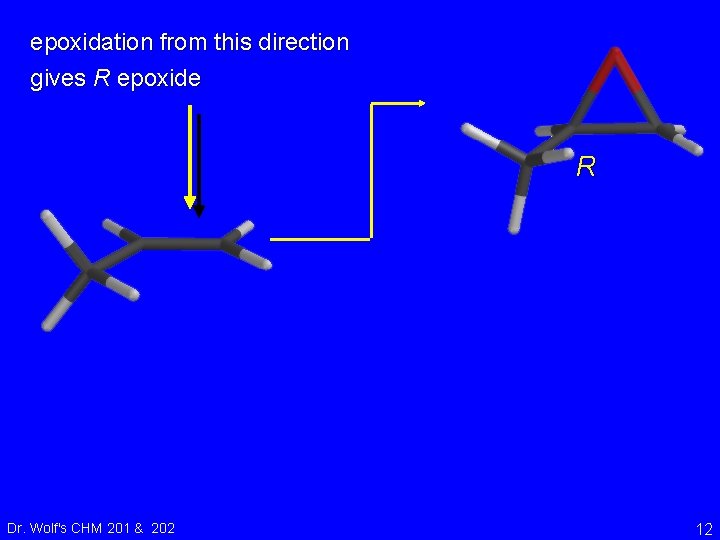
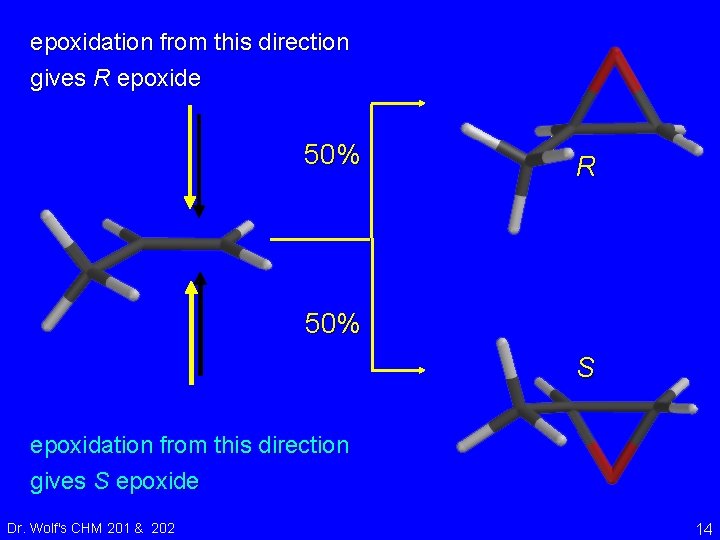
epoxidation from this direction gives R epoxide R Dr. Wolf's CHM 201 & 202 12

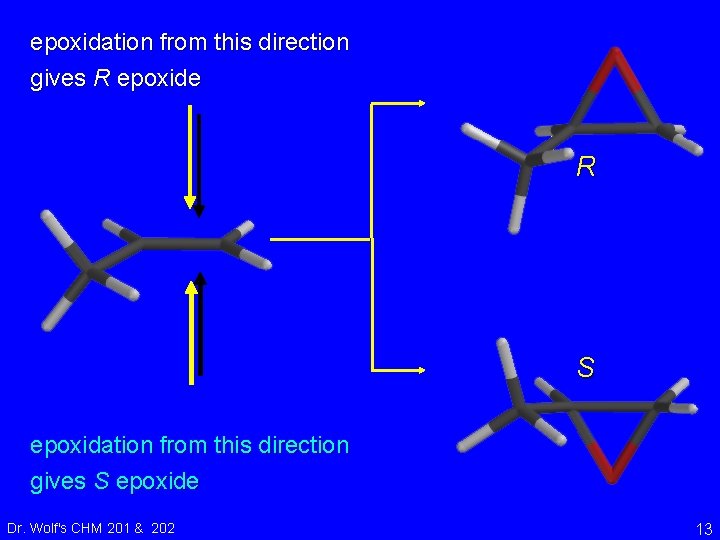
epoxidation from this direction gives R epoxide R S epoxidation from this direction gives S epoxide Dr. Wolf's CHM 201 & 202 13

epoxidation from this direction gives R epoxide 50% R 50% S epoxidation from this direction gives S epoxide Dr. Wolf's CHM 201 & 202 14

Example Br 2, H 2 O CH 3 CH CH 2 CH 3 CHCH 2 Br OH Achiral Dr. Wolf's CHM 201 & 202 Chiral, but racemic 15

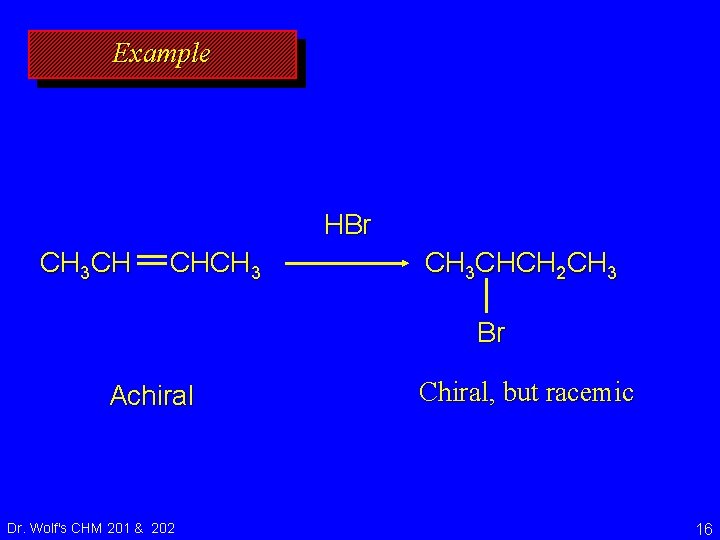
Example HBr CH 3 CH CHCH 3 CHCH 2 CH 3 Br Achiral Dr. Wolf's CHM 201 & 202 Chiral, but racemic 16

Many reactions convert chiral reactants to chiral products. However, if the reactant is racemic, the product will be racemic also. Remember: "Optically inactive starting materials can't give optically active products. " Dr. Wolf's CHM 201 & 202 17

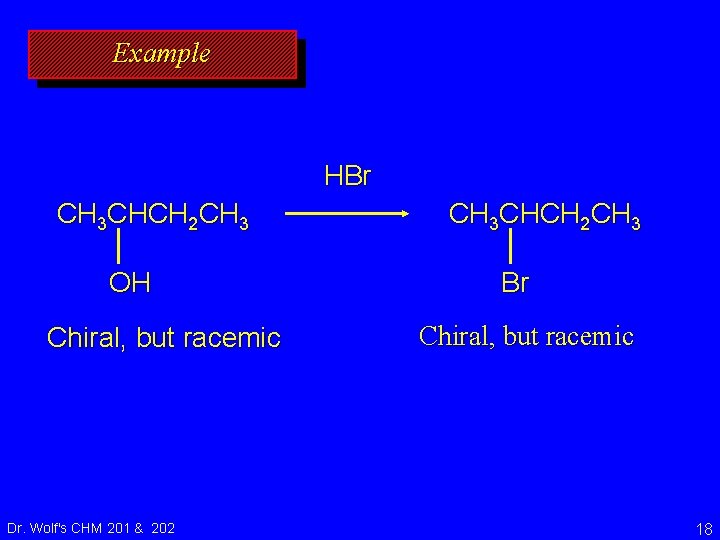
Example HBr CH 3 CHCH 2 CH 3 OH Chiral, but racemic Dr. Wolf's CHM 201 & 202 CH 3 CHCH 2 CH 3 Br Chiral, but racemic 18

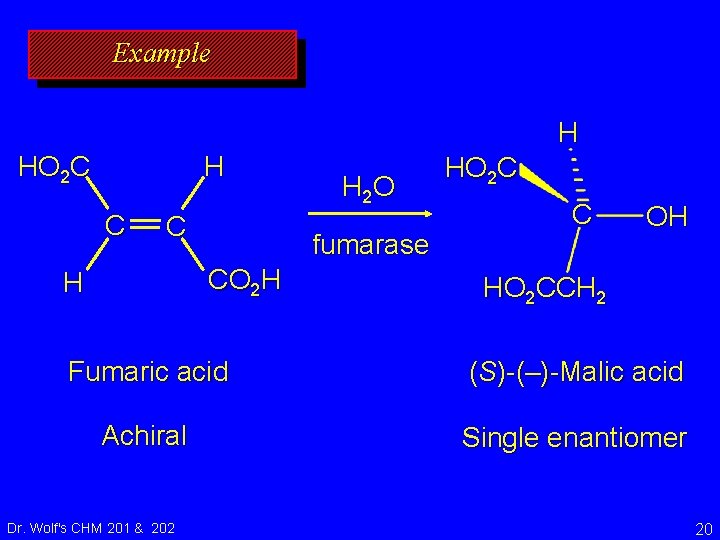
Many biochemical reactions convert an achiral reactant to a single enantiomer of a chiral product Reactions in living systems are catalyzed by enzymes, which are enantiomerically homogeneous. The enzyme (catalyst) is part of the reacting system, so such reactions don't violate the generalization that "Optically inactive starting materials can't give optically active products. " Dr. Wolf's CHM 201 & 202 19

Example H HO 2 C H C C fumarase CO 2 H H H 2 O HO 2 C C OH HO 2 CCH 2 Fumaric acid (S)-(–)-Malic acid Achiral Single enantiomer Dr. Wolf's CHM 201 & 202 20

7. 10 Chiral Molecules with Two Chirality Centers How many stereoisomers when a particular molecule contains two chiral centers? Dr. Wolf's CHM 201 & 202 21

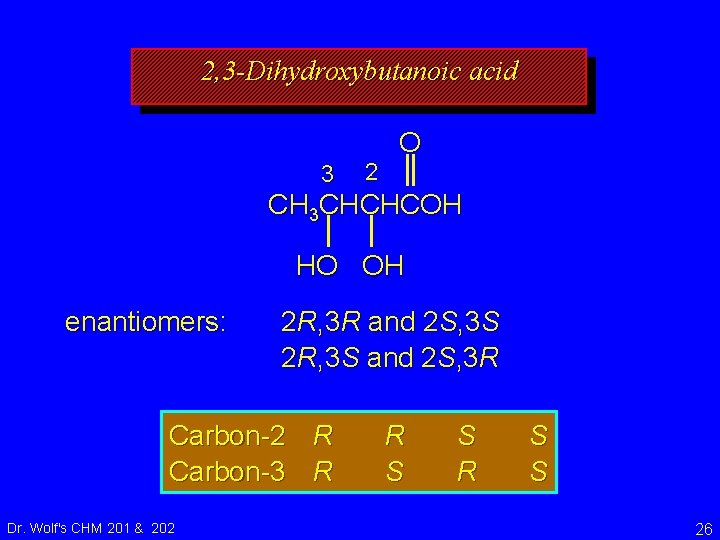
2, 3 -Dihydroxybutanoic acid 3 2 O CH 3 CHCHCOH HO OH What are all the possible R and S combinations of the two chirality centers in this molecule? Dr. Wolf's CHM 201 & 202 22

2, 3 -Dihydroxybutanoic acid 3 2 O CH 3 CHCHCOH HO OH What are all the possible R and S combinations of the two chirality centers in this molecule? Carbon-2 R Carbon-3 R Dr. Wolf's CHM 201 & 202 R S S 23

2, 3 -Dihydroxybutanoic acid 3 2 O CH 3 CHCHCOH HO OH 4 Combinations = 4 Stereoisomers Carbon-2 R Carbon-3 R Dr. Wolf's CHM 201 & 202 R S S 24

2, 3 -Dihydroxybutanoic acid 3 2 O CH 3 CHCHCOH HO OH 4 Combinations = 4 Stereoisomers What is the relationship between these stereoisomers? Carbon-2 R Carbon-3 R Dr. Wolf's CHM 201 & 202 R S S 25

2, 3 -Dihydroxybutanoic acid 3 2 O CH 3 CHCHCOH HO OH enantiomers: 2 R, 3 R and 2 S, 3 S 2 R, 3 S and 2 S, 3 R Carbon-2 R Carbon-3 R Dr. Wolf's CHM 201 & 202 R S S 26
![CO 2 H HO H a 9 5 CO 2 H a CO 2 H HO H [a] = -9. 5° CO 2 H [a] =](https://slidetodoc.com/presentation_image_h2/bd07b7a33d2b594bfa1e589bf3782ccd/image-27.jpg)
CO 2 H HO H [a] = -9. 5° CO 2 H [a] = +9. 5° R H OH R enantiomers H HO S H S CH 3 CO 2 H HO HO S R H H S CH 3 OH Dr. Wolf's CHM 201 & 202 enantiomers [a] = +17. 8° [a] = -17. 8° OH H H OH R CH 3 27

2, 3 -Dihydroxybutanoic acid O 3 2 CH 3 CHCHCOH HO OH but not all relationships are enantiomeric stereoisomers that are not enantiomers are: diastereomers……. similar but not identical chemical and physical properties Carbon-2 R Carbon-3 R Dr. Wolf's CHM 201 & 202 R S S 28

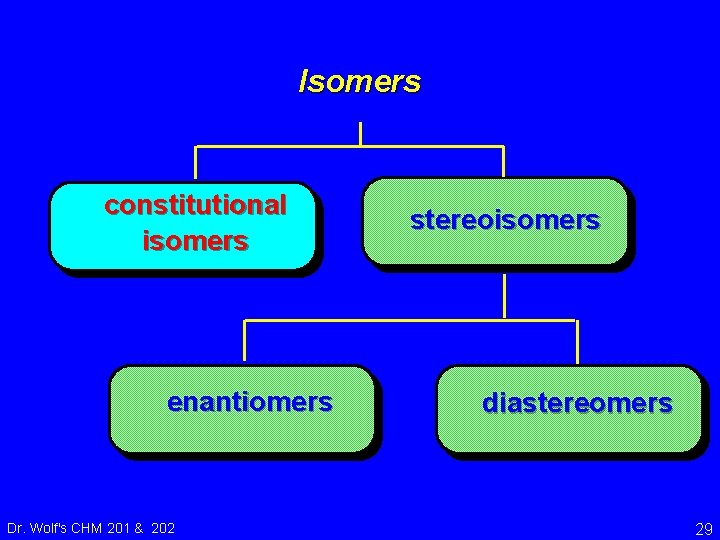
Isomers constitutional isomers enantiomers Dr. Wolf's CHM 201 & 202 stereoisomers diastereomers 29
![CO 2 H HO H a 9 5 CO 2 H a CO 2 H HO H [a] = -9. 5° CO 2 H [a] =](https://slidetodoc.com/presentation_image_h2/bd07b7a33d2b594bfa1e589bf3782ccd/image-30.jpg)
CO 2 H HO H [a] = -9. 5° CO 2 H [a] = +9. 5° R H OH R enantiomers H HO S H S CH 3 diastereomers CO 2 H HO HO S R H H S CH 3 OH Dr. Wolf's CHM 201 & 202 enantiomers [a] = +17. 8° [a] = -17. 8° OH H H OH R CH 3 30

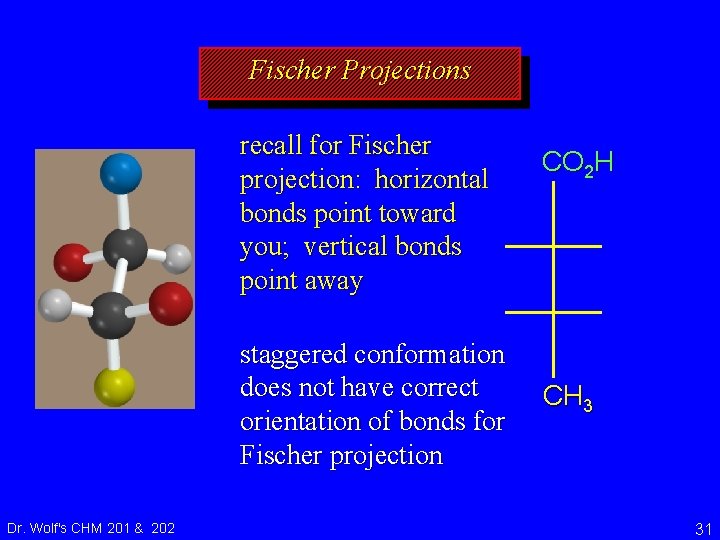
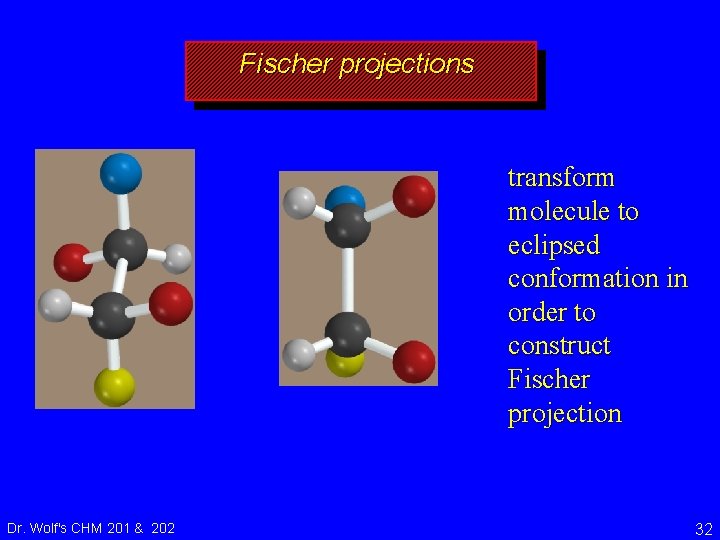
Fischer Projections recall for Fischer projection: horizontal bonds point toward you; vertical bonds point away staggered conformation does not have correct orientation of bonds for Fischer projection Dr. Wolf's CHM 201 & 202 CO 2 H CH 3 31

Fischer projections transform molecule to eclipsed conformation in order to construct Fischer projection Dr. Wolf's CHM 201 & 202 32

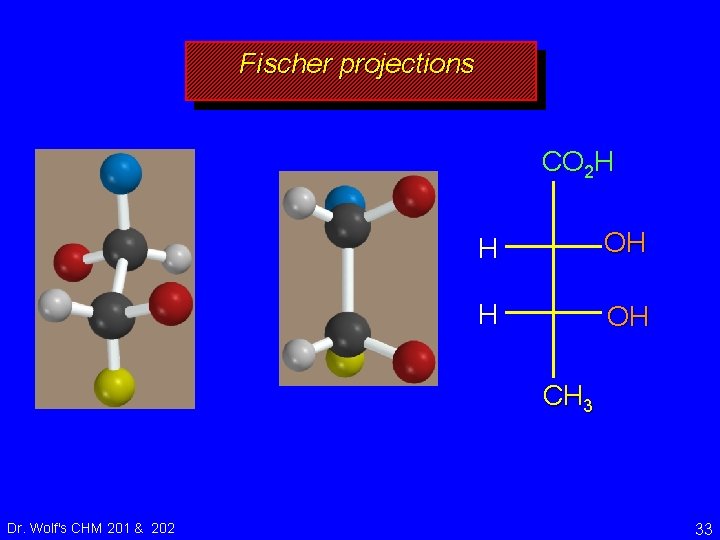
Fischer projections CO 2 H H OH CH 3 Dr. Wolf's CHM 201 & 202 33

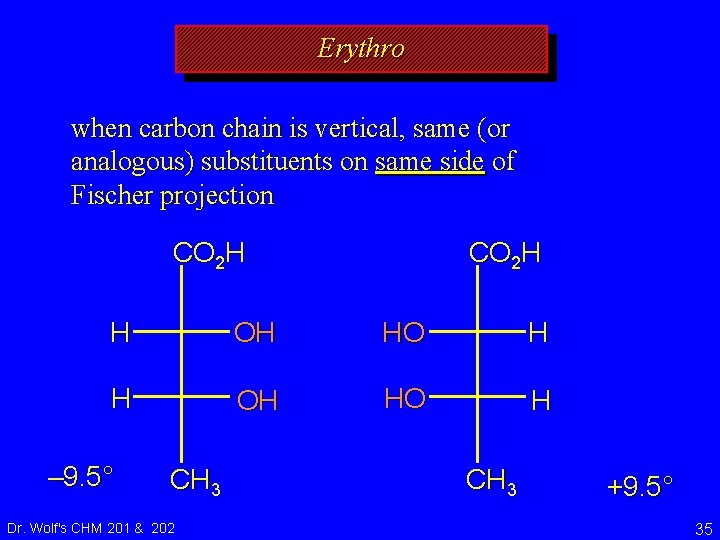
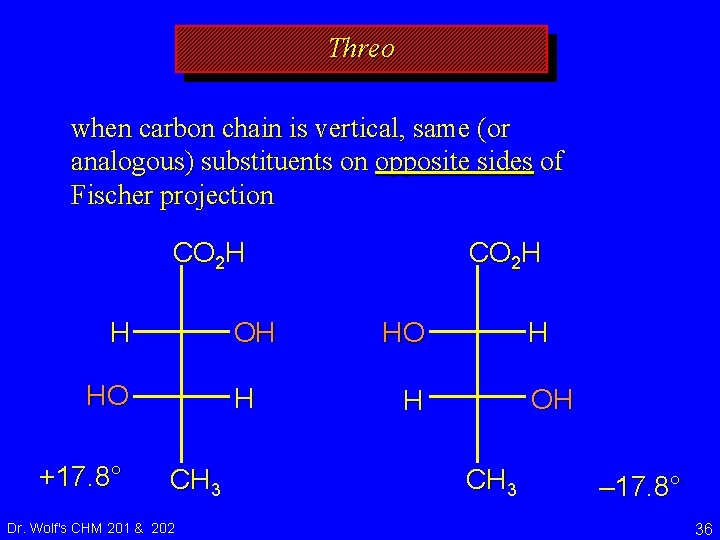
Erythro and Threo stereochemical prefixes used to specify relative configuration in molecules with two chirality centers easiest to apply using Fischer projections orientation: vertical carbon chain Dr. Wolf's CHM 201 & 202 34

Erythro when carbon chain is vertical, same (or analogous) substituents on same side of Fischer projection CO 2 H H OH HO H – 9. 5° CH 3 Dr. Wolf's CHM 201 & 202 CH 3 +9. 5° 35

Threo when carbon chain is vertical, same (or analogous) substituents on opposite sides of Fischer projection CO 2 H OH H HO +17. 8° H CH 3 Dr. Wolf's CHM 201 & 202 CO 2 H H HO OH H CH 3 – 17. 8° 36

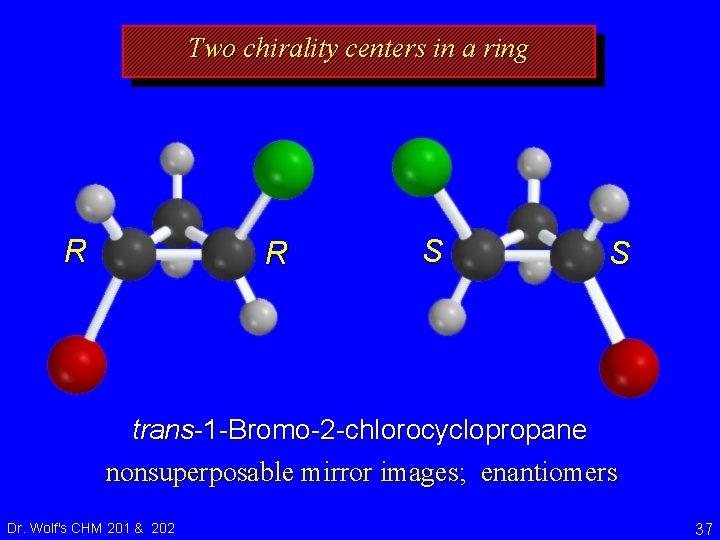
Two chirality centers in a ring R R S S trans-1 -Bromo-2 -chlorocyclopropane nonsuperposable mirror images; enantiomers Dr. Wolf's CHM 201 & 202 37

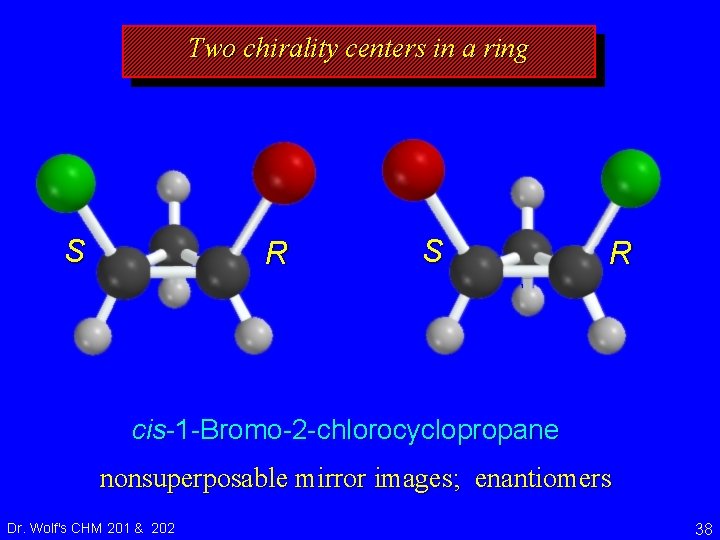
Two chirality centers in a ring S R cis-1 -Bromo-2 -chlorocyclopropane nonsuperposable mirror images; enantiomers Dr. Wolf's CHM 201 & 202 38

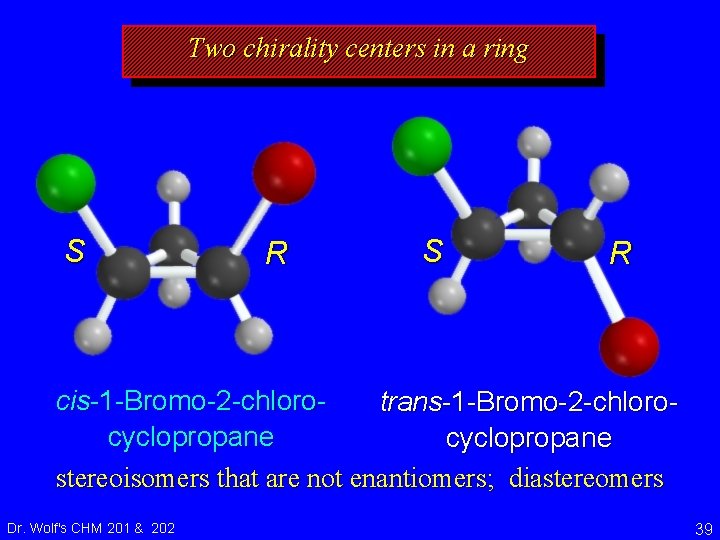
Two chirality centers in a ring S R cis-1 -Bromo-2 -chlorotrans-1 -Bromo-2 -chlorocyclopropane stereoisomers that are not enantiomers; diastereomers Dr. Wolf's CHM 201 & 202 39

7. 11 Achiral Molecules with Two Chirality Centers It is possible for a molecule to have chirality centers yet be achiral. Dr. Wolf's CHM 201 & 202 40

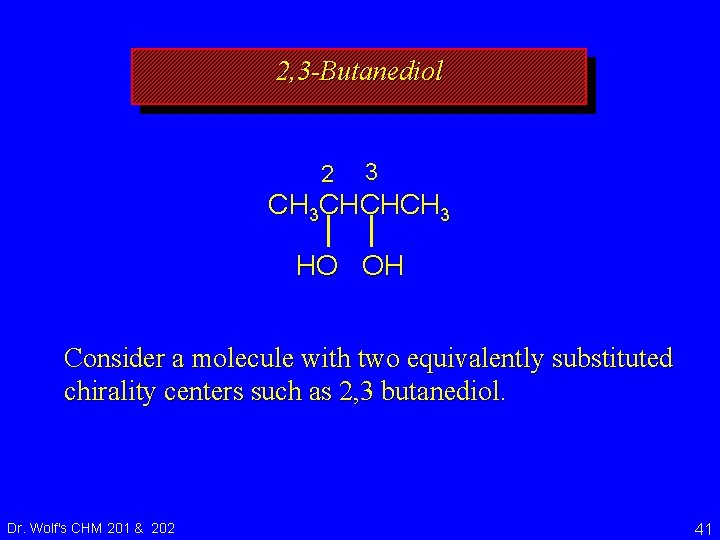
2, 3 -Butanediol 2 3 CH 3 CHCHCH 3 HO OH Consider a molecule with two equivalently substituted chirality centers such as 2, 3 butanediol. Dr. Wolf's CHM 201 & 202 41

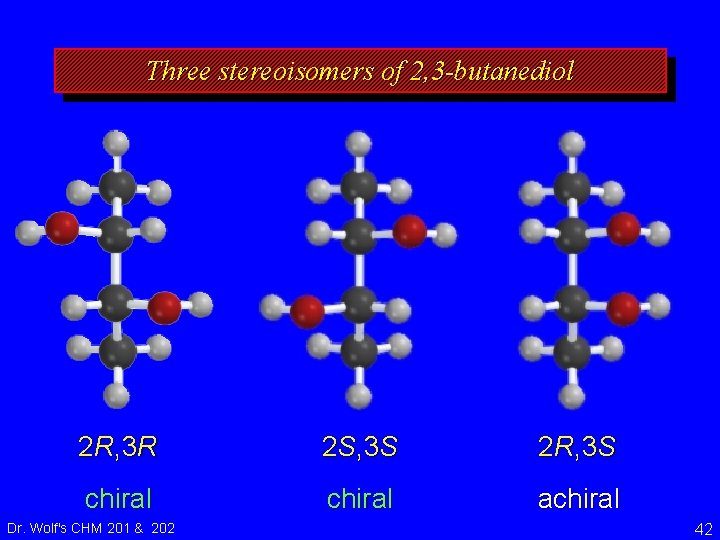
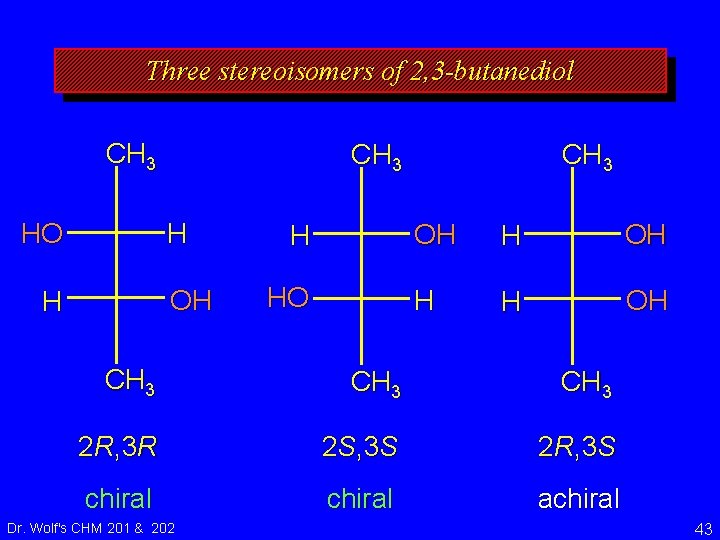
Three stereoisomers of 2, 3 -butanediol 2 R, 3 R 2 S, 3 S 2 R, 3 S chiral achiral Dr. Wolf's CHM 201 & 202 42

Three stereoisomers of 2, 3 -butanediol CH 3 H HO OH H H HO CH 3 OH H H OH CH 3 2 R, 3 R 2 S, 3 S 2 R, 3 S chiral achiral Dr. Wolf's CHM 201 & 202 43

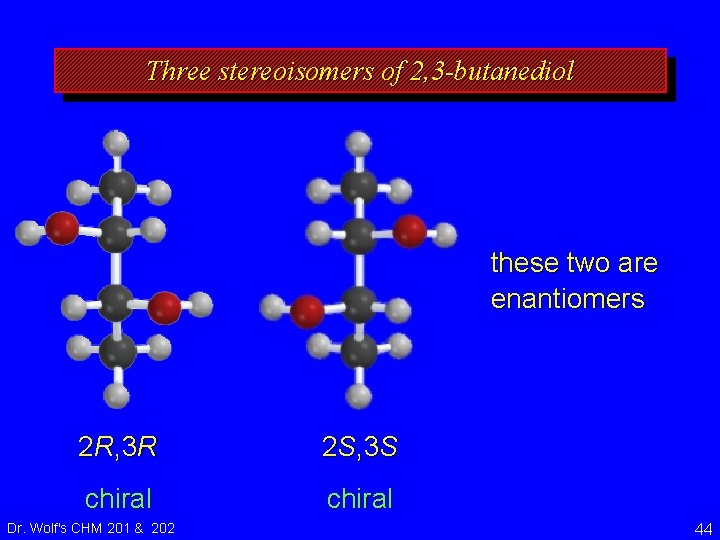
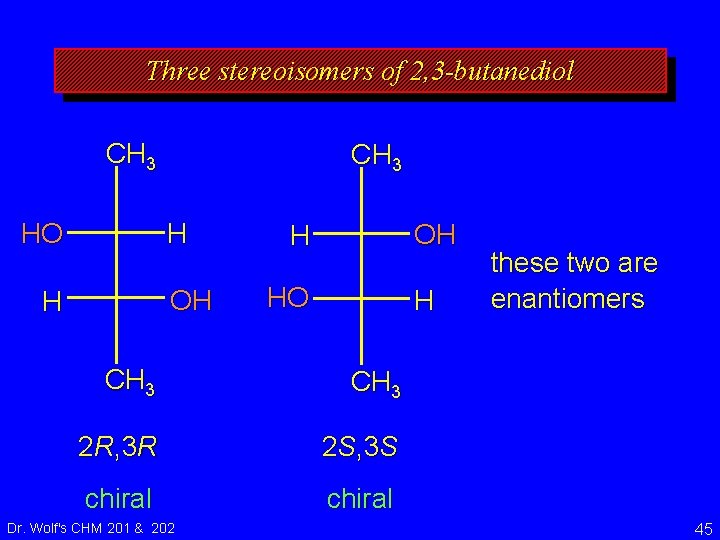
Three stereoisomers of 2, 3 -butanediol these two are enantiomers 2 R, 3 R 2 S, 3 S chiral Dr. Wolf's CHM 201 & 202 44

Three stereoisomers of 2, 3 -butanediol CH 3 H HO OH H HO H CH 3 2 R, 3 R 2 S, 3 S chiral Dr. Wolf's CHM 201 & 202 these two are enantiomers 45

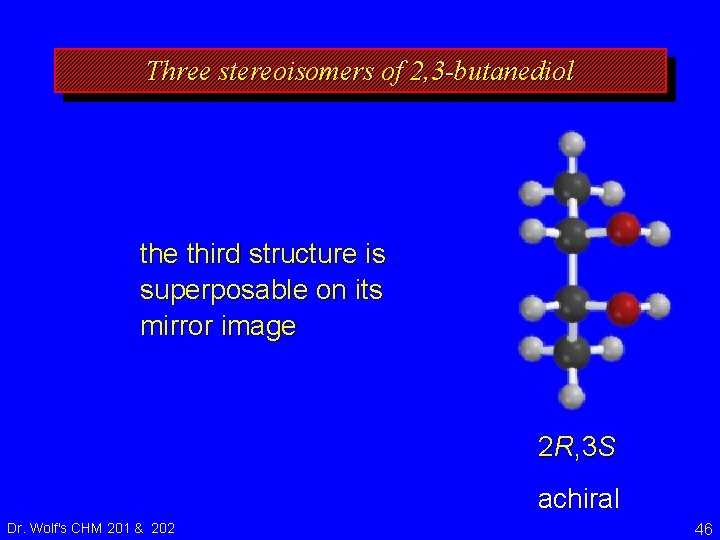
Three stereoisomers of 2, 3 -butanediol the third structure is superposable on its mirror image 2 R, 3 S achiral Dr. Wolf's CHM 201 & 202 46

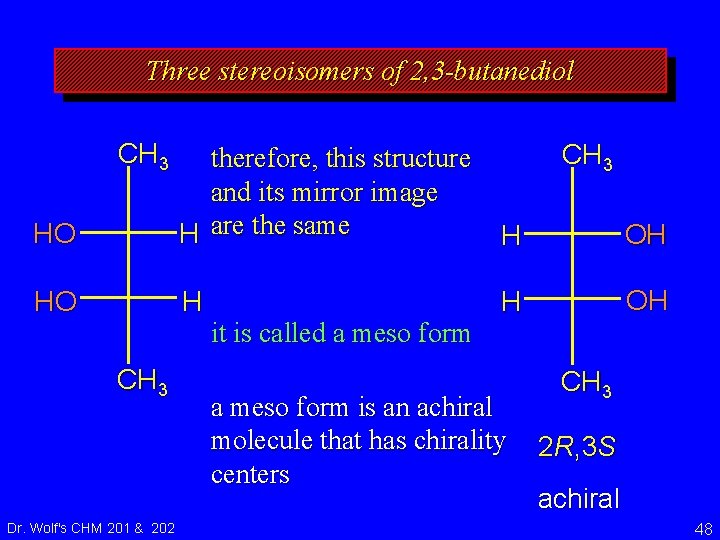
Three stereoisomers of 2, 3 -butanediol therefore, this structure and its mirror image are the same it is called a meso form is an achiral molecule that has chirality centers Dr. Wolf's CHM 201 & 202 2 R, 3 S achiral 47

Three stereoisomers of 2, 3 -butanediol CH 3 HO therefore, this structure and its mirror image H are the same H OH HO H H OH CH 3 Dr. Wolf's CHM 201 & 202 it is called a meso form is an achiral molecule that has chirality centers CH 3 2 R, 3 S achiral 48

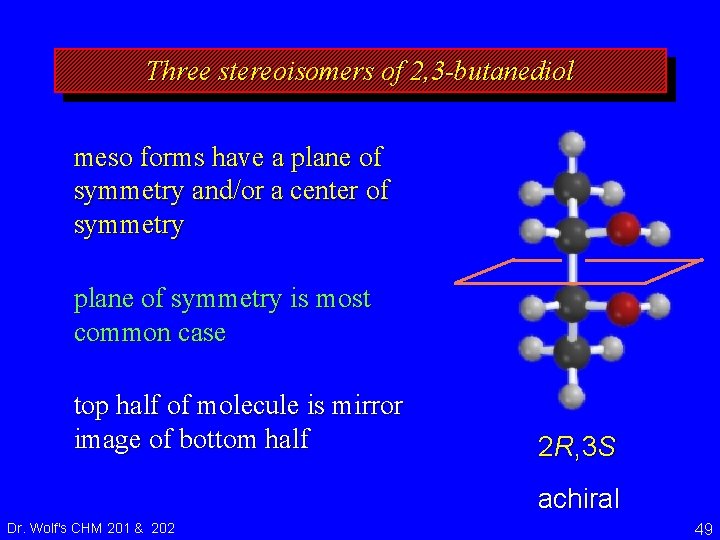
Three stereoisomers of 2, 3 -butanediol meso forms have a plane of symmetry and/or a center of symmetry plane of symmetry is most common case top half of molecule is mirror image of bottom half 2 R, 3 S achiral Dr. Wolf's CHM 201 & 202 49

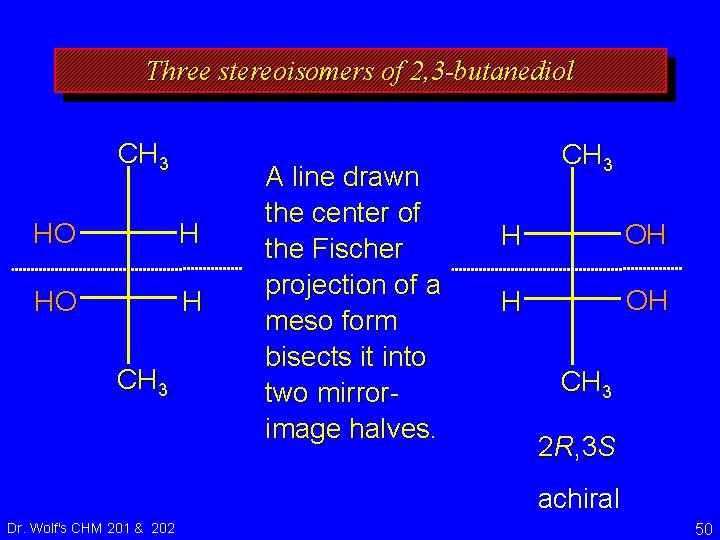
Three stereoisomers of 2, 3 -butanediol CH 3 HO H CH 3 A line drawn the center of the Fischer projection of a meso form bisects it into two mirrorimage halves. CH 3 H OH CH 3 2 R, 3 S achiral Dr. Wolf's CHM 201 & 202 50

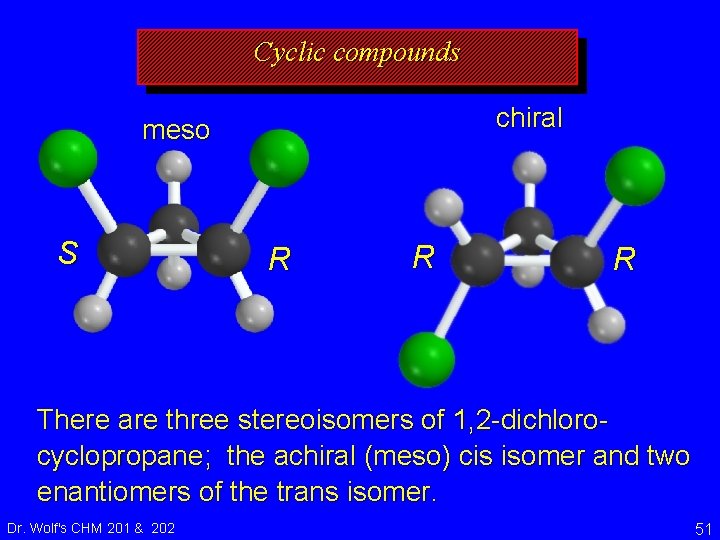
Cyclic compounds chiral meso S R R R There are three stereoisomers of 1, 2 -dichlorocyclopropane; the achiral (meso) cis isomer and two enantiomers of the trans isomer. Dr. Wolf's CHM 201 & 202 51