Module 6 Flowsheet Environmental Impact Assessment Chapter 11
















- Slides: 33

Module 6: Flowsheet Environmental Impact Assessment Chapter 11 David R. Shonnard Hui Chen Department of Chemical Engineering Michigan Technological University of Texas at Austin Michigan Technological University 1

Module 6: Outline After the flowsheet input output structure, unit operation designations, and mass/heat integration have been completed, the last step in the process to improve the environmental performance of a chemical process design is to perform a detailed environmental impact assessment l Educational goals and topics covered in the module l Potential uses of the module in chemical engineering courses l Review of environmental impact assessment methods l Application of Tier 3 environmental impact assessment to a detailed flowsheet - Chapter 11 University of Texas at Austin Michigan Technological University 2

Module 6: Educational goals and topics covered in the module Students will: l learn to apply a systematic risk assessment methodology to the evaluation of chemical process designs l integrate emission estimation, environmental fate and transport calculation, and relative risk assessment to rank process design alternatives University of Texas at Austin Michigan Technological University 3

Module 6: Potential uses of the module in chemical engineering courses Process Design course: • develop and use environmental objective functions to rank process design alternatives • rank process designs quantitatively based on environmental criteria Transport phenomena course: • Module on interphase mass transfer in the environment University of Texas at Austin Michigan Technological University 4

Module 6: Essential features of environmental impact assessment for chemical process design Computationally efficient Environmental performance metrics quickly calculated using output from commercial process simulators Link waste generation and release to environmental impacts Environmental metrics linked to process parameters Impacts based on a systematic risk assessment methodology Release estimates fate and transport exposure risk University of Texas at Austin Michigan Technological University 5

Module 6: Systematic risk assessment methodology National Academy of Sciences, 1983 1. Hazard Identification (which chemicals are important? ) 2. Exposure assessment (release estimation, fate and transport, dose assessment) 3. Toxicity assessment (chemical dose - response relationships) 4. Risk Characterization (magnitude and uncertainty of risk) Result: Quantitative risk assessment (e. g. excess cancers) Atmospheric dispersion Model, Ca Thibodeaux, L. J. 1996, Environmental Chemodynamics, John Wiley & Sons University of Texas at Austin Michigan Technological University 6

Module 6: Quantitative risk calculation Carcinogenic Risk Example (inhalation route) Exposure Dose - Response Relationship, Slope Factor Result: # excess cancers per 106 cases in the population; 10 -4 to 10 -6 acceptable Disadvantage: Only a single compartment is modeled / Computationally inefficient Highly uncertain prediction of risk University of Texas at Austin Michigan Technological University 7

Module 6: Relative risk calculation Carcinogenic Risk Example (inhalation route) Result: Risk of a chemical relative to a well-studied benchmark compound Advantage: If C is calculated for all compartments using a multimedia compartment model, computationally efficient University of Texas at Austin Michigan Technological University 8

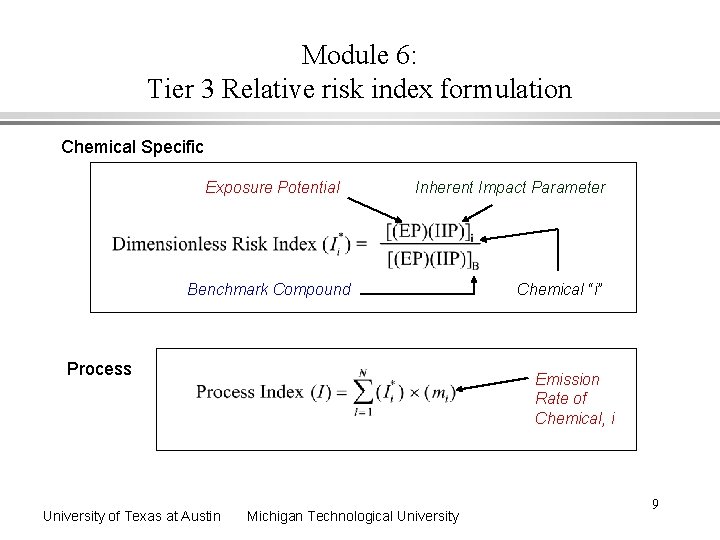
Module 6: Tier 3 Relative risk index formulation Chemical Specific Exposure Potential Inherent Impact Parameter Benchmark Compound Process University of Texas at Austin Chemical “i” Emission Rate of Chemical, i Michigan Technological University 9

Module 6: Airborne emissions estimation o Unit Specific EPA Emission Factors n n n o Correlation (AP- 42, EPA) n n o Storage tanks, wastewater treatment Fugitive sources (pumps, valves, fittings) Criteria Pollutants from Utility Consumption n n o Distillation/stripping column vents Reactor vents Fugitive sources Factors for CO 2, CO, SO 2, NOx, AP- 42 (EPA) factors Process Simulators (e. g. HYSYS ) University of Texas at Austin Michigan Technological University 10

Module 6: Release estimates based on surrogate processes Waste stream summaries based on past experience 1. Hedley, W. H. et al. 1975, “Potential Pollutants from Petrochemical Processes”, Technomics, Westport, CT 2. AP-42 Document, Chapters 5 and 6 on petroleum and chemical industries, Air CHIEF CD, www. epa. gov/ttn/chief/airchief. htm 3. Other sources i. Kirk-Othmer Encyclopedia of Chemical Technology, 1991 ii. Hydrocarbon Processing, “Petrochemical Processes ‘ 99”, March 1999. University of Texas at Austin Michigan Technological University 11

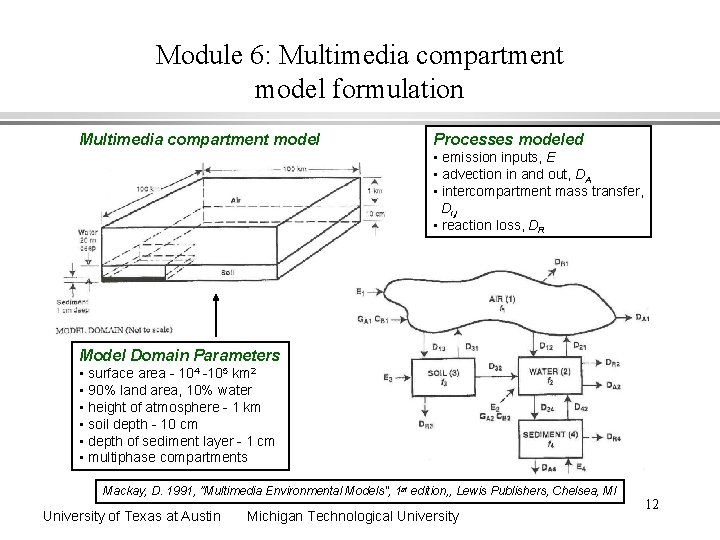
Module 6: Multimedia compartment model formulation Multimedia compartment model Processes modeled • emission inputs, E • advection in and out, DA • intercompartment mass transfer, Di, j • reaction loss, DR Model Domain Parameters • surface area - 104 -105 km 2 • 90% land area, 10% water • height of atmosphere - 1 km • soil depth - 10 cm • depth of sediment layer - 1 cm • multiphase compartments Mackay, D. 1991, ”Multimedia Environmental Models", 1 st edition, , Lewis Publishers, Chelsea, MI University of Texas at Austin Michigan Technological University 12

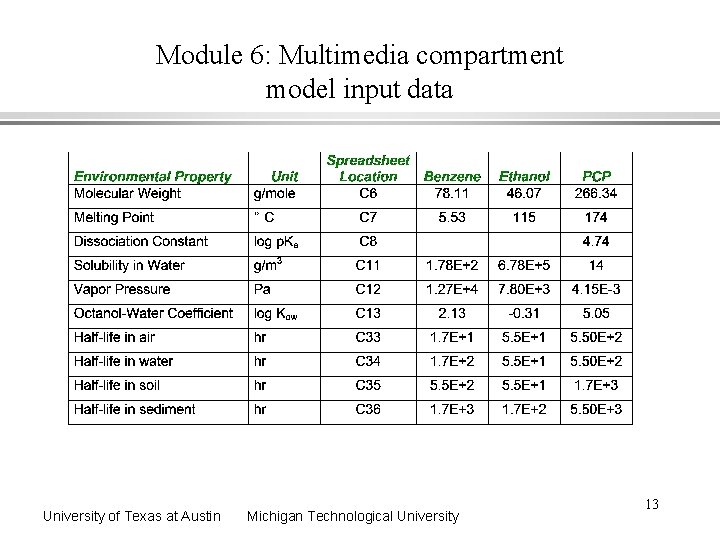
Module 6: Multimedia compartment model input data University of Texas at Austin Michigan Technological University 13

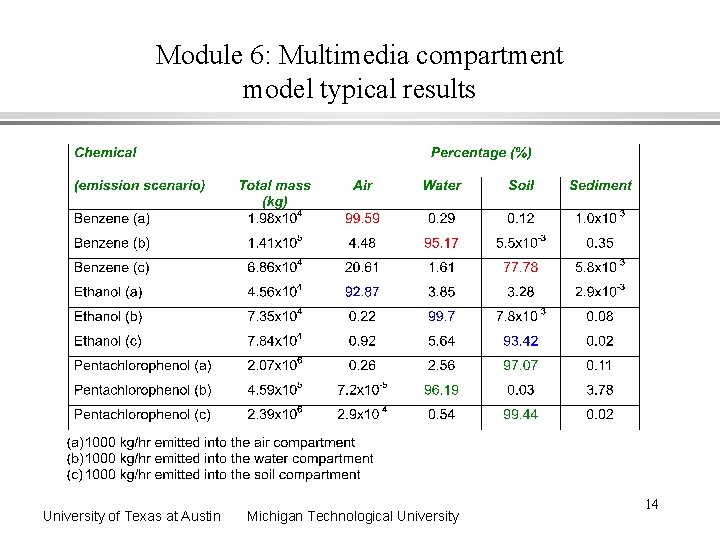
Module 6: Multimedia compartment model typical results University of Texas at Austin Michigan Technological University 14

Module 6: Multimedia compartment model typical results - interpretations 1. The percentages in each environmental compartment depend upon the emission scenario a) the highest air concentrations result from emission into the air b) the highest water concentrations are from emission into water c) the highest soil concentrations are from emission into soil d) highest sediment concentrations are from emission into water 2. Chemical properties dictate percentages and amounts a) high KH results in high air concentrations b) high KOW results in high soil concentrations c) high reactions half lives results in highest pollutant amounts University of Texas at Austin Michigan Technological University 15

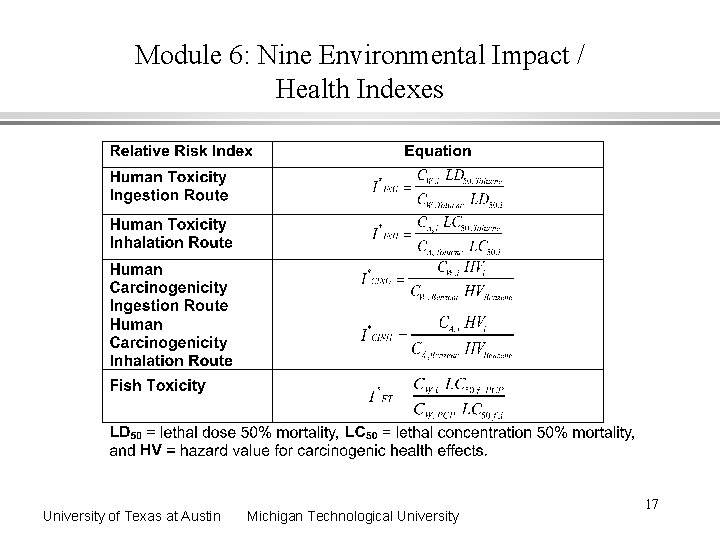
Module 6: Nine Environmental Impact / Health Indexes University of Texas at Austin Michigan Technological University 16

Module 6: Nine Environmental Impact / Health Indexes University of Texas at Austin Michigan Technological University 17

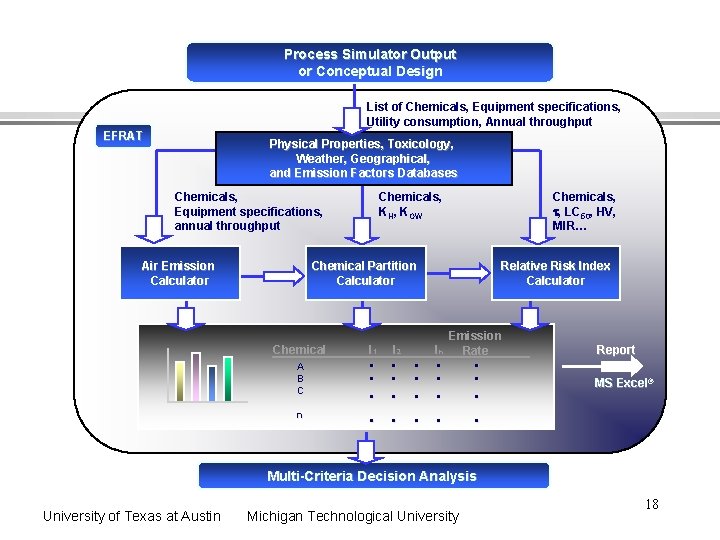
Process Simulator Output or Conceptual Design List of Chemicals, Equipment specifications, Utility consumption, Annual throughput EFRAT Physical Properties, Toxicology, Weather, Geographical, and Emission Factors Databases Chemicals, Equipment specifications, annual throughput Air Emission Calculator Chemicals, KH, KOW Chemicals, t, LC 50, HV, MIR… Chemical Partition Calculator Chemical A B C n I 1 . . I 2 . . . . Relative Risk Index Calculator In . . Emission Rate . . Report MS Excel® Multi-Criteria Decision Analysis University of Texas at Austin Michigan Technological University 18

Module 6: Software tools for environmental impact assessment of process designs Environmental Fate and Risk Assessment Tool (EFRAT) • links with HYSYS for automated assessments WAste Reduction Algorithm (WAR) • reported to be linked with Chem. CAD • US EPA National Risk Management Research Laboratory Cincinnati, OH Dr. Heriberto Cabezas and Dr. Douglas Young US Environmental Protection Agency National Risk Management Research Laboratory 26 W. Martin Luther King Dr. Cincinnati, OH 45268 University of Texas at Austin Michigan Technological University 19

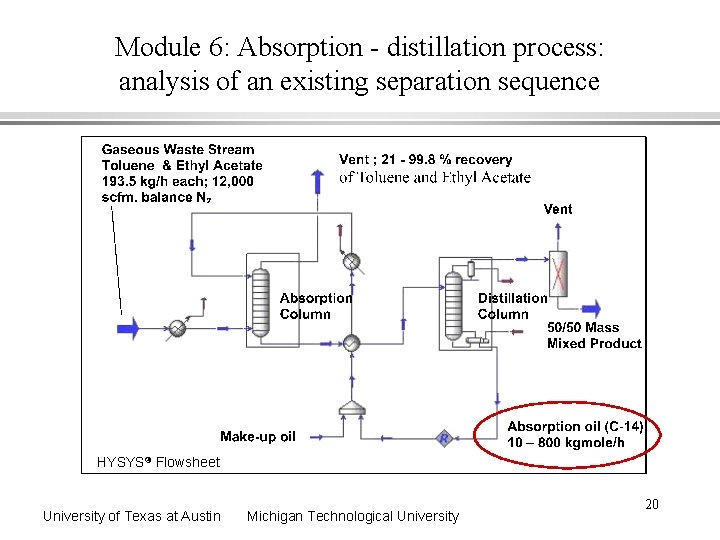
Module 6: Absorption - distillation process: analysis of an existing separation sequence HYSYS Flowsheet University of Texas at Austin Michigan Technological University 20

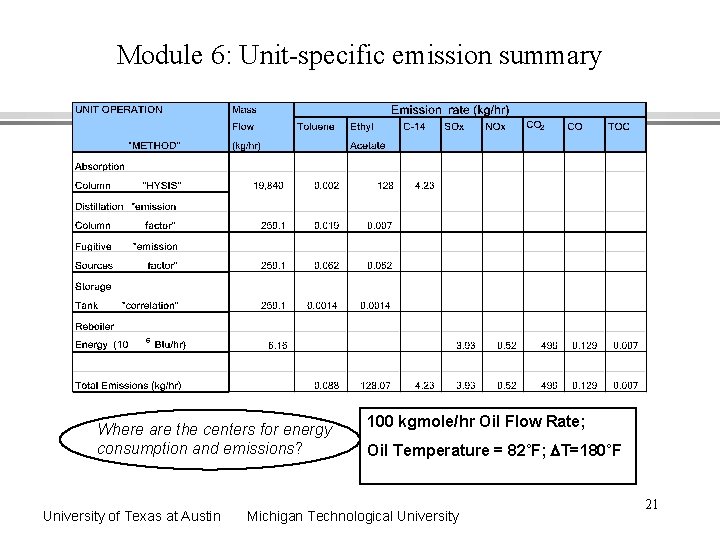
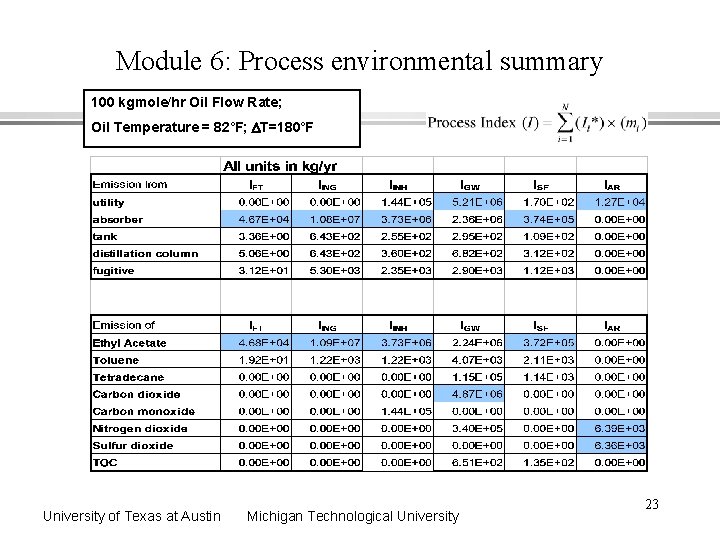
Module 6: Unit-specific emission summary Where are the centers for energy consumption and emissions? University of Texas at Austin 100 kgmole/hr Oil Flow Rate; Oil Temperature = 82˚F; DT=180˚F Michigan Technological University 21

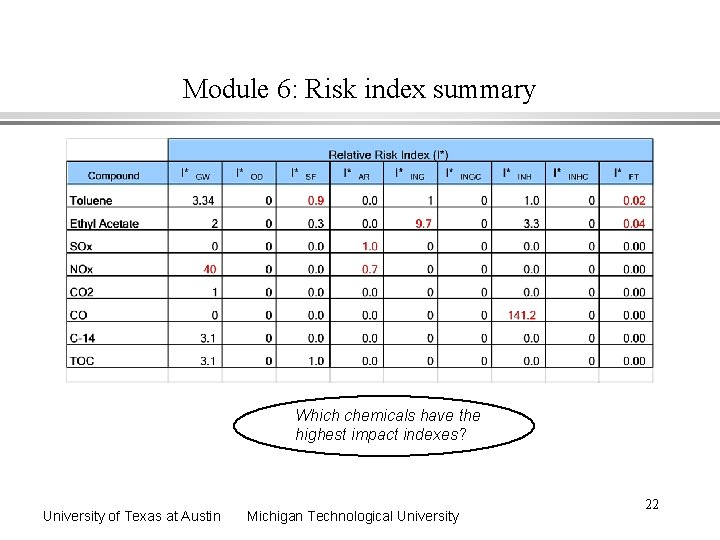
Module 6: Risk index summary Which chemicals have the highest impact indexes? University of Texas at Austin Michigan Technological University 22

Module 6: Process environmental summary 100 kgmole/hr Oil Flow Rate; Oil Temperature = 82˚F; DT=180˚F University of Texas at Austin Michigan Technological University 23

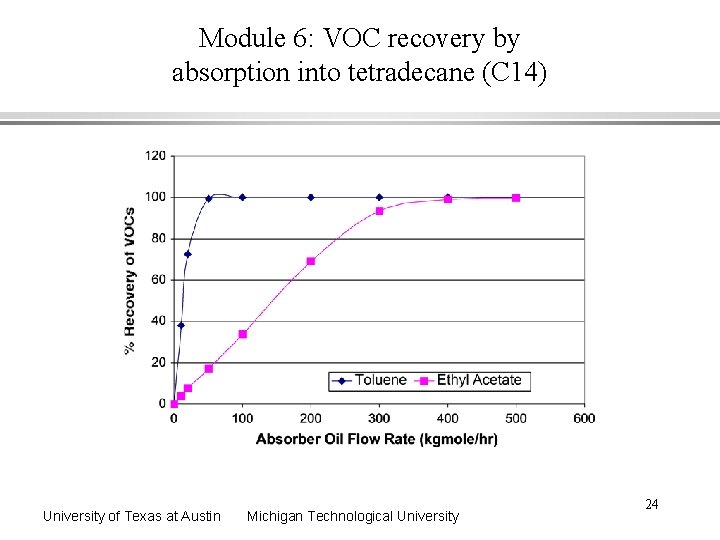
Module 6: VOC recovery by absorption into tetradecane (C 14) University of Texas at Austin Michigan Technological University 24

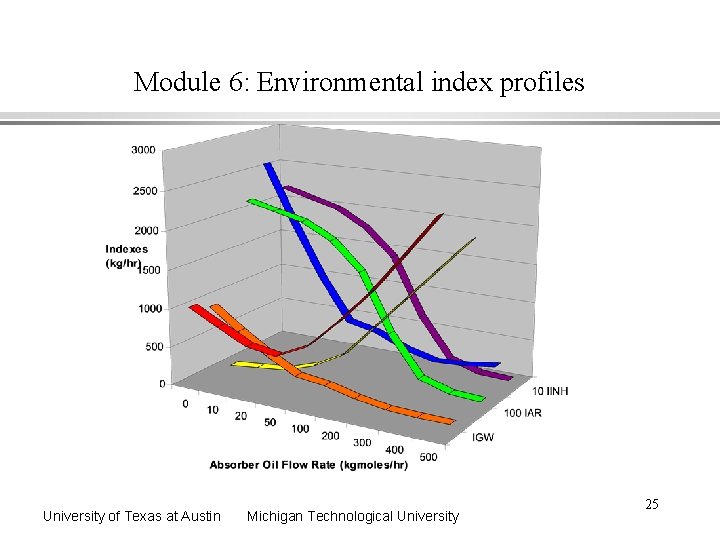
Module 6: Environmental index profiles University of Texas at Austin Michigan Technological University 25

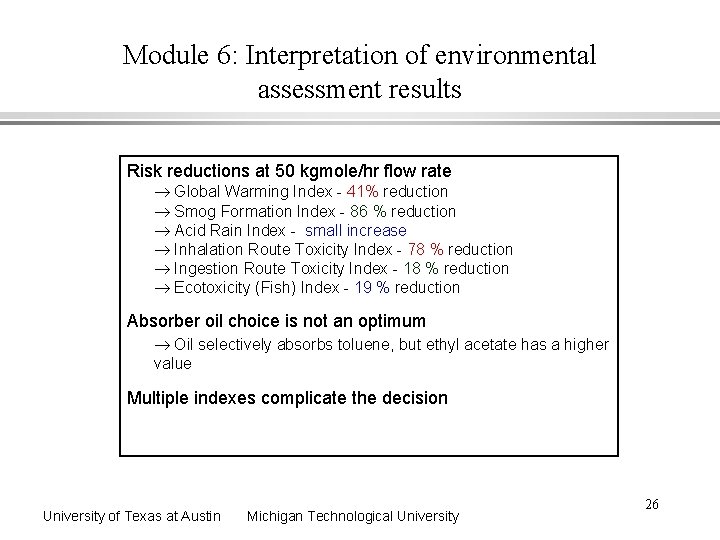
Module 6: Interpretation of environmental assessment results Risk reductions at 50 kgmole/hr flow rate Global Warming Index - 41% reduction Smog Formation Index - 86 % reduction Acid Rain Index - small increase Inhalation Route Toxicity Index - 78 % reduction Ingestion Route Toxicity Index - 18 % reduction Ecotoxicity (Fish) Index - 19 % reduction Absorber oil choice is not an optimum Oil selectively absorbs toluene, but ethyl acetate has a higher value Multiple indexes complicate the decision University of Texas at Austin Michigan Technological University 26

Module 6: Maleic anhydride from n-butane process flowsheet evaluation l Use of EFRAT : evaluate the MA process l Basecase (Dibutyl phthalate absorber oil) with and without heat integration l Simulate 3 case studies using heat integrated flowsheet » Dibutyl phthalate absorber oil » Dibenzyl ether absorber oil » Diethylene glycol butyl ether acetate absorber oil University of Texas at Austin Michigan Technological University 27

Module 6: Maleic anhydride from n-butane: Use of EFRAT on basecase flowsheet l Follow the tutorial instructions given in the notebook! l The SCENE file has been linked to a HYSYS case file l Add three additional emission sources l Complete the relative risk assessment calculations University of Texas at Austin Michigan Technological University 28

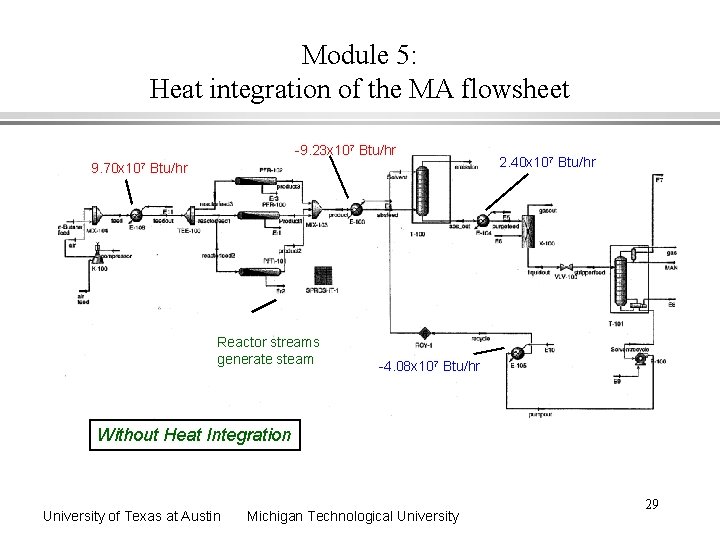
Module 5: Heat integration of the MA flowsheet -9. 23 x 107 Btu/hr 9. 70 x 107 Btu/hr Reactor streams generate steam 2. 40 x 107 Btu/hr -4. 08 x 107 Btu/hr Without Heat Integration University of Texas at Austin Michigan Technological University 29

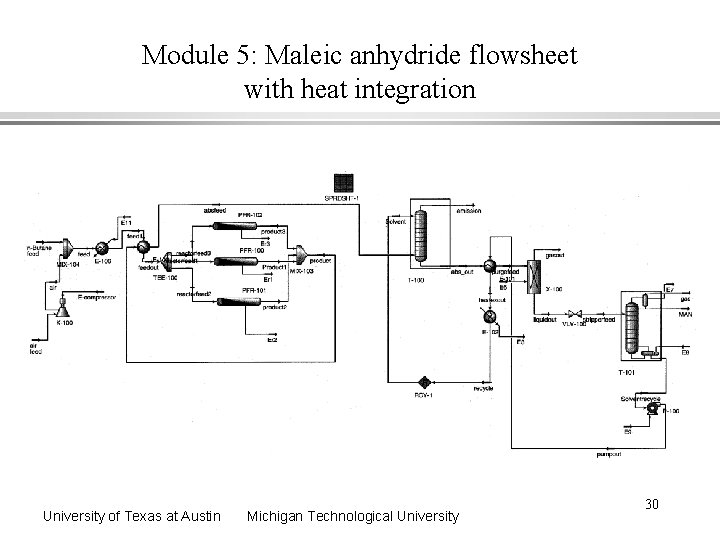
Module 5: Maleic anhydride flowsheet with heat integration University of Texas at Austin Michigan Technological University 30

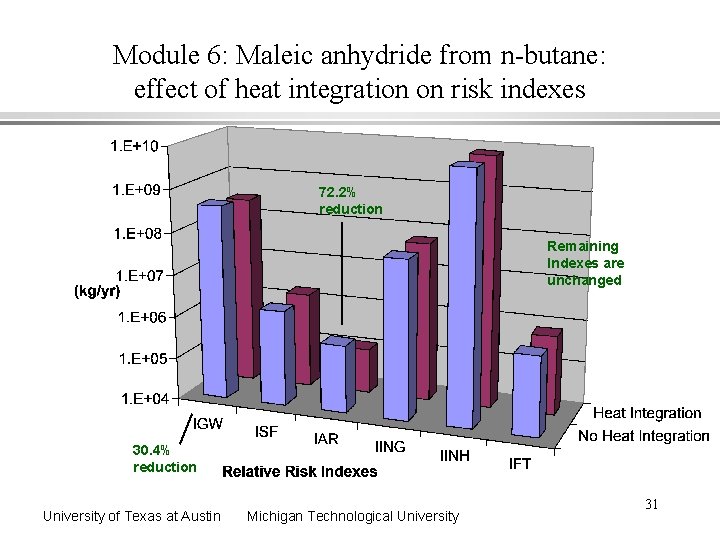
Module 6: Maleic anhydride from n-butane: effect of heat integration on risk indexes 72. 2% reduction Remaining Indexes are unchanged 30. 4% reduction University of Texas at Austin Michigan Technological University 31

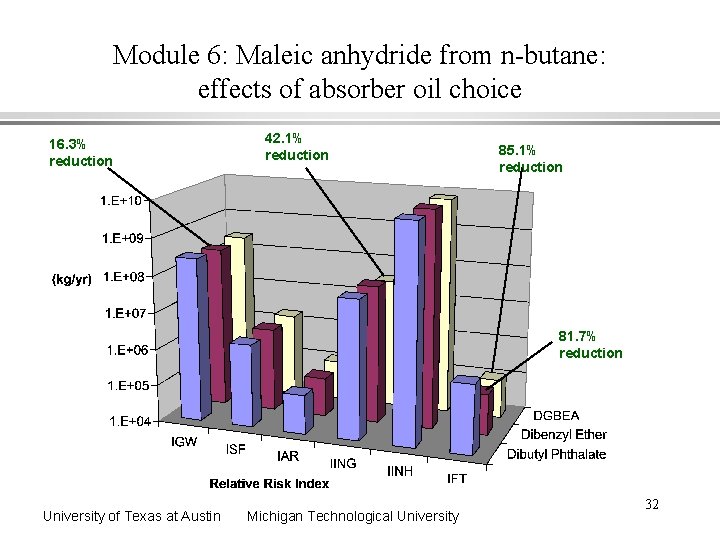
Module 6: Maleic anhydride from n-butane: effects of absorber oil choice 16. 3% reduction 42. 1% reduction 85. 1% reduction 81. 7% reduction University of Texas at Austin Michigan Technological University 32

Module 6: Summary / Conclusions l Educational goals and topics covered in the module l Potential uses of the module in chemical engineering courses l Review of environmental impact assessment methods l Application of Tier 3 environmental impact assessment to a detailed flowsheet - Chapter 11 » Heat integration of the Maleic Anhydride flowsheet » Effects of absorber oil choice for the MA flowsheet University of Texas at Austin Michigan Technological University 33