Holes Human Anatomy and Physiology 12 th Edition











- Slides: 16

Hole’s Human Anatomy and Physiology 12 th Edition Chapter 2, pp. 51 -55 Introduction to Chemistry, part I Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. 1 -1

Chemical Basis of Life Why study chemistry in an Anatomy and Physiology class? 2 -2

Matter • takes up space + has weight • solids, liquids and gases • atom = smallest stable unit of matter • different types of atoms form from different elements 2 -3

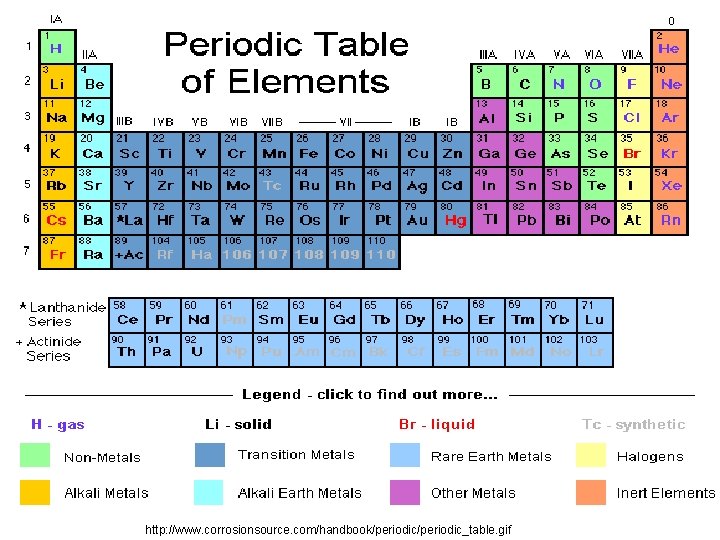
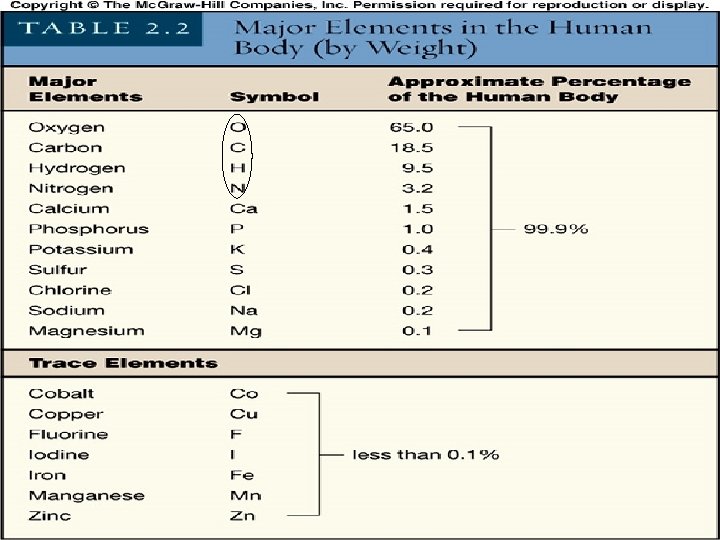
Elements • 92 naturally occurring elements • additional created by man • form all matter, both living and non-living • 26 elements found in humans • each type of element is composed of chemically identical atoms • an atom is the smallest particle of an element 2 -3

http: //www. corrosionsource. com/handbook/periodic_table. gif


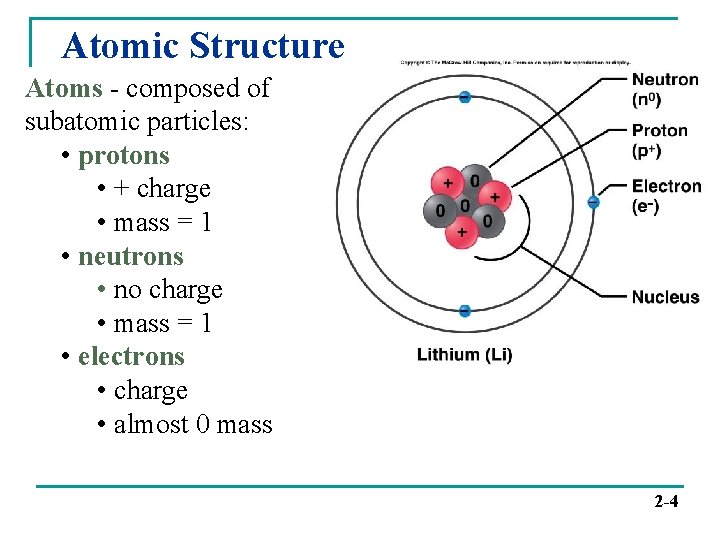
Atomic Structure Atoms - composed of subatomic particles: • protons • + charge • mass = 1 • neutrons • no charge • mass = 1 • electrons • charge • almost 0 mass 2 -4

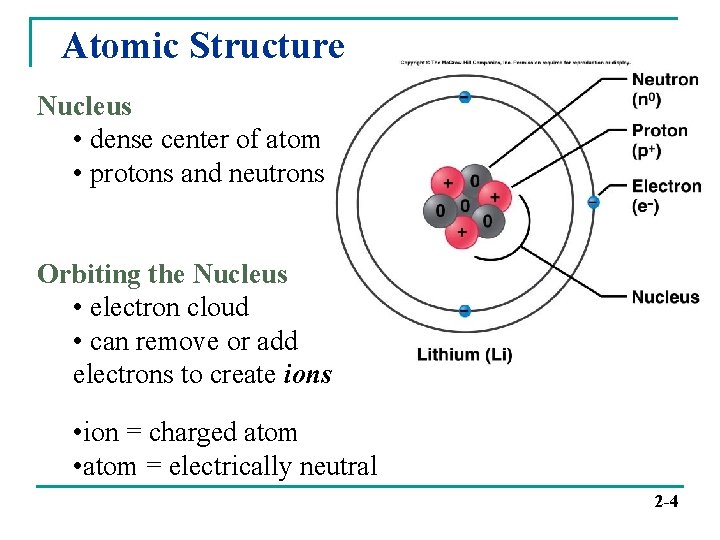
Atomic Structure Nucleus • dense center of atom • protons and neutrons Orbiting the Nucleus • electron cloud • can remove or add electrons to create ions • ion = charged atom • atom = electrically neutral 2 -4

COMPARISON OF SUB-ATOMIC PARTICLES Location Charge Mass Can transfer from one atom to another? Proton Neutron Electrons An _____ has # protons = # electrons. A) ion C) both A & B are correct B) atom D) neither A nor B are correct If an atom loses or gains an electron, is it electrically neutral? What is it then named?

Atomic Weight n Atomic Weight q # protons + # neutrons q electrons do not contribute weight n Ex. oxygen has 8 protons and 8 neutrons q Atomic weight = O 16

Isotopes n atoms have same number of protons but a different number of neutrons q same atomic number, different atomic weight n ex. oxygen forms isotopes (O 16, O 17, O 18) n same element! n How many protons in O 16? O 17? O 18? How many neutrons? unstable isotopes are radioactive and emit energy – used in medicine and research n

Atomic Number Determines the Element Atomic Number • # protons • each element has a unique atomic number • will the number of protons in an atom also determine how many electrons there are in an atom? • f an atom has 9 protons, how many electrons will it have? 2 -5

Electrons • found in electron shells (energy shells) • each shell holds a limited number of electrons • 1 st shell holds up to 2 electrons • 2 nd shell holds up to 8 electrons “Octet rule” • 3 rd shell holds up to 8 electrons • lower shells are filled first • outermost shell is known as the “valence” shell • a full valence shell stable atom (unreactive) • unstable atoms will react with each other to form stable compounds or molecules • Will an unstable atom have a full valence shell? 2 -8

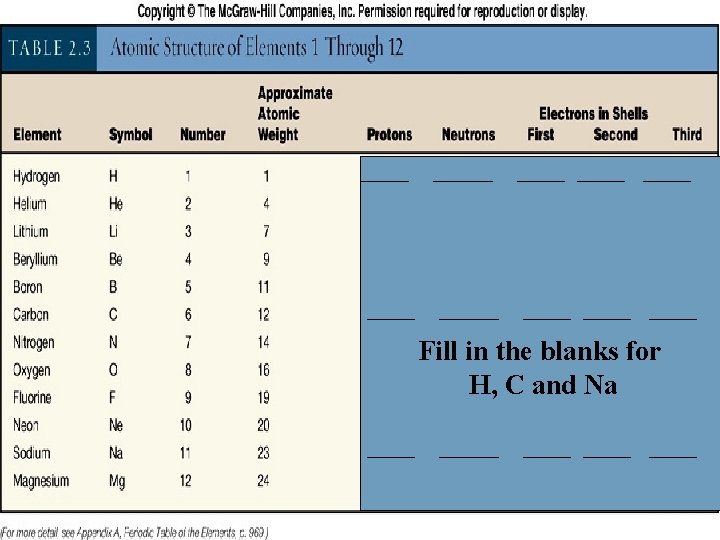
_____ ____ ____ Fill in the blanks for H, C and Na _____ ____

Molecules and Compounds Molecule –when two or more atoms chemically combine Compound –when two or more atoms of different elements chemically combine in defined proportions. Molecular formulas – elements present and the number of each atom present in the molecule H 2 C 6 H 12 O 6 H 2 O 2 -7

Concept Review 1. Which particle of the atom: 2 - can transfer from one atom to another? - creates isotopes? - has almost no mass? - gives the atom its properties? 2. Which of the following are “stable” atoms? 3. What do you think will happen to each of the nonstable atoms below?