EPID 503 Class 12 Cohort Study Design Hierarchy















- Slides: 23

EPID 503 – Class 12 Cohort Study Design

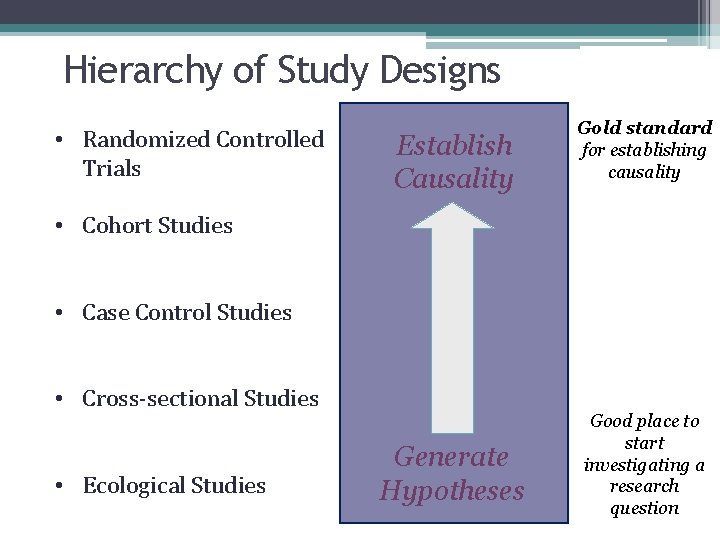
Hierarchy of Study Designs • Randomized Controlled Trials Establish Causality Gold standard for establishing causality • Cohort Studies • Case Control Studies • Cross-sectional Studies • Ecological Studies Generate Hypotheses Good place to start investigating a research question

Last Class • Explored randomized controlled trials (RCTs) • Randomized to try to address the question – what would have happened if same individual had not been exposed (counterfactual)

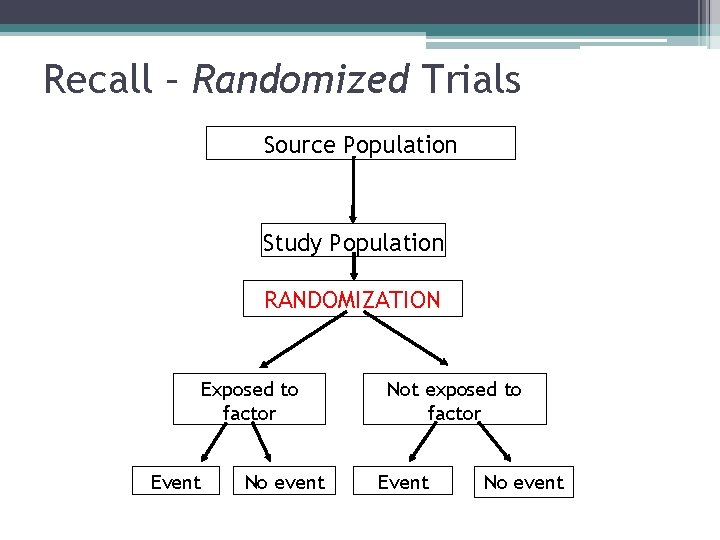
Recall – Randomized Trials Source Population Study Population RANDOMIZATION Exposed to factor Event No event Not exposed to factor Event No event

5 Randomized Trials Not Always Appropriate

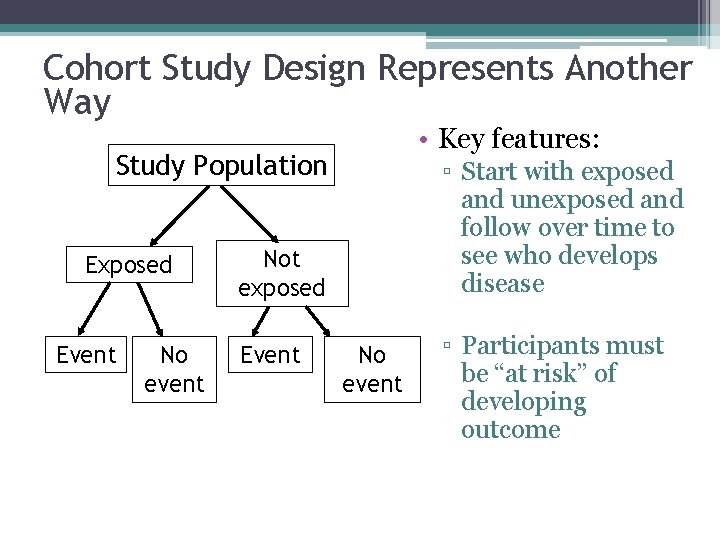
Cohort Study Design Represents Another Way • Key features: Study Population Exposed Event No event ▫ Start with exposed and unexposed and follow over time to see who develops disease Not exposed Event No event ▫ Participants must be “at risk” of developing outcome

Advantages • Can estimate incidence (risk or rate) of disease • Exposure precedes development of disease • Multiple outcomes (and sometimes multiple exposures) can be studied

Disadvantages • • • Not good for rare outcomes Long-term follow-up required Expensive Generally large sample sizes needed Changes over time in criteria and methods can lead to problems with inferences • People self-select exposures so exposed and unexposed may differ with respect to important characteristics

When is a Cohort Useful? • When the outcome is common or rare? • When the exposure is common or rare? • When the time between exposure and disease is long or short? • To study one outcome or many? • To study one exposure or many?

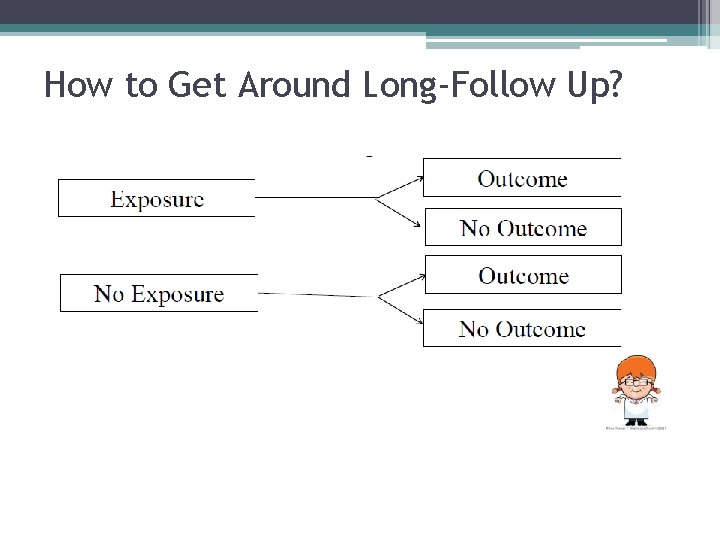
How to Get Around Long-Follow Up?

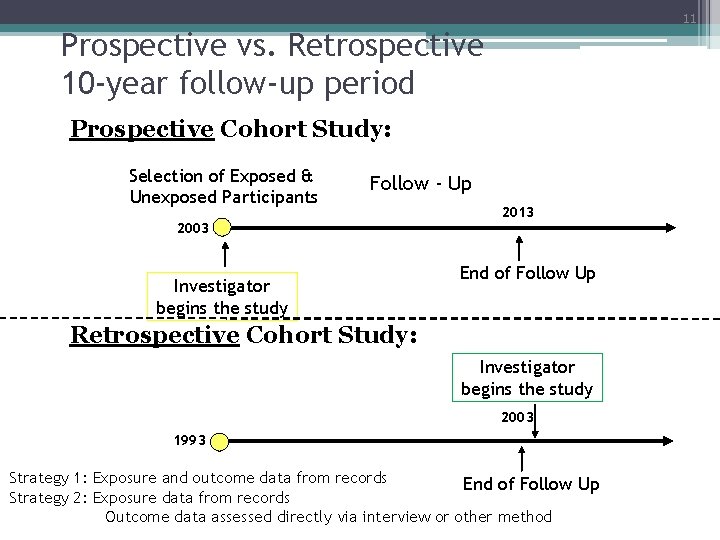
11 Prospective vs. Retrospective 10 -year follow-up period Prospective Cohort Study: Selection of Exposed & Unexposed Participants Follow - Up 2003 Investigator begins the study 2013 End of Follow Up Retrospective Cohort Study: Investigator begins the study 2003 1993 Strategy 1: Exposure and outcome data from records End of Follow Up Strategy 2: Exposure data from records Outcome data assessed directly via interview or other method

How do we analyze and summarize data from a cohort?

Exposure Yes No Total Yes a Disease No Total b a+b c d c+d a+c b+d a + b+ c+ d What measure can we calculate here?

Exposure Yes No Total Disease Yes No a b Total a+b c d c+d a+c b+d a + b+ c+ d Cumulative Incidence exp = a/(a+b) = Risk Ratio Cumulative Incidence no exp = c/(c+d)

Example Risk Ratio Interpretation • Relative Risk = 1. 25 Among the population studied during the time frame of interest, exposed persons were 1. 25 times more likely to develop disease than unexposed persons. Among the population studied during the time frame of interest, exposed persons were 25% more likely to develop disease than unexposed persons.

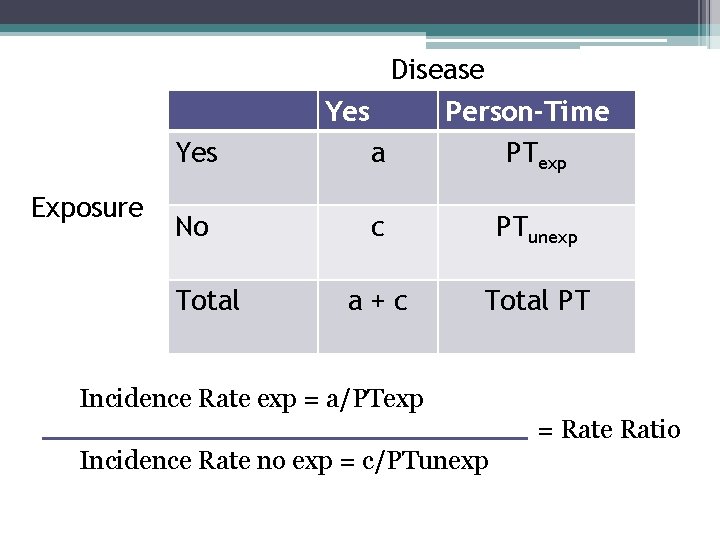
Disease Yes Exposure Person-Time PTexp Yes a No c PTunexp a+c Total PT Total What measure can we calculate here?

Yes Exposure No Total Disease Yes Person-Time a PTexp c PTunexp a+c Total PT Incidence Rate exp = a/PTexp = Rate Ratio Incidence Rate no exp = c/PTunexp

Example Ratio Interpretation • Rate Ratio = 0. 9 Among the population studied during the time frame of interest, the rate of disease in exposed persons was 0. 9 times that of unexposed persons. Among the population studied during the time frame of interest, the rate of disease in exposed persons was 10% less than that in unexposed persons.

Confidence Intervals

What is a confidence interval? A 95% confidence interval tells us that, with 95% probability, the true value of the variable is contained within the interval. The greater the desired level of confidence, the wider the confidence interval. 90% confidence 95% confidence 99% confidence * Point Estimate

Confidence Interval of the Relative Risk Disease No Disease Total Exposed a b a+b Unexposed c d c+d a+c b+d a+b+c+d Total RR = 95% Confidence interval = a/(a+b) c/(c+d)

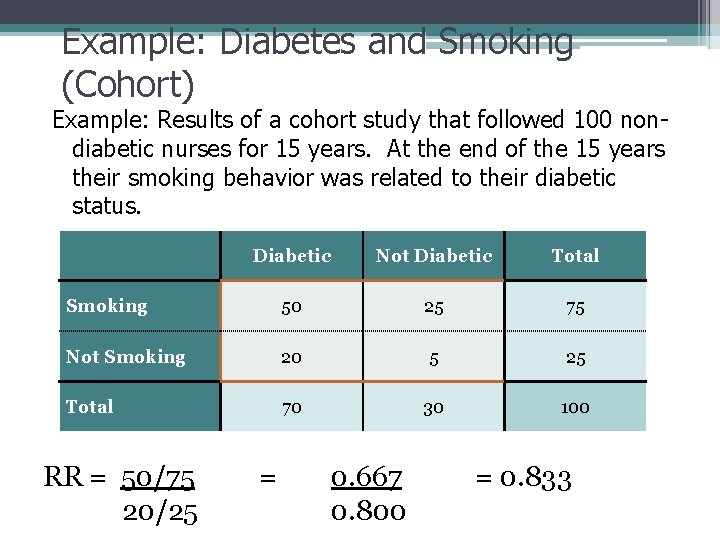
Example: Diabetes and Smoking (Cohort) Example: Results of a cohort study that followed 100 nondiabetic nurses for 15 years. At the end of the 15 years their smoking behavior was related to their diabetic status. Diabetic Not Diabetic Total Smoking 50 25 75 Not Smoking 20 5 25 Total 70 30 100 RR = 50/75 20/25 = 0. 667 0. 800 = 0. 833

Example: Diabetes and Smoking (Cohort) 95% CI for the RR = = (0. 647, 1. 073) Interpretation: The confidence interval contains the null value of 1, therefore there is NOT a statistically significant association between smoking and diabetes in this study population with 95% confidence.