The Carbohydrates CH 2 On Emil Hermann Fischer
![The Carbohydrates [C(H 2 O)]n Emil Hermann Fischer (1852 -1919) The Carbohydrates [C(H 2 O)]n Emil Hermann Fischer (1852 -1919)](https://slidetodoc.com/presentation_image/0c08ccdbb75e2da0f04e07fc4dc5edb1/image-1.jpg)
The Carbohydrates [C(H 2 O)]n Emil Hermann Fischer (1852 -1919)
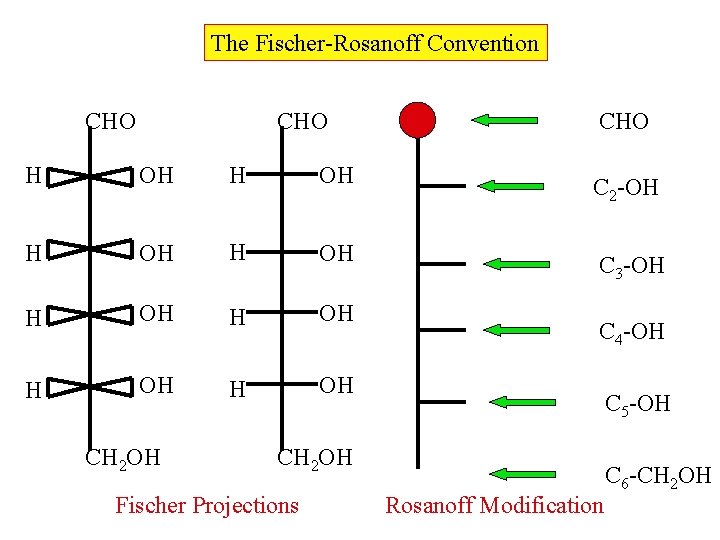
The Fischer-Rosanoff Convention CHO H OH H OH CH 2 OH CHO C 2 -OH C 3 -OH C 4 -OH C 5 -OH CH 2 OH Fischer Projections Rosanoff Modification C 6 -CH 2 OH
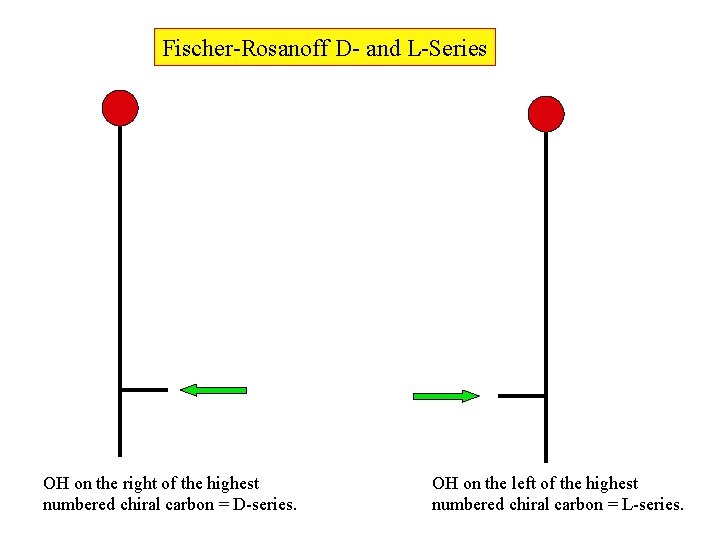
Fischer-Rosanoff D- and L-Series OH on the right of the highest numbered chiral carbon = D-series. OH on the left of the highest numbered chiral carbon = L-series.
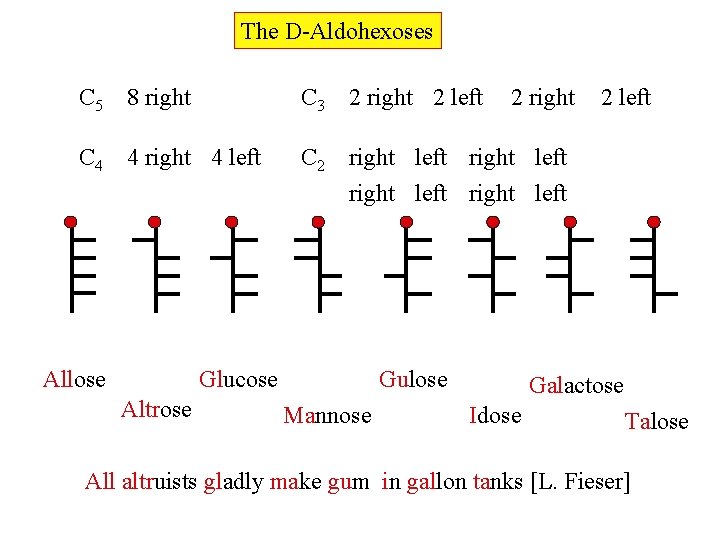
The D-Aldohexoses C 5 8 right C 3 2 right 2 left C 4 4 right 4 left C 2 right left Allose Glucose Altrose 2 right Gulose Mannose 2 left Galactose Idose Talose All altruists gladly make gum in gallon tanks [L. Fieser]
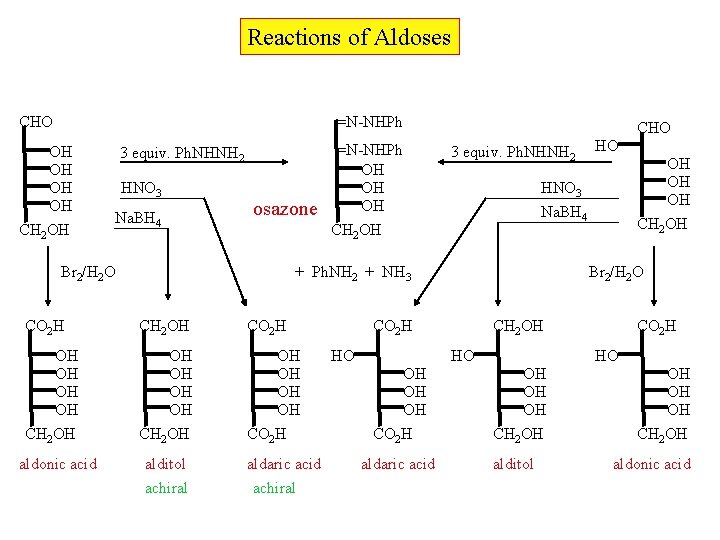
Reactions of Aldoses CHO =N-NHPh OH OH CH 2 OH 3 equiv. Ph. NHNH 2 HNO 3 Na. BH 4 osazone Br 2/H 2 O 3 equiv. Ph. NHNH 2 CH 2 OH CO 2 H OH OH CH 2 OH OH OH CO 2 H alditol achiral aldaric acid achiral Br 2/H 2 O CH 2 OH HO OH OH OH CO 2 H aldaric acid OH OH OH CH 2 OH Na. BH 4 CO 2 H HO HO HNO 3 + Ph. NH 2 + NH 3 CO 2 H aldonic acid =N-NHPh OH OH OH CH 2 OH CHO CO 2 H HO OH OH OH CH 2 OH alditol OH OH OH CH 2 OH aldonic acid
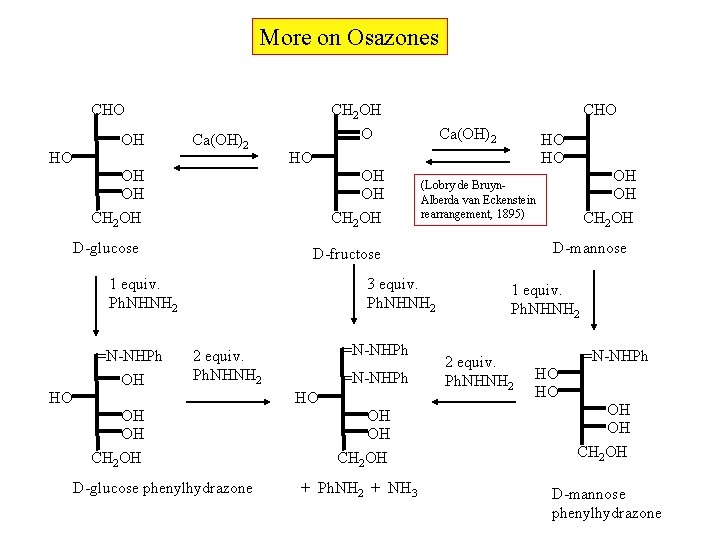
More on Osazones CH 2 OH O CHO OH HO Ca(OH)2 OH OH CH 2 OH HO HO 3 equiv. Ph. NHNH 2 =N-NHPh 2 equiv. Ph. NHNH 2 =N-NHPh HO HO OH OH CH 2 OH D-glucose phenylhydrazone OH OH CH 2 OH + Ph. NH 2 + NH 3 OH OH CH 2 OH (Lobry de Bruyn. Alberda van Eckenstein rearrangement, 1895) D-mannose D-fructose 1 equiv. Ph. NHNH 2 =N-NHPh OH Ca(OH)2 HO OH OH CH 2 OH D-glucose CHO 1 equiv. Ph. NHNH 2 2 equiv. Ph. NHNH 2 =N-NHPh HO HO OH OH CH 2 OH D-mannose phenylhydrazone
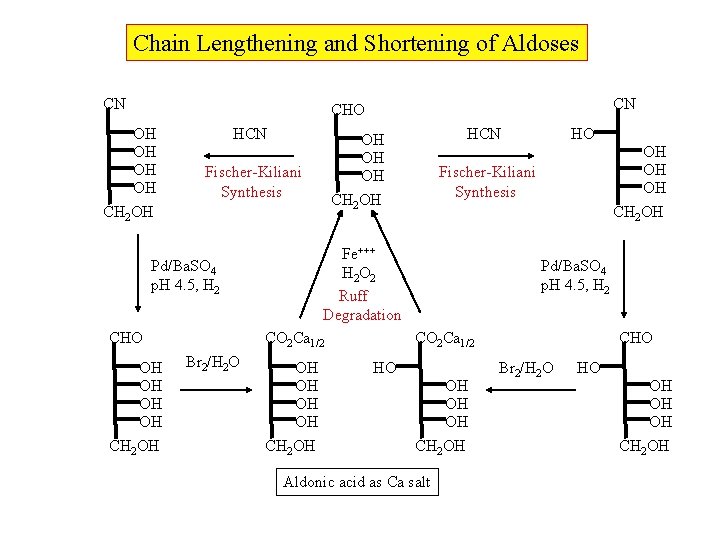
Chain Lengthening and Shortening of Aldoses CN CN CHO OH OH CH 2 OH HCN Fischer-Kiliani Synthesis OH OH CH 2 OH CO 2 Ca 1/2 Br 2/H 2 O OH OH CH 2 OH HO OH OH OH Fischer-Kiliani Synthesis CH 2 OH Fe+++ H 2 O 2 Ruff Degradation Pd/Ba. SO 4 p. H 4. 5, H 2 CHO HCN OH OH OH Pd/Ba. SO 4 p. H 4. 5, H 2 CO 2 Ca 1/2 HO OH OH OH CH 2 OH Aldonic acid as Ca salt CHO Br 2/H 2 O HO OH OH OH CH 2 OH
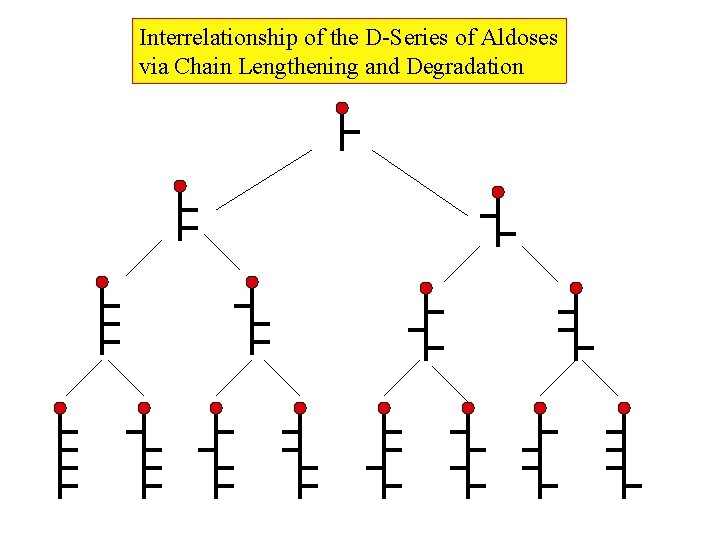
Interrelationship of the D-Series of Aldoses via Chain Lengthening and Degradation
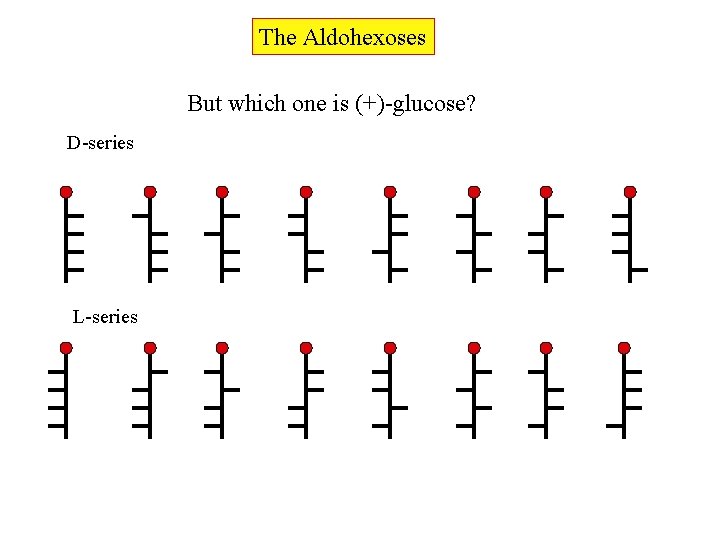
The Aldohexoses But which one is (+)-glucose? D-series L-series
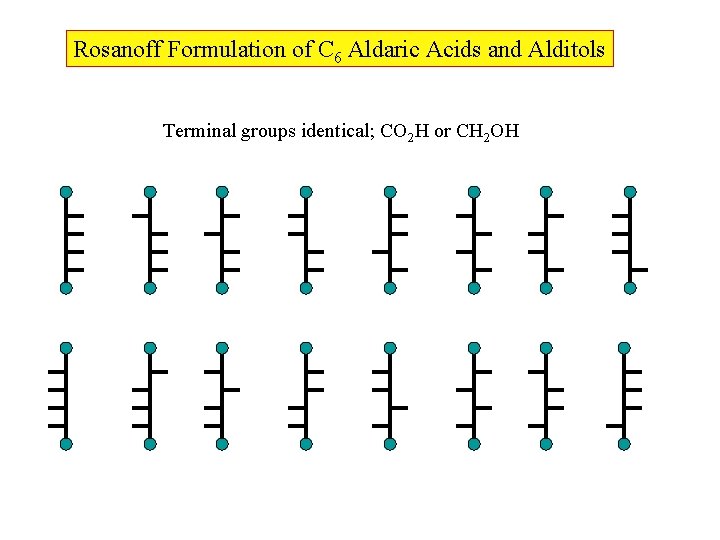
Rosanoff Formulation of C 6 Aldaric Acids and Alditols Terminal groups identical; CO 2 H or CH 2 OH
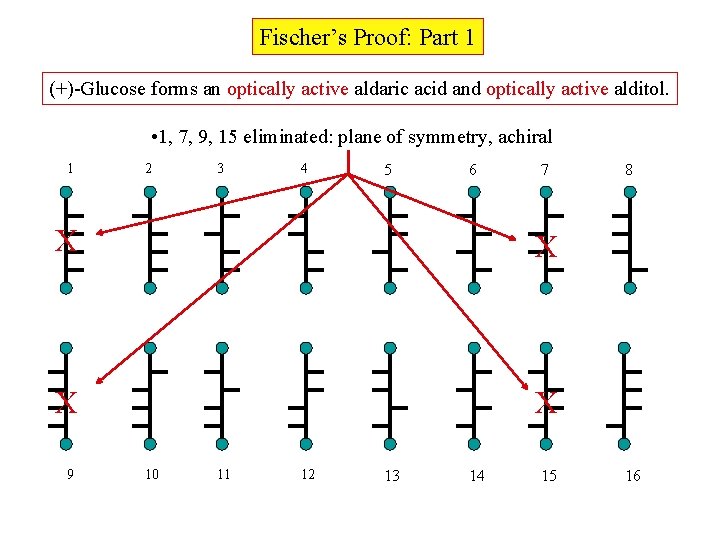
Fischer’s Proof: Part 1 (+)-Glucose forms an optically active aldaric acid and optically active alditol. • 1, 7, 9, 15 eliminated: plane of symmetry, achiral 1 2 3 4 5 6 7 X X 9 10 11 12 13 14 15 8 16
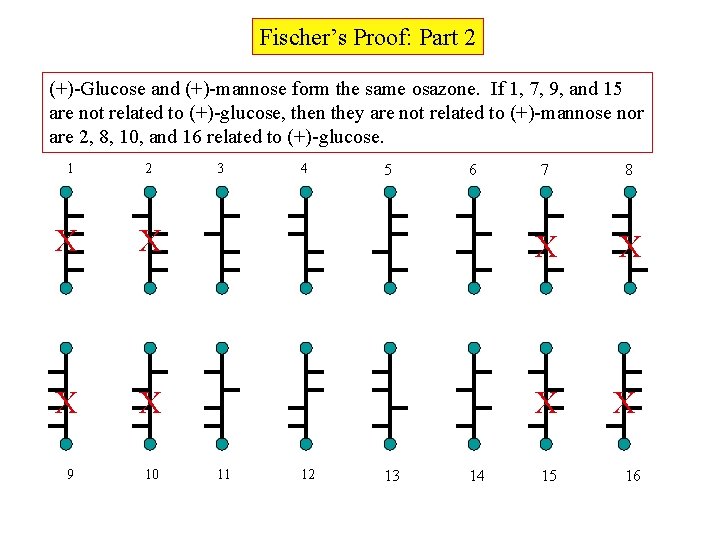
Fischer’s Proof: Part 2 (+)-Glucose and (+)-mannose form the same osazone. If 1, 7, 9, and 15 are not related to (+)-glucose, then they are not related to (+)-mannose nor are 2, 8, 10, and 16 related to (+)-glucose. 1 2 X 3 4 7 8 X X X X 9 10 11 12 5 13 6 14 15 16

Fischer’s Proof: Part 3 (+)-Arabinose affords (-)-glucose and (-)-mannose by Kiliani-Fischer synthesis. (+)-Arabinose must be of the opposite series (D/L)as (+)-glucose and have the same absolute configuration at C 3 -5 as (-)-glucose and (-)-mannose. What is the structure of arabinose?
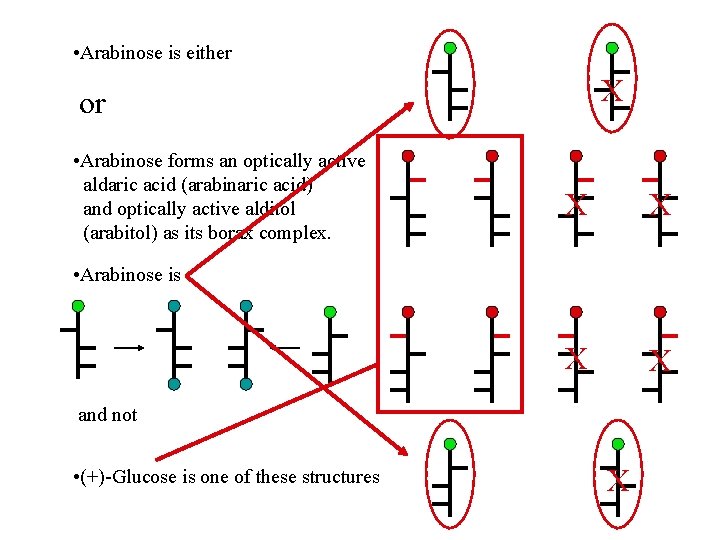
• Arabinose is either X or • Arabinose forms an optically active aldaric acid (arabinaric acid) and optically active alditol (arabitol) as its borax complex. X X • Arabinose is and not • (+)-Glucose is one of these structures X

Fischer’s Proof: Part 4 The pentose (+)-xylose affords optically inactive xylaric acid and optically inactive xylitol as its borax complex. • (+)-Xylose can only be one of the following: X X Fischer-Kiliani synthesis of (+)-xylose leads to two new hexoses, (+)-gulose and (+)-idose, both of which form optically active aldaric acids. • These enantiomers cannot be (+)-xylose because their Fischer-Kiliani hexoses (already eliminated) would lead to one optically active and one optically inactive aldaric acid. • (+)-Xylose must be one of the remaining two structures.
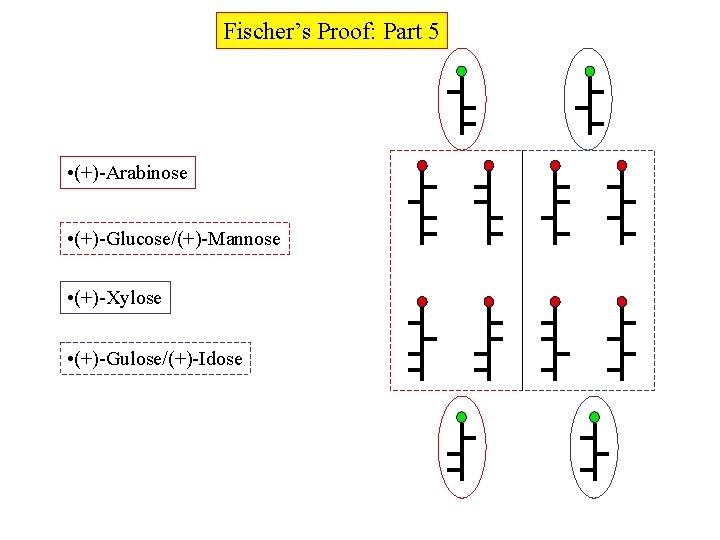
Fischer’s Proof: Part 5 • (+)-Arabinose • (+)-Glucose/(+)-Mannose • (+)-Xylose • (+)-Gulose/(+)-Idose
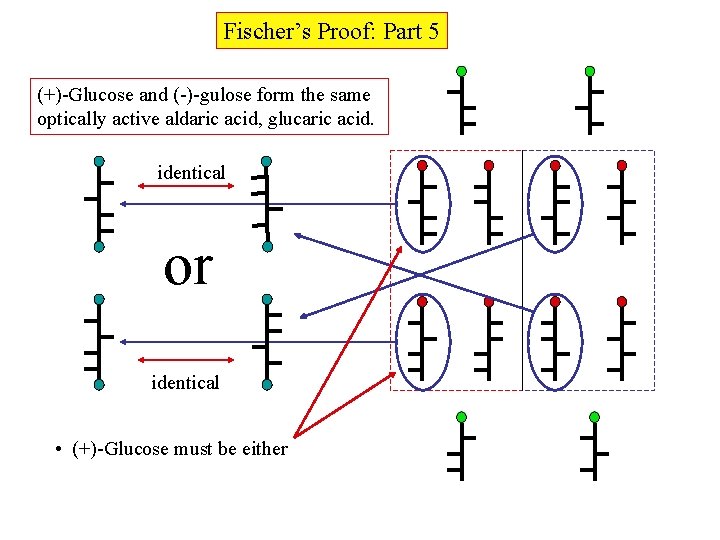
Fischer’s Proof: Part 5 (+)-Glucose and (-)-gulose form the same optically active aldaric acid, glucaric acid. identical or identical • (+)-Glucose must be either
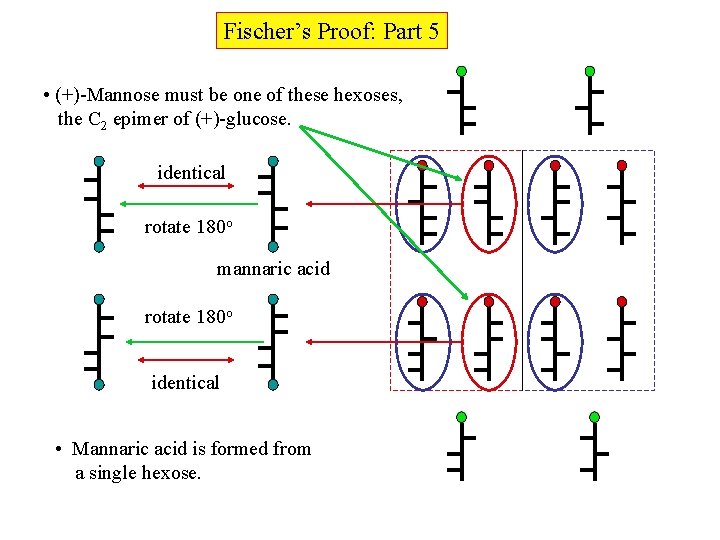
Fischer’s Proof: Part 5 • (+)-Mannose must be one of these hexoses, the C 2 epimer of (+)-glucose. identical rotate 180 o mannaric acid rotate 180 o identical • Mannaric acid is formed from a single hexose.
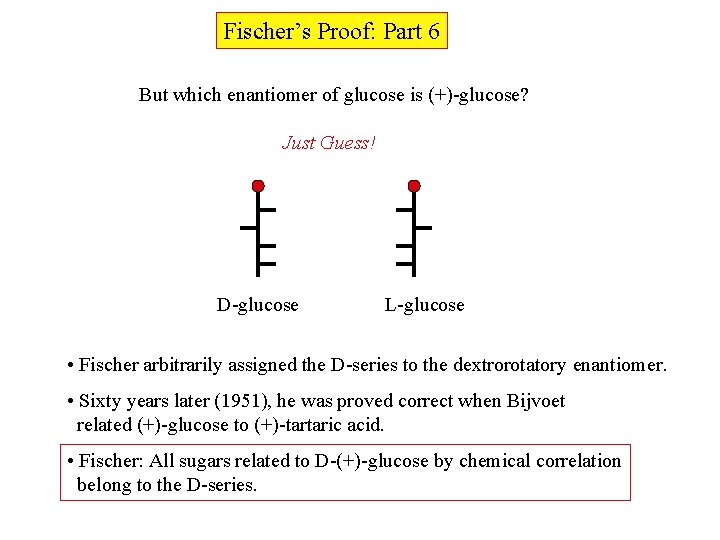
Fischer’s Proof: Part 6 But which enantiomer of glucose is (+)-glucose? Just Guess! D-glucose L-glucose • Fischer arbitrarily assigned the D-series to the dextrorotatory enantiomer. • Sixty years later (1951), he was proved correct when Bijvoet related (+)-glucose to (+)-tartaric acid. • Fischer: All sugars related to D-(+)-glucose by chemical correlation belong to the D-series.
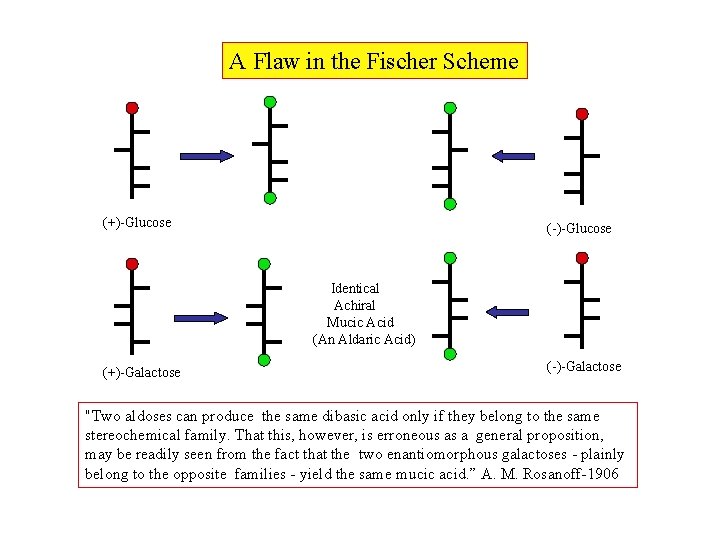
A Flaw in the Fischer Scheme (+)-Glucose (-)-Glucose Identical Achiral Mucic Acid (An Aldaric Acid) (+)-Galactose (-)-Galactose "Two aldoses can produce the same dibasic acid only if they belong to the same stereochemical family. That this, however, is erroneous as a general proposition, may be readily seen from the fact that the two enantiomorphous galactoses - plainly belong to the opposite families - yield the same mucic acid. ” A. M. Rosanoff-1906

Rosanoff’s Reorganization of the Carbohydrates • Arranged by successive Kiliani syntheses • The D- and L- assignments were Fischer’s based on chemical correlation with D-(+)-glucose, an unreliable scheme. 2 4 8 -series 16 mirror plane -series
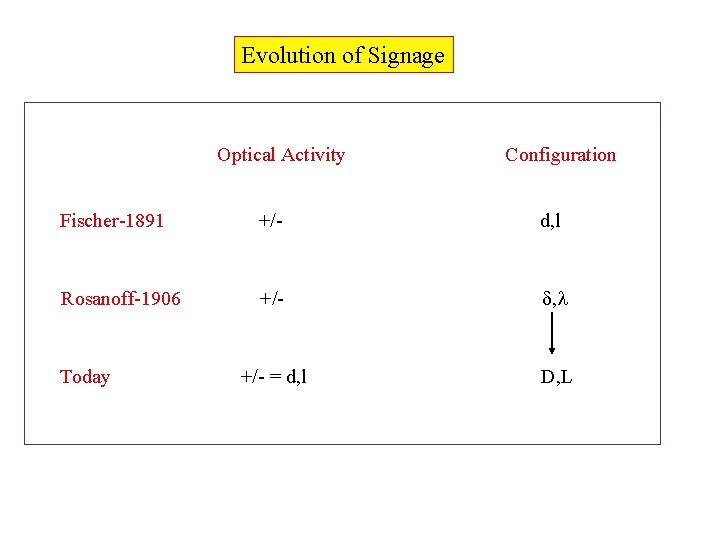
Evolution of Signage Optical Activity Configuration Fischer-1891 +/- d, l Rosanoff-1906 +/- = d, l D, L Today
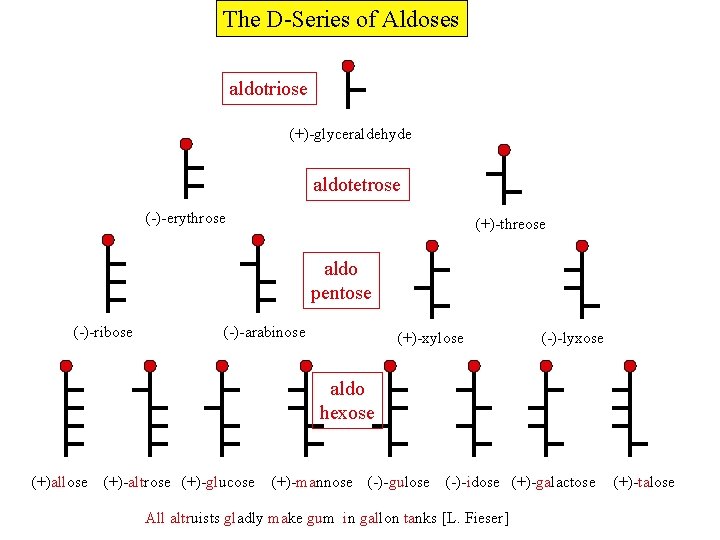
The D-Series of Aldoses aldotriose (+)-glyceraldehyde aldotetrose (-)-erythrose (+)-threose aldo pentose (-)-ribose (-)-arabinose (+)-xylose (-)-lyxose aldo hexose (+)allose (+)-altrose (+)-glucose (+)-mannose (-)-gulose (-)-idose (+)-galactose All altruists gladly make gum in gallon tanks [L. Fieser] (+)-talose
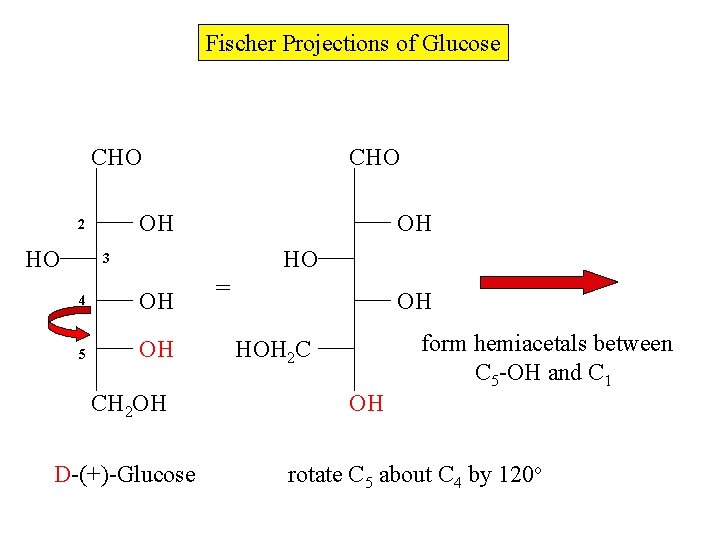
Fischer Projections of Glucose CHO OH 2 HO CHO OH HO 3 4 OH 5 OH CH 2 OH D-(+)-Glucose = OH form hemiacetals between C 5 -OH and C 1 HOH 2 C OH rotate C 5 about C 4 by 120 o
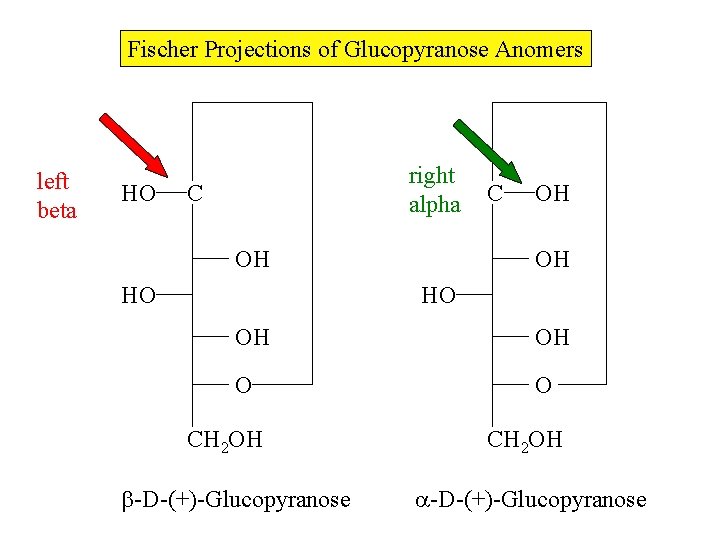
Fischer Projections of Glucopyranose Anomers left beta HO right alpha C OH HO C OH OH HO OH OH O O CH 2 OH -D-(+)-Glucopyranose
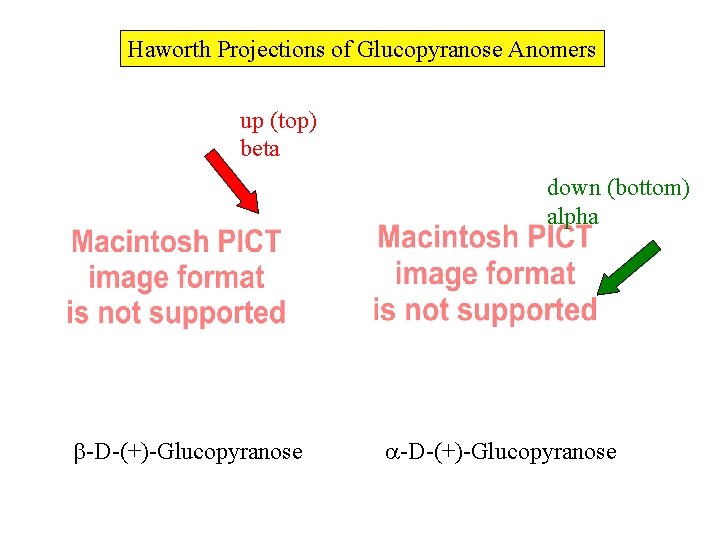
Haworth Projections of Glucopyranose Anomers up (top) beta down (bottom) alpha -D-(+)-Glucopyranose
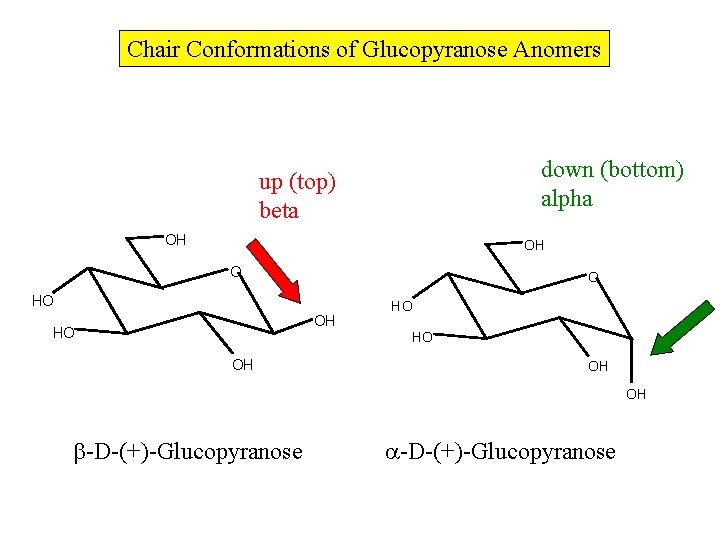
Chair Conformations of Glucopyranose Anomers down (bottom) alpha up (top) beta OH OH O O HO OH HO HO HO OH OH OH -D-(+)-Glucopyranose
How an Old Salt Remembers port starboard left right red light green light fewer letters more letters beta alpha up down top bottom
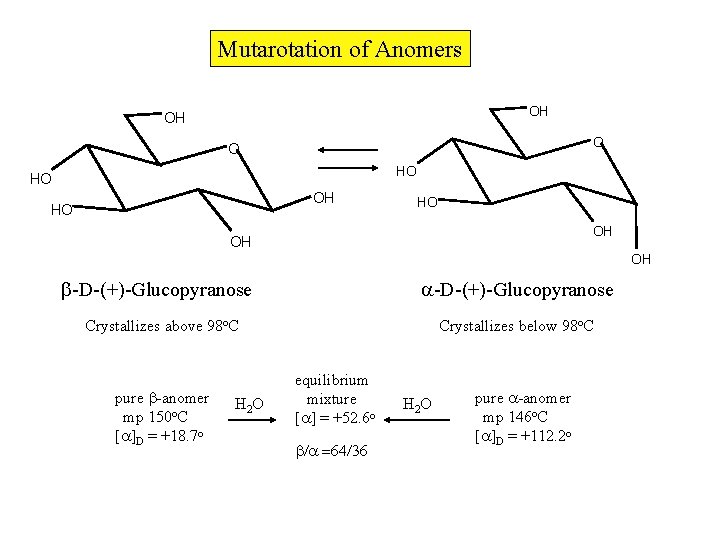
Mutarotation of Anomers OH OH O O HO HO OH OH OH -D-(+)-Glucopyranose Crystallizes above 98 o. C pure -anomer mp 150 o. C [ ]D = +18. 7 o H 2 O Crystallizes below 98 o. C equilibrium mixture [ ] = +52. 6 o H 2 O pure -anomer mp 146 o. C [ ]D = +112. 2 o
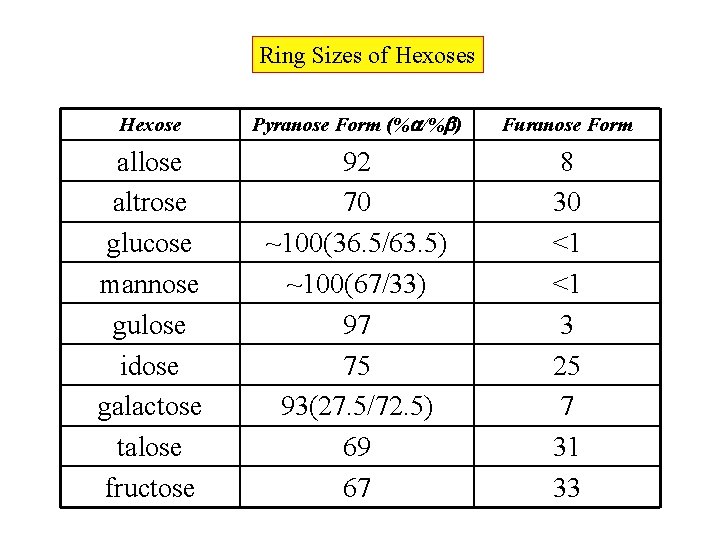
Ring Sizes of Hexoses Hexose Pyranose Form (% /% ) Furanose Form allose altrose glucose mannose gulose idose galactose talose fructose 92 70 ~100(36. 5/63. 5) ~100(67/33) 97 75 93(27. 5/72. 5) 69 67 8 30 <1 <1 3 25 7 31 33

Periodic Acid Cleavage of Carbohydrates as a Diagnostic Tool OH HIO 4 2 CH 2=O OH OH OH HIO 4 CHO HIO 4 CH 2=O + OH OH HO H 2 O OH HIO 4 OH Formaldehyde (CH 2 O) arises from a primary alcohols Formic acid (HCO 2 H) arises from a secondary alcohols HCO 2 H
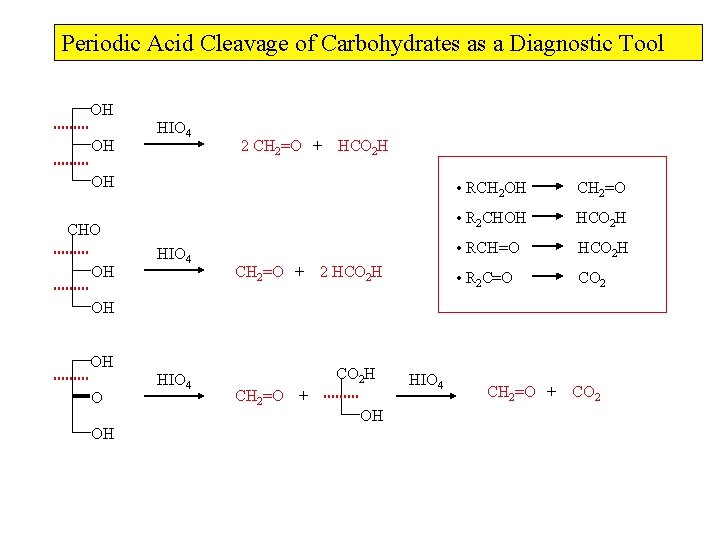
Periodic Acid Cleavage of Carbohydrates as a Diagnostic Tool OH OH HIO 4 2 CH 2=O + HCO 2 H OH CHO OH HIO 4 CH 2=O + 2 HCO 2 H • RCH 2 OH CH 2=O • R 2 CHOH HCO 2 H • RCH=O HCO 2 H • R 2 C=O CO 2 OH OH O OH HIO 4 CO 2 H CH 2=O + OH HIO 4 CH 2=O + CO 2
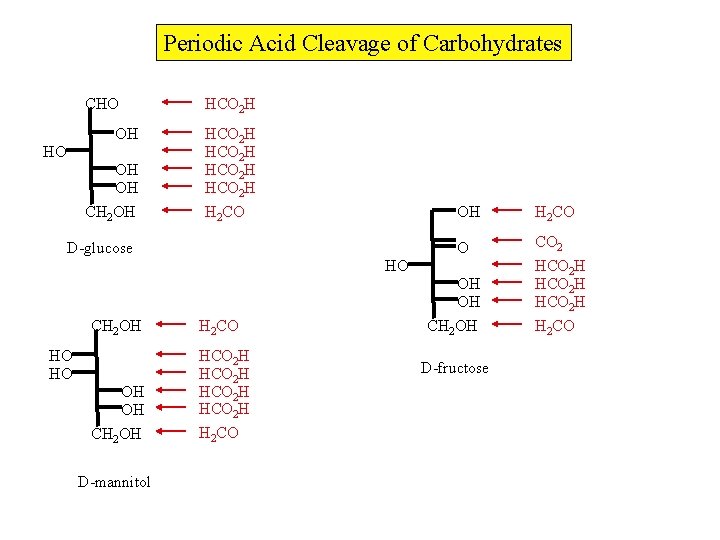
Periodic Acid Cleavage of Carbohydrates CHO OH OH CH 2 OH HCO 2 H HCO 2 H H 2 CO D-glucose OH H 2 CO O CO 2 HCO 2 H H 2 CO HO OH OH CH 2 OH H 2 CO OH OH CH 2 OH HCO 2 H H 2 CO HO HO D-mannitol CH 2 OH D-fructose
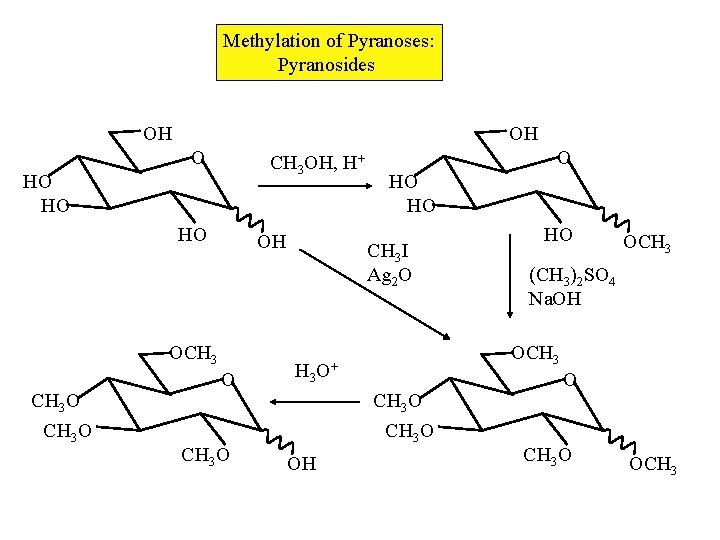
Methylation of Pyranoses: Pyranosides OH OH O CH 3 OH, H+ HO HO HO OH OCH 3 O O HO HO CH 3 I Ag 2 O O H 3 CH 3 O OH HO OCH 3 (CH 3)2 SO 4 Na. OH OCH 3 O+ CH 3 O OCH 3
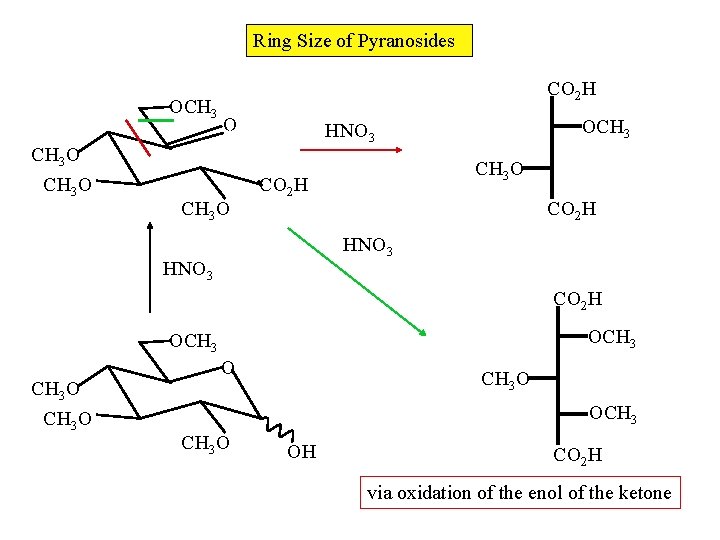
Ring Size of Pyranosides OCH 3 O CO 2 H O CH 3 O OCH 3 HNO 3 CH 3 O CO 2 H HNO 3 CO 2 H OCH 3 O O CH 3 O OCH 3 O OH CO 2 H via oxidation of the enol of the ketone
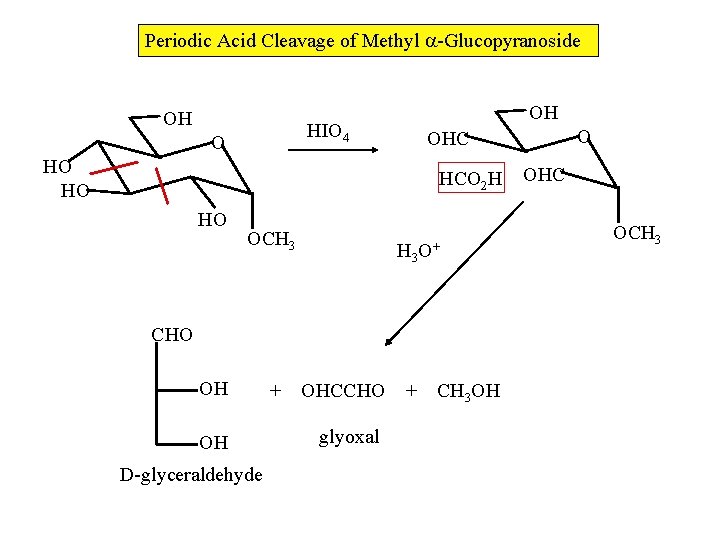
Periodic Acid Cleavage of Methyl -Glucopyranoside OH OH HIO 4 O HO HO HCO 2 H HO OCH 3 O+ CHO OH OH D-glyceraldehyde + OHCCHO + glyoxal O OHC CH 3 OH OHC OCH 3
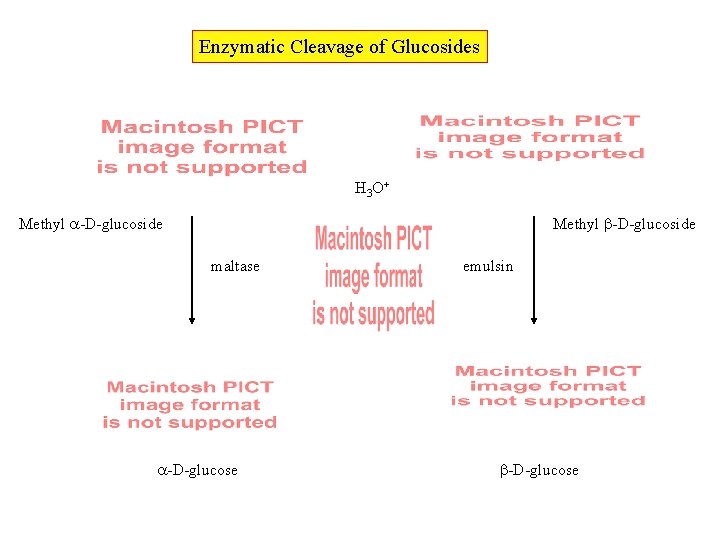
Enzymatic Cleavage of Glucosides H 3 O+ Methyl -D-glucoside maltase -D-glucose emulsin -D-glucose
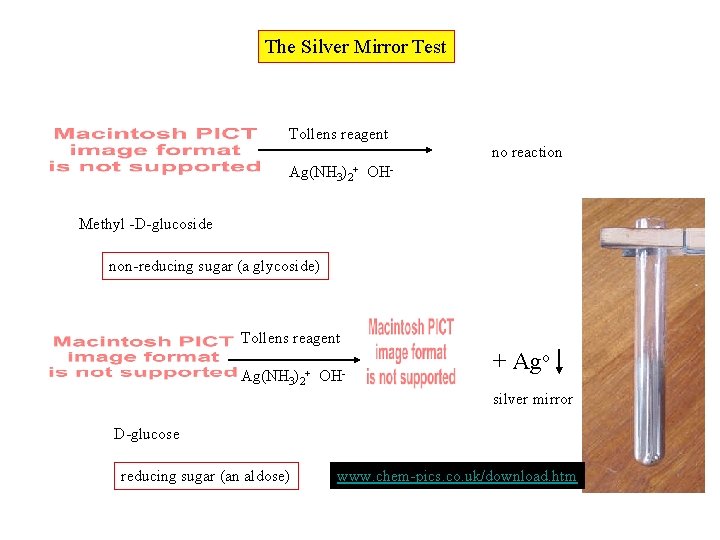
The Silver Mirror Test Tollens reagent no reaction Ag(NH 3)2+ OHMethyl -D-glucoside non-reducing sugar (a glycoside) Tollens reagent Ag(NH 3)2+ OH- + Ago silver mirror D-glucose reducing sugar (an aldose) www. chem-pics. co. uk/download. htm
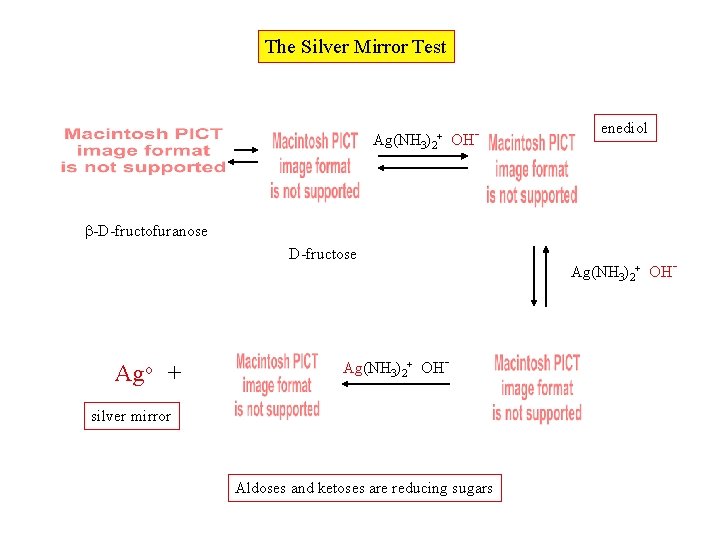
The Silver Mirror Test Ag(NH 3)2+ OH- enediol -D-fructofuranose D-fructose Ago + Ag(NH 3)2+ OH- silver mirror Aldoses and ketoses are reducing sugars Ag(NH 3)2+ OH-

Disaccharides and Polysaccharides

-D-Glucopyranosyl- -D-fructofuranoside or -D-Fructofuranosyl- -D-glucopyranoside OOCR O OH O HO HO acetal HO HO RCOO O HO O ketal RCOO OOCR OH HO Sucrose (non-reducing sugar) RCOO Olestra (R=n-Cn. H 2 n+1; n=6 -8)

Sucrose is Formed from Glucose and Fructose This discussion brings to mind a wonderful story told to me by Professor Harry Wasserman (Yale), who during the late 1940's was a graduate student of Professor R. B. Woodward at Harvard. Apparently Woodward had received a notice of a $1, 000 prize for the first person to accomplish a chemical synthesis of sucrose. He went into the laboratory and said to his students that all they had to do was connect two molecules of glucose together [. . . and lose a molecule of water] and they would have themselves $1, 000. One student, obviously not overwhelmed by Woodward's stature in the field even at such a young age, replied that if you did it that way, the prize would be $2, 000!
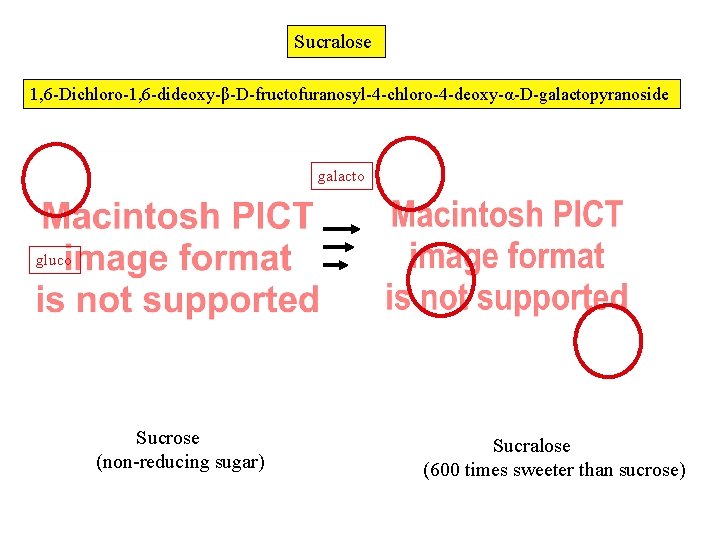
Sucralose 1, 6 -Dichloro-1, 6 -dideoxy-β-D-fructofuranosyl-4 -chloro-4 -deoxy-α-D-galactopyranoside galacto gluco Sucrose (non-reducing sugar) Sucralose (600 times sweeter than sucrose)
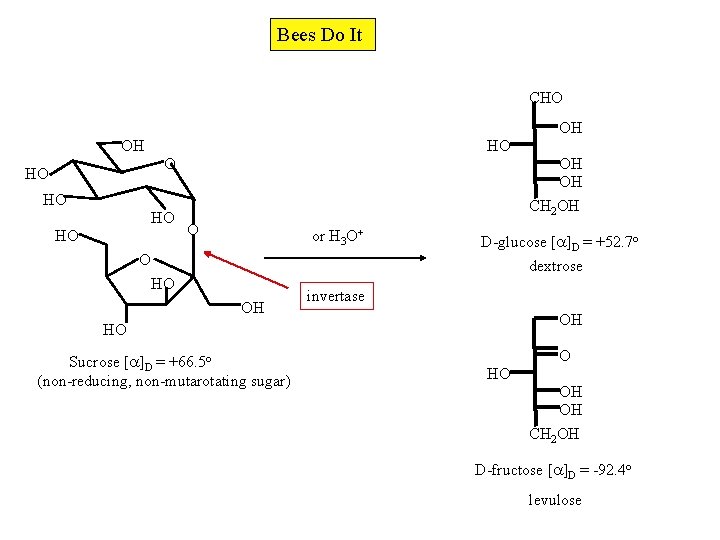
Bees Do It CHO OH OH HO HO O OH OH CH 2 OH HO HO HO O or H 3 O+ O D-glucose [ ]D = +52. 7 o dextrose HO OH invertase OH HO Sucrose [ ]D = +66. 5 o (non-reducing, non-mutarotating sugar) O HO OH OH CH 2 OH D-fructose [ ]D = -92. 4 o levulose
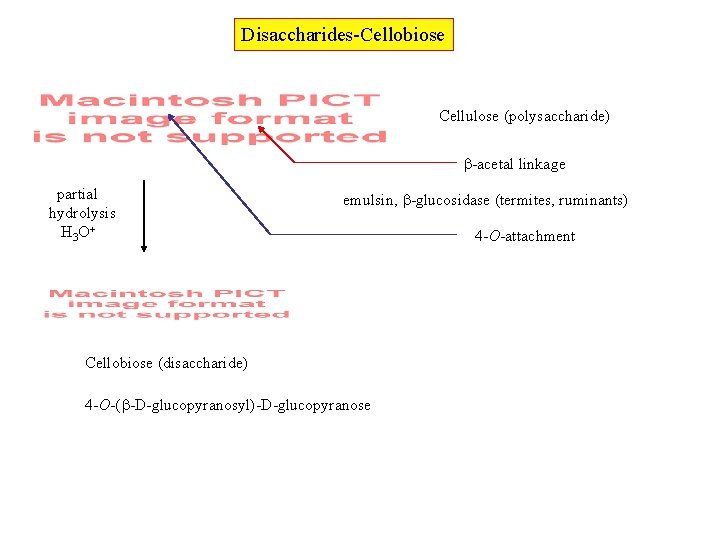
Disaccharides-Cellobiose Cellulose (polysaccharide) -acetal linkage partial hydrolysis H 3 O+ emulsin, -glucosidase (termites, ruminants) Cellobiose (disaccharide) 4 -O-( -D-glucopyranosyl)-D-glucopyranose 4 -O-attachment
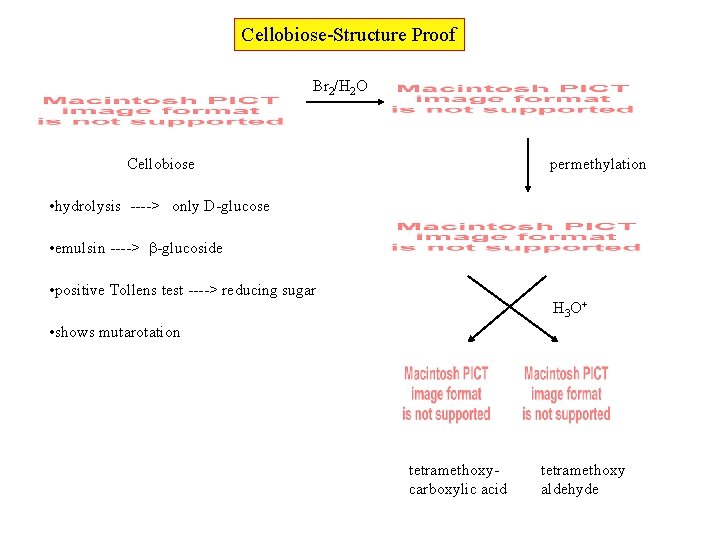
Cellobiose-Structure Proof Br 2/H 2 O Cellobiose permethylation • hydrolysis ----> only D-glucose • emulsin ----> -glucoside • positive Tollens test ----> reducing sugar H 3 O+ • shows mutarotation tetramethoxycarboxylic acid tetramethoxy aldehyde
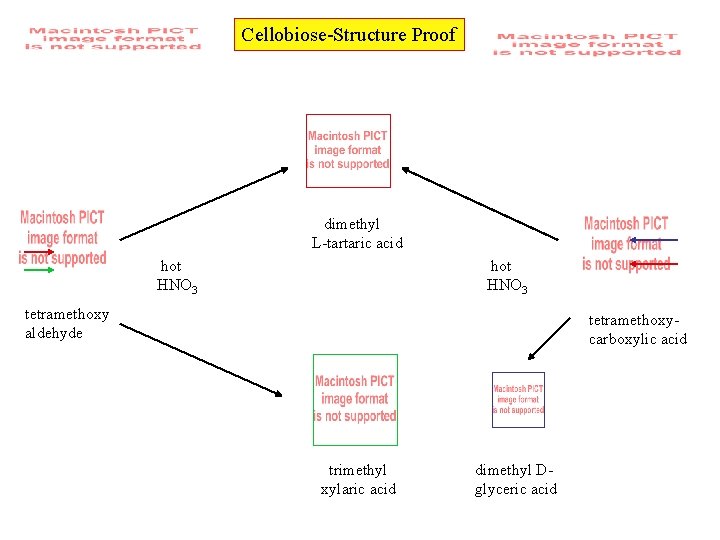
Cellobiose-Structure Proof dimethyl L-tartaric acid hot HNO 3 tetramethoxy aldehyde tetramethoxycarboxylic acid trimethyl xylaric acid dimethyl Dglyceric acid

Disaccharides: Maltose Starches: poly- -D-glucosides Malt (barley) maltase • hydrolysis ----> only D-glucose • maltase ----> -glucoside • positive Tollens test ----> reducing sugar • shows mutarotation • differs from cellobiose at the glycosidic anomeric center

Disaccharides: Lactose 4 -O-( -D-galactopyranosyl)-D-glucose ~5% of human and cow milk • hydrolysis ----> D-glucose and D-galactose • -galactosidase (lactase) ----> -galactoside • lactose intolerance • positive Tollens test ----> reducing sugar • shows mutarotation

Disaccharides: Amygdalin Laetrile (laevorotatory mandelonitrile), “vitamin 17” 1, 6 - -linkage cyanohydrin of benzaldehyde 1836 - Isolated from bitter almonds by Wohler. Demonstrated that emulsin produces glucose, benzaldehyde and prussic acid (HCN) Touted in some circles as a treatment for cancer.
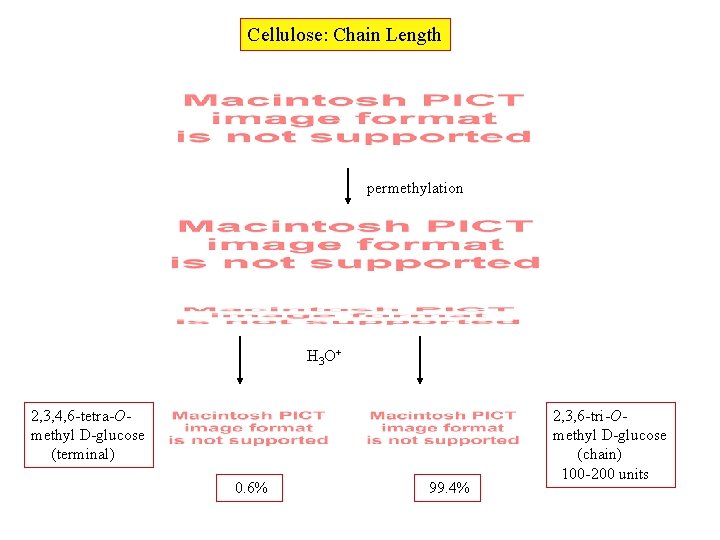
Cellulose: Chain Length permethylation H 3 O+ 2, 3, 4, 6 -tetra-Omethyl D-glucose (terminal) 0. 6% 99. 4% 2, 3, 6 -tri-Omethyl D-glucose (chain) 100 -200 units

Starches: Plant Polysaccharides Amylose : • ~20% water soluble starch; poly 4 -O-( -D-glucoside) • forms helical structure; blue complex with iodine

Starches: Plant Polysaccharides Amylopectin: • ~80% water insoluble starch branched poly-4 -O-( -D-glucoside) • permethylation/hydrolysis 90% 2, 3, 6 -tri-O-methyl-D-glucose ~5% 2, 3 -di-O-methyl-D-glucose ~5% 2, 3, 4, 6 -tetra-O-methyl-D-glucose chain junction Average 20 glucose units /chain terminus

And Finally, a True Story In March of 1986 I was in California visiting several universities. While at Stanford University, I stopped at the health center to have a swollen foot examined. The young resident was very attentive. To assess his qualifications, I asked him where he had attended college. “M. I. T. , ” he responded. “So you must have had Professor Kemp for organic chemistry, ” I countered. “Yes, I did, ” he said. Then I asked, “What D-aldohexose forms the same osazone as glucose? ” Like Diogenes the Cynic in search of an honest man (person), I have posed this question to many a practitioner of the medical and dental professions. Neither Diogenes nor I have fulfilled our quests. However, the responses to my query were often amusing. Response: My thought: “I really enjoyed organic chemistry!” Really? “Organic, don’t remind me!” An honest man? “I know the mechanism of the aldol condensation. ” Wrong chapter! “I know what a Grignard reagent is. ” Wrong test! “Wait! Give me some time. ” “It’s only an hour exam. ”

The Moral of the Story Somewhere, …sometime…someone might ask you this question. What D-aldohexose forms the same osazone as D-glucose? Your answer will be. . . D-Mannose!

The End
- Slides: 56