Motifs Part of MATERIALS SCIENCE A Learners Guide










- Slides: 16

Motifs Part of MATERIALS SCIENCE & A Learner’s Guide ENGINEERING AN INTRODUCTORY E-BOOK Anandh Subramaniam & Kantesh Balani Materials Science and Engineering (MSE) Indian Institute of Technology, Kanpur- 208016 Email: anandh@iitk. ac. in, URL: home. iitk. ac. in/~anandh http: //home. iitk. ac. in/~anandh/E-book. htm

Motifs q Basis is a synonym for Motif. q Any entity which is associated with each lattice point is a motif. q This entity could be a geometrical object or a physical property (or a combination). q This could be a shape like a pentagon (in 2 D), cube (in 3 D) or something more complicated. q Typically in atomic crystals an atom (or group of atoms) ions (or groups of ions) molecules (or group of Molecules) associated with each lattice point constitutes a motif. q The motif should be positioned identically at each lattice point (i. e. should not be rotated or distorted from point to point). Note: If the atom has spherical symmetry, rotations would not matter!

Revision: MOTIFS Geometrical Entity or a combination Shapes, atoms, ions… Physical Property Magnetization vector, field vortices, light intensity… § What is the role of the symmetry of the motif on the symmetry of the crystal?

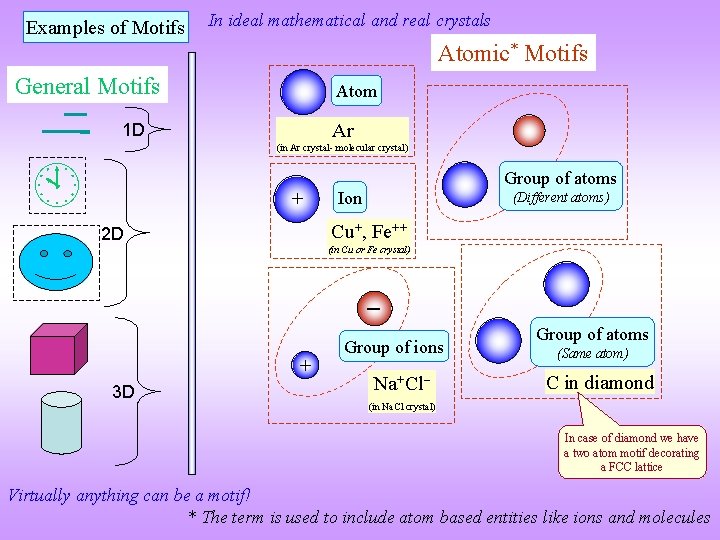
Examples of Motifs In ideal mathematical and real crystals Atomic* Motifs General Motifs Atom Ar 1 D (in Ar crystal- molecular crystal) + Group of atoms Ion (Different atoms) Cu+, Fe++ 2 D (in Cu or Fe crystal) + 3 D Group of ions Na+Cl Group of atoms (Same atom) C in diamond (in Na. Cl crystal) In case of diamond we have a two atom motif decorating a FCC lattice Virtually anything can be a motif! * The term is used to include atom based entities like ions and molecules

q Viruses can be crystallized and the motif now is an individual virus (a entity much larger than the usual atomic motifs) A complete virus is sitting as a motif on each lattice position (instead of atoms or ions!) We get a crystal of ‘virus’ Crystal of Tobacco Mosaic Virus [1] Crystal Physics, G. S. Zhdanov, Oliver & Boyd, Ediburgh, 1965

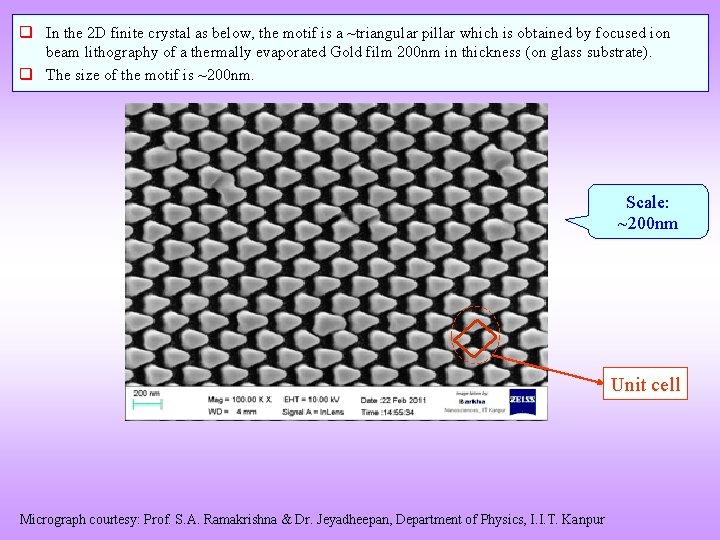
q In the 2 D finite crystal as below, the motif is a ~triangular pillar which is obtained by focused ion beam lithography of a thermally evaporated Gold film 200 nm in thickness (on glass substrate). q The size of the motif is ~200 nm. Scale: ~200 nm Unit cell Micrograph courtesy: Prof. S. A. Ramakrishna & Dr. Jeyadheepan, Department of Physics, I. I. T. Kanpur

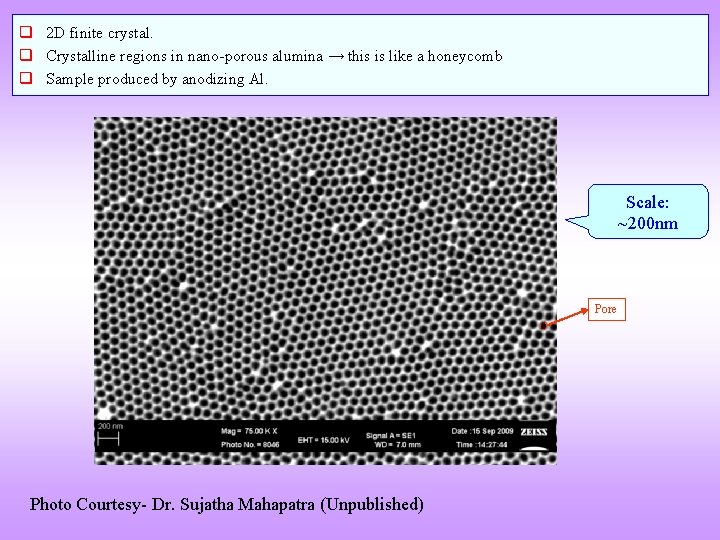
q 2 D finite crystal. q Crystalline regions in nano-porous alumina → this is like a honeycomb q Sample produced by anodizing Al. Scale: ~200 nm Pore Photo Courtesy- Dr. Sujatha Mahapatra (Unpublished)

Chip of the LED light sensing assembly of a mouse

q 3 D Finite crystal of metallic balls → motif is one brown metallic ball and one metallic ball (uncolored) [lattice is FCC]. Scale: ~mm

q Crystals have been synthesized with silver nanocrystals as the motif in an FCC lattice. Each lattice point is occupied by a silver nanocrystal having the shape of a truncated octahedron- a tetrakaidecahedron (with orientational and positional order). q The orientation relation between the particles and the lattice is as follows: [110] lattice || [110]Ag, [001]lattice || [1 10]Ag Ag nanocrystal as the motif

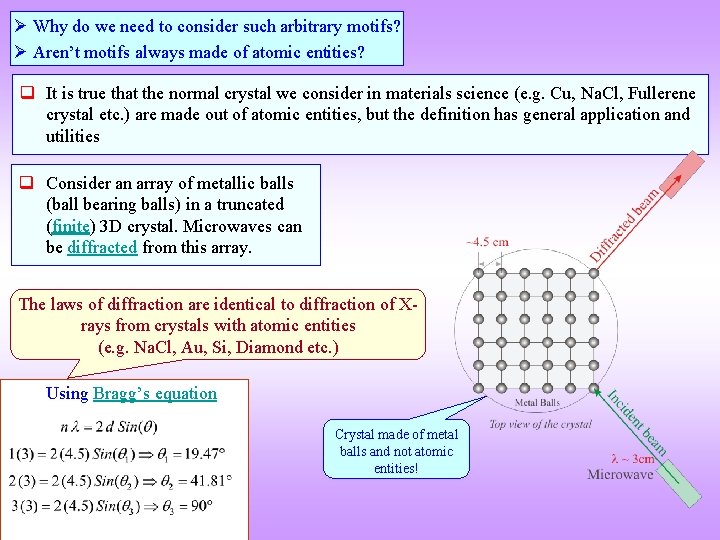
Why do we need to consider such arbitrary motifs? Aren’t motifs always made of atomic entities? q It is true that the normal crystal we consider in materials science (e. g. Cu, Na. Cl, Fullerene crystal etc. ) are made out of atomic entities, but the definition has general application and utilities q Consider an array of metallic balls (ball bearing balls) in a truncated (finite) 3 D crystal. Microwaves can be diffracted from this array. The laws of diffraction are identical to diffraction of Xrays from crystals with atomic entities (e. g. Na. Cl, Au, Si, Diamond etc. ) Using Bragg’s equation Crystal made of metal balls and not atomic entities!

q Example of complicated motifs include: Opaque and transparent regions in a photo-resist material which acts like an element in opto-electronics q A physical property can also be a motif decorating a lattice point q Experiments have been carried out wherein matter beams (which behave like waves) have been diffracted from ‘LASER Crystals’! Matter being diffracted from electromagnetic radiation! = + Lattice Motif Is now a physical property (electromagnetic flux density)* Scale: ~cm Ofcourse, it has a a certain geometry! An actual LASER crystal created by making LASER beams visible by smoke Things are little approximate in real life! * Yes, the physical property has a ‘geometry’!

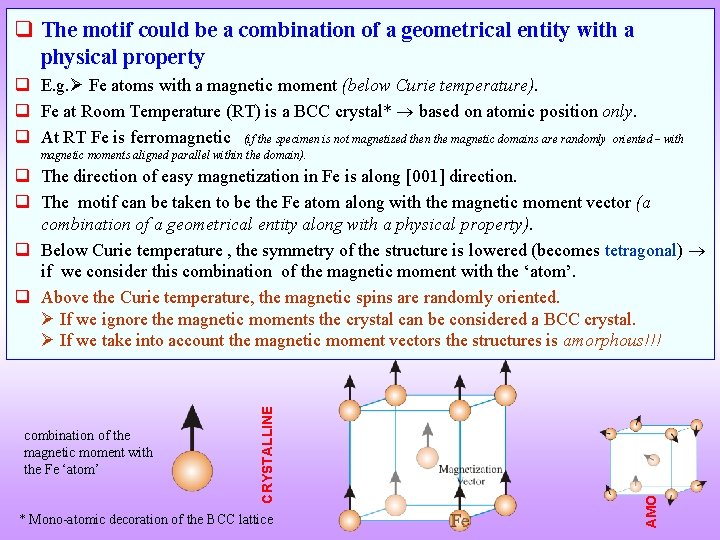
q The motif could be a combination of a geometrical entity with a physical property q E. g. Fe atoms with a magnetic moment (below Curie temperature). q Fe at Room Temperature (RT) is a BCC crystal* based on atomic position only. q At RT Fe is ferromagnetic (if the specimen is not magnetized then the magnetic domains are randomly oriented with magnetic moments aligned parallel within the domain). * Mono-atomic decoration of the BCC lattice AMORPHOUS combination of the magnetic moment with the Fe ‘atom’ CRYSTALLINE q The direction of easy magnetization in Fe is along [001] direction. q The motif can be taken to be the Fe atom along with the magnetic moment vector (a combination of a geometrical entity along with a physical property). q Below Curie temperature , the symmetry of the structure is lowered (becomes tetragonal) if we consider this combination of the magnetic moment with the ‘atom’. q Above the Curie temperature, the magnetic spins are randomly oriented. If we ignore the magnetic moments the crystal can be considered a BCC crystal. If we take into account the magnetic moment vectors the structures is amorphous!!!

q Wigner crystal q Electrons repel each other and can get ordered to this repulsive interaction. This is a Wigner crystal! (here, we ignore the atomic entitles). q We can also visualize other purely repulsively ordered crystals.

Ordering of Nuclear spins q We had seen that electron spin (magnetic moment arising from the spin) can get ordered (e. g. ferromagnetic ordering of spins in solid Fe at room temperature). q Similarly, nuclear spin can also get ordered.

The case of the vortices in type-II superconductors q In type-II superconductors the transition to non-superconducting state occurs gradually via penetration of flux lines via magnetic vortices. q These vortices can ‘crystallize’ in a hexagonal Abrikosov lattice (lattice decorated with a physical property). This is a repulsively ordered crystal. Causing disorder in this perfect crystal cost ‘elastic energy’. q On heating or increasing the magnetic field strength the crystal may ‘melt’ (i. e. melting of a physical property lattice). Before melting the flux tubes become ‘wiggly’.