Designing observational studies descriptive crosssectional case control and

Designing observational studies (descriptive, crosssectional, case control and cohort studies) Prof Dr Meltem ÇÖL Department of Public Health Ankara University Medical Faculty 1

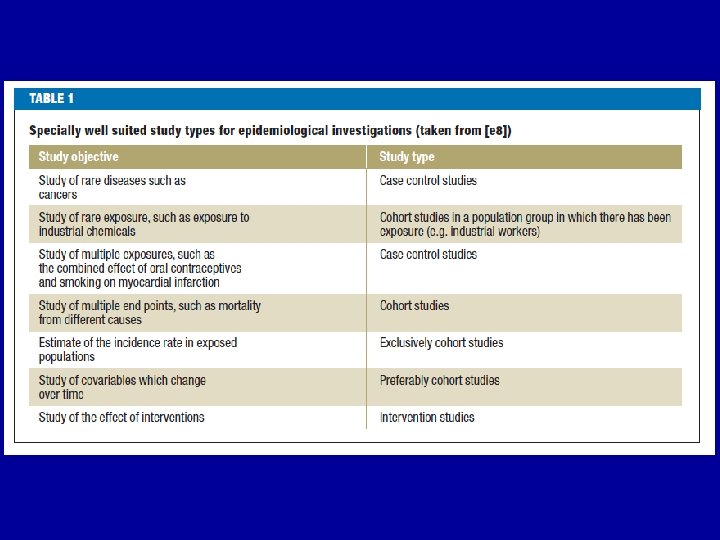
RESEARCH TYPES IN MEDICINE Overview of design strategies • Observational Studies – Descriptive studies Correlational studies Case reports Case series – Analytic studies • Cross-sectional surveys • Case-control studies • Cohort studies • Intervention studies • Clinical trial • Field trial • Community trial 2

Descriptive Studies • Correlational studies • Case reports or Case series Provides information on various characteristics of: Person, Place or Time Answer the question of: Who? Where? When? Each has advantages and disadvantages. . . 3

Descriptive Studies • Describe; Person, Place and Time: • Often uses data from routinely collected and readily available. • Less expensive, less time consuming 4

Descriptive Studies • It is used for – Planning of health services, Determination of priorities – First step for analytic studies (Description provides some informations about etiological factors) 5

Correlational Studies • Uses data from entire populations to compare disease frequencies between different groups. • “ r “ is calculated • The value of r : between (+1) and (-1) 6

Correlational Studies Example: • Death rates from 44 states were correlated with per capita cigarette sales • Correlation between per capita daily consumption of meat and rates of colon cancer from a large number of countries 7

Correlational Studies Advantages • First step in investigating a possible exposure-disease relationship • Can be done quickly-inexpensively • Often uses already available information 8

Correlational Studies Disadvantages • Inability to link exposure with disease in particular individuals • The lack of ability to control for the effects of potential confounding factors 9

Case reports • Describe the experience of a single patient 10

Case reports • Most common types of articles in medical journals are case reports and case series • Document unusual medical occurrences • Represent the first clues of new diseases or adverse effects of exposures 11

Case reports • Can formulate Hypothesis but • Cannot test Hypothesis 12

Case series • Collection of more than 1 case which occure in a short period. • Emergence New disease or epidemic 13

Case series Example: • Creech and Johnson (1974) reported: 3 men with angiosarcoma of the liver among workers at a vinyl chloride plant Hypothesis: “Vinyl chloride” caused “Hepatic angiosarcoma” Later, two analytic studies supported it • AIDS from 5 cases recognized as a new disease 14

Conclusion • Descriptive epidemiological studies are; – useful to public health administrators planning for health care utilization and resource allocation – important information to the formulation of etiologic hypothesis 15

Conclusion – each type of study provide valuable information on who is getting the disease? , where it is more and less common? , when it is occurring? – They generally can not test etiologic hypothesis 16

RESEARCH TYPES IN MEDICINE Overview of design strategies • Observational Studies – Descriptive studies Correlational studies Case reports Case series - Analytic studies • Cross-sectional studies • Case-control studies • Cohort studies • Intervention studies • Randomized controlled trial • Field trial • Community trial 17

Analytic studies • Try to find answer for question ; WHY ? • Test hypothesis (appropriate comparison group allows testing of hypothesis) 18

Analytic studies Three basic types; Cross sectional Case - control Cohort study • Each has advantages & disadvantages • Decision is based on: features of the exposure and disease current state of knowledge, available time, resources, etc. 19

Cross-sectional studies • • Population survey Community survey Epidemiologic survey Screening 20

Cross- sectional studies Community Sampling method Sample Questionnaire, Physical examination, Laboratory methods Disease (+) Disease (-) Exposure(+) Exposure (-) Exposure (+) Exposure (-) 21

Cross-sectional studies Disease (+) Prevalence= ---------------- X 100 Disease (+) + Disease (-) 22

Cross-sectional studies • Since exposure and disease status are assessed at a single point in time: – Not-possible to determine whether the exposure preceded or resulted from the disease • Provide information about; – the frequency and characteristics of a disease – health care needs and health status of a population 23

Cross-sectional studies General view: • In the population (country, city, district, school, workplace, etc. ) • Especially on the sample • Prevalence rate • High participation needed • Source of data: records or questionnaire, physical exam, lab test results, etc. 24

Cross-sectional studies Advantages • • In a short time Not expensive Useful data Results can be generalized (because on the whole population or on sample) 25

Cross-sectional studies Disadvantages • EXPOSURE - RESULT Relationship may not be safe (Because they are assessed at the same time) • May be memory bias (forgetfulness) 26
![Case-control studies Population or Hospital Cases [Disease (+)] Exposure (+) Exposure (-) Controls [Disease Case-control studies Population or Hospital Cases [Disease (+)] Exposure (+) Exposure (-) Controls [Disease](http://slidetodoc.com/presentation_image_h2/4af1efb1f068b4baeb4953686f87602d/image-27.jpg)
Case-control studies Population or Hospital Cases [Disease (+)] Exposure (+) Exposure (-) Controls [Disease (-)] Exposure (+) Exposure (-) • Compare each group’s exposure rates • From disease to causes Retrospective 27
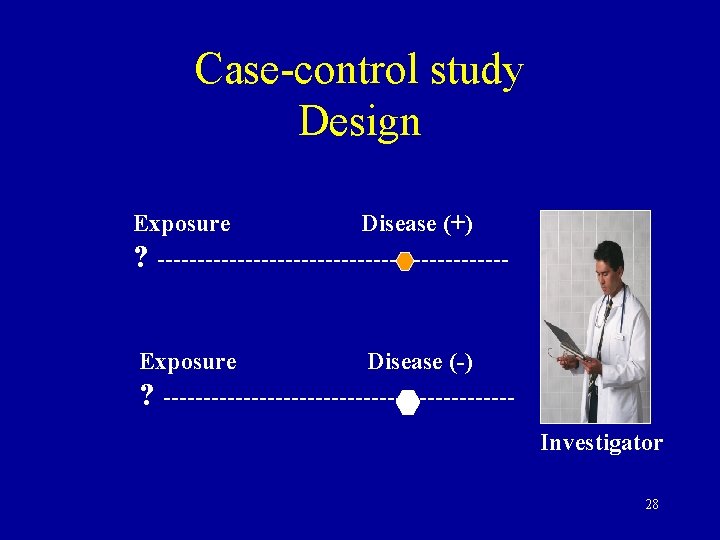
Case-control study Design Exposure Disease (+) ? ----------------------Exposure Disease (-) ? ----------------------Investigator 28

Case control study Exposure rate: a b In case group= ----- x 100 In control group=------ x 100 a+c b+d 29

Case-control study Smoking in case group = Smoking in control group = OR = 70 x 60 /30 x 40 = 3. 7 70 % 40 % 30

Case-control studies Selection of cases 1. From hospital - clinics during a specified period of time Hospital based case-control study 2. All persons with the disease in a defined general population Population based case-control study 31

Case-control studies Selection of controls 1. Hospital controls (people in the same hospital admitted for other conditions or other clinics) 2. Friends, neighbors, relatives of cases 3. General population controls Age, sex, socio-economical status must be similar with case group. 32

Case control studies Advantages • Relatively quick and inexpensive compared with other analytic designs • suitable for long latent periods and rare diseases • can examine multiple etiologic factors for a single disease 33

Case control studies Disadvantages • Some measurements can’t be calculated if it is hospital based. (Morbidity-mortality rates, relative risk, etc. ) • In some situations difficult to establish Exposure Disease relationship • possible BIAS – – records interview different institutions selected people 34

Cohort studies • • Incidence studies Longitudinal studies Follow-up studies Prospective studies 35

Cohort studies (From Exposure to Disease) • Two types of Cohort studies – 1. Prospective Cohort study – 2. Retrospective Cohort study 36

Prospective & Retrospective cohort study • Classify subjects in the basis of presence or absence of exposure • At the beginning of prospective cohort study exposed-unexposed subjects are present, but the disease has not yet occurred. Investigator must follow-up • In a retrospective cohort study both the exposure and disease have already occurred 37

Prospective cohort study EXPOSURE (+) --------DİSEASE ? EXPOSURE (-) --------DİSEASE ? Investigator at beginning of study 38
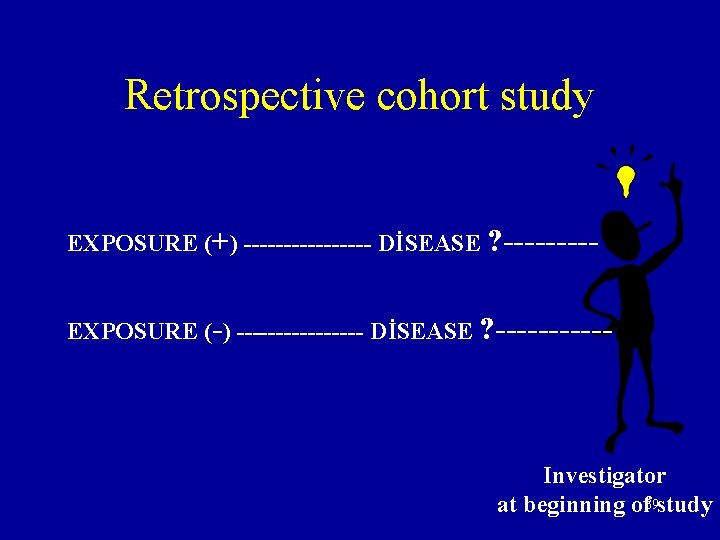
Retrospective cohort study EXPOSURE (+) -------- DİSEASE ? ----EXPOSURE (-) -------- DİSEASE ? ------ Investigator at beginning of 39 study

Cohort study Population People who don’t have disease Exposure (+) Disease (-) Disease (+) a b c Total incidence = ( a+c) / (a+b+c+d) x 100 Incidence exp. (+) = (a) / (a+b) x 100 Incidence exp. (-) = (c) / (c+d) x 100 RR = Incidence exp (+) / Incidence exp (-) Exposure (-) Disease (-) d 40

Prospective cohort study Example: Framingham heart study • 5127 men & women, aged 30 - 59 years • free from coronary heart disease • information collected on demographic variables, medical history, smoking and some clinical & laboratory parameters. • Members of cohort have been followed and examined at regular intervals to monitor the development of cardiovascular disease. 41

Retrospective cohort study Example: Enterline (1965) Asbestos exposure and lung cancer mortality • Mortality from lung cancer among the asbest workers was compared with a group of cotton textile workers. • Indicated increased lung cancer mortality among asbestos workers 42

Cohort study Selection of exposed population • Specific geographic locations • Specific groups (pregnant etc) • Specific exposure population - particular occupations (uranium mine, coal-mine workers etc) 43

Cohort study Selection of comparison group (Non exposed indivudials) • General population • According to the exposure status group can be divided into subgroups (exposure+and exposure -) • Specific comparison group Example: for effect of radiation if radiologists cohort group comparison group may be internalists. Textile workers for asbest workers, 44

Cohort studies Advantages • Exposure - disease relation is very reliable • Can examine multiple effect of a single exposure • Prospective type minimizes BIAS • Incidence rates and relative risk can be calculated 45

Cohort studies Disadvantages • If incidence rate is low and latent period is long it is inefficient - large number, more time are needed • If prospective, can be expensive and time consuming • If retrospective requires the availability of adequate records • Loses to follow-up 46

GUIDELINES FOR CLINICAL RESEARCHERS

STROBE Guideline • The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) Statement: guidelines for reporting observational studies • http: //www. strobe-statement. org/
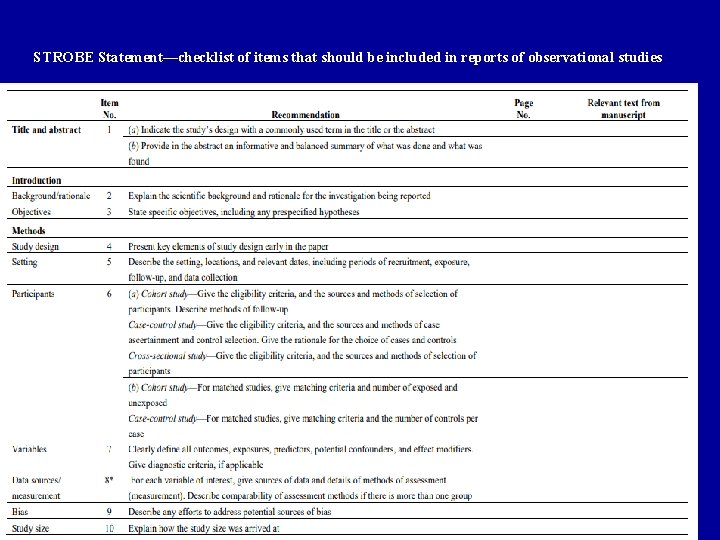
STROBE Statement—checklist of items that should be included in reports of observational studies



The CARE Guidelines • The CARE Guidelines: Consensus-based Clinical Case Reporting Guideline Development • Reference: Gagnier JJ, Kienle G, Altman DA, Moher D, Sox H, Riley D; the CARE Group. The CARE Guidelines: Consensus-based Clinical Case Reporting Guideline Development.
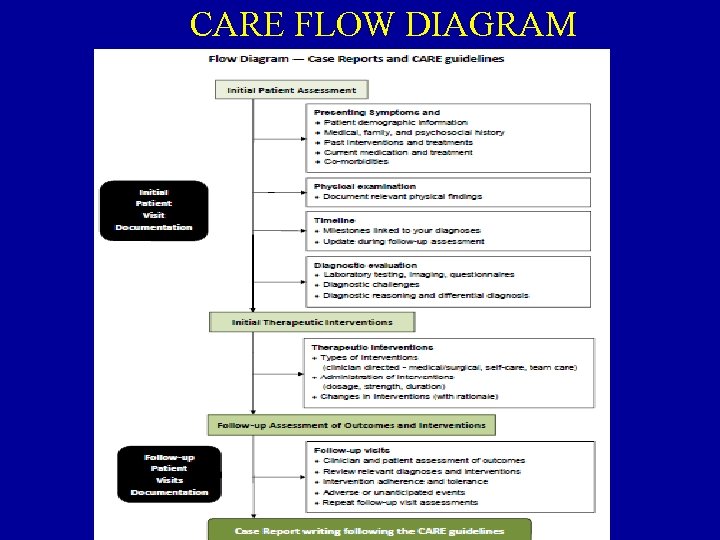
CARE FLOW DIAGRAM

The CARE Guidelines • Title – The diagnosis or intervention of primary focus followed by the words “case report”. • Key Words – 2 to 5 key words that identify diagnoses or interventions in this case report (including "case report"). • Abstract – (structured or unstructured) – Introduction – What is unique about this case and what does it add to the scientific literature? – The patient’s main concerns and important clinical findings. – The primary diagnoses, interventions, and outcomes. – Conclusion – What are one or more “take-away” lessons from this case report?

• • The CARE Guidelines Introduction – Briefly summarizes why this case is unique and may include medical literature references. Patient Information – De-identified patient specific information. – Primary concerns and symptoms of the patient. – Medical, family, and psychosocial history including relevant genetic information. – Relevant past interventions and their outcomes. Clinical Findings – Describe significant physical examination (PE) and important clinical findings. Timeline – Historical and current information from this episode of care organized as a timeline (figure or table).

The CARE Guidelines • Diagnostic Assessment – Diagnostic methods (PE, laboratory testing, imaging, surveys). – Diagnostic challenges. – Diagnosis (including other diagnoses considered). – Prognostic characteristics when applicable. • Therapeutic Intervention – Types of therapeutic intervention (pharmacologic, surgical, preventive). – Administration of therapeutic intervention (dosage, strength, duration). – Changes in therapeutic interventions with explanations.

• • The CARE Guidelines Follow-up and Outcomes – Clinician- and patient-assessed outcomes if available. – Important follow-up diagnostic and other test results. – Intervention adherence and tolerability. (How was this assessed? ) – Adverse and unanticipated events. Discussion – Strengths and limitations in your approach to this case. – Discussion of the relevant medical literature. – The rationale for your conclusions. – The primary “take-away” lessons from this case report (without references) in a one paragraph conclusion. Patient Perspective – The patient should share their perspective on the treatment(s) they received. Informed Consent – The patient should give informed consent. (Provide if requested. )

CONSORT 2010 • CONSORT 2010 Statement: updated guidelines for reporting parallel group randomised trials • http: //www. consort-statement. org/
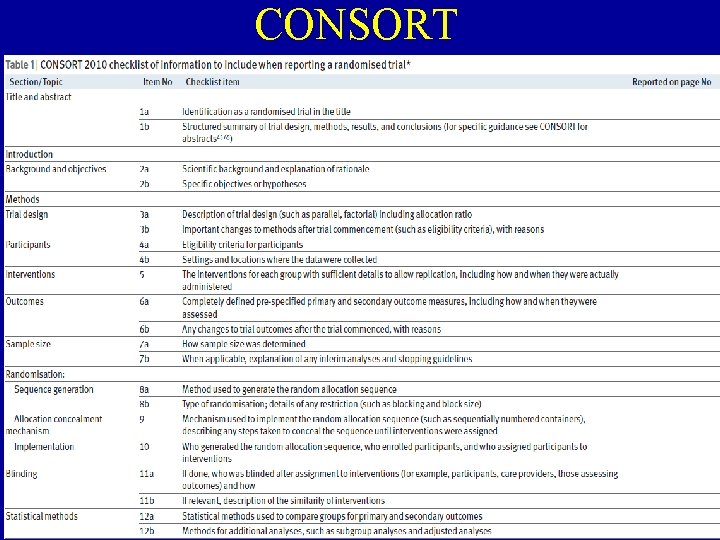
CONSORT
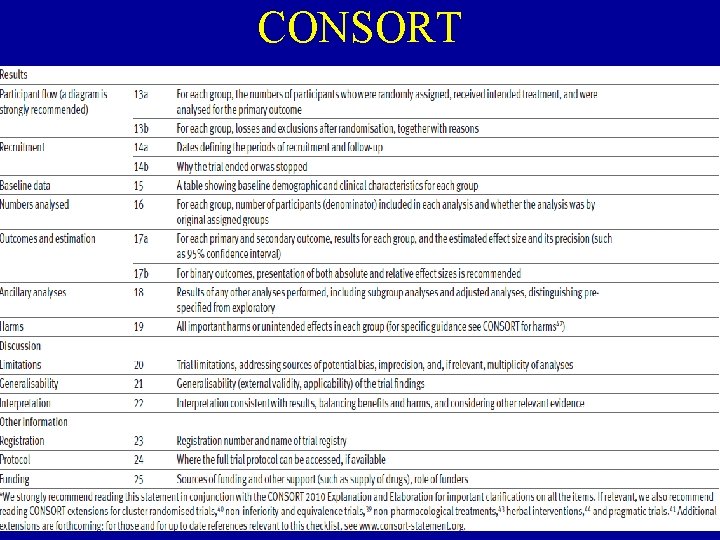
CONSORT

ARRIVE • ARRIVE Guidelines - Animal Research Reporting of In Vivo Experiments • Referans: Kilkenny C, Browne WJ, Cuthill IC, Emerson M, Altman DG. Improving bioscience research reporting: the ARRIVE guidelines for reporting animal research.
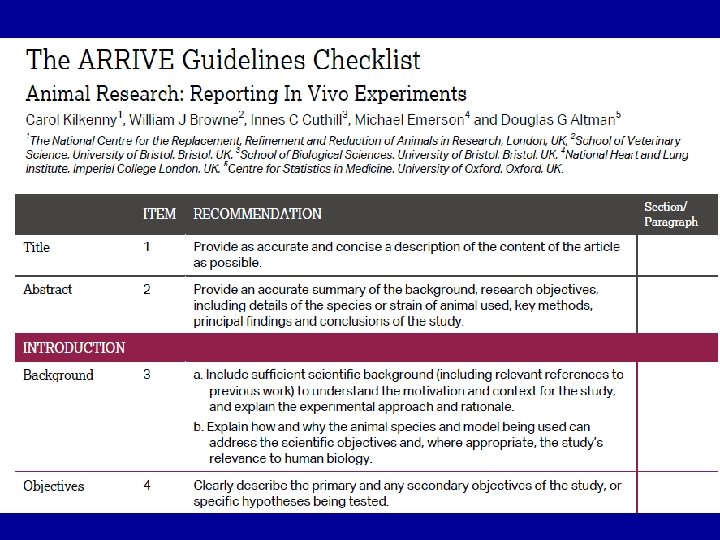

ARRIVE GUIDELINE

ARRIVE
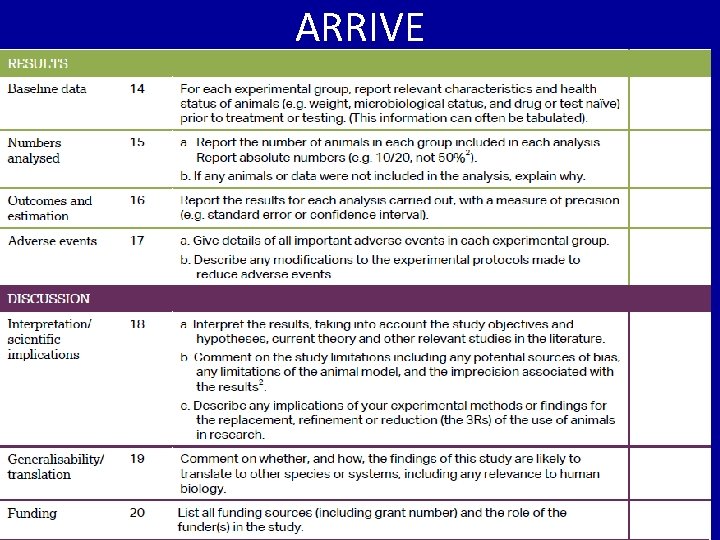
ARRIVE

STARD • Towards complete and accurate reporting of studies of diagnostic accuracy: the STARD initiative. Standards for Reporting of Diagnostic Accuracy • http: //www. stard-statement. org/
STARD
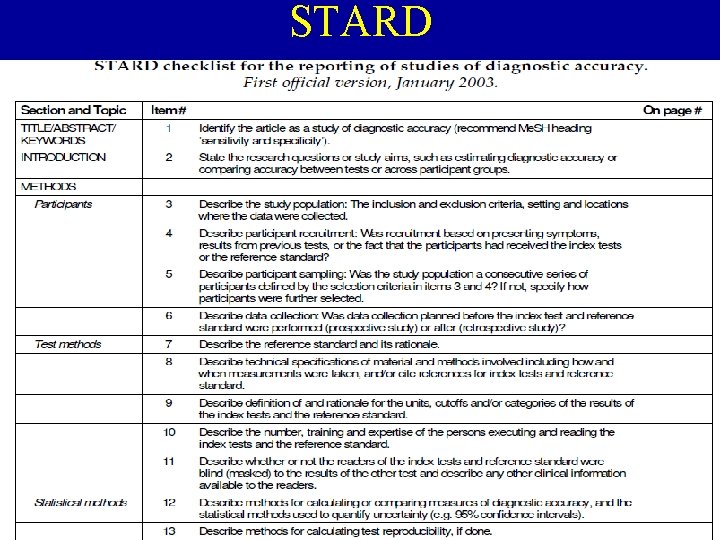
STARD
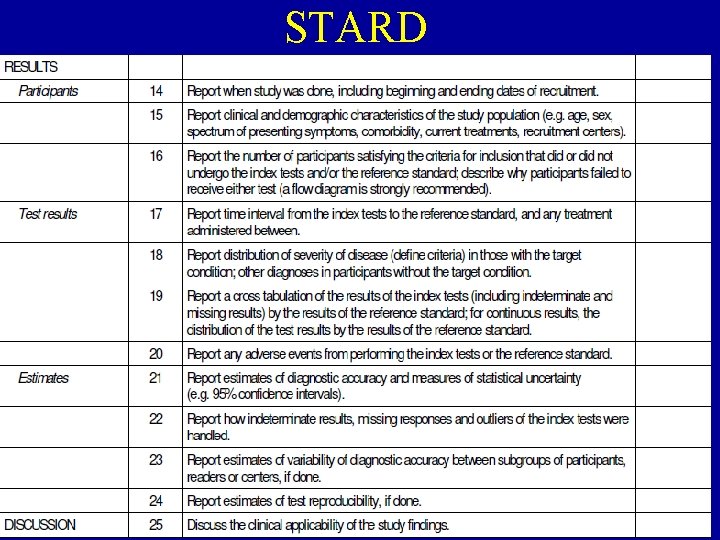
STARD

PRISMA • Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA Statement • http: //www. prisma-statement. org/statement. htm • *The others for systematic review and metaanalysis: AMSTAR, MOOSE ve CRITICAL APPRAISAL CHECKLIST.
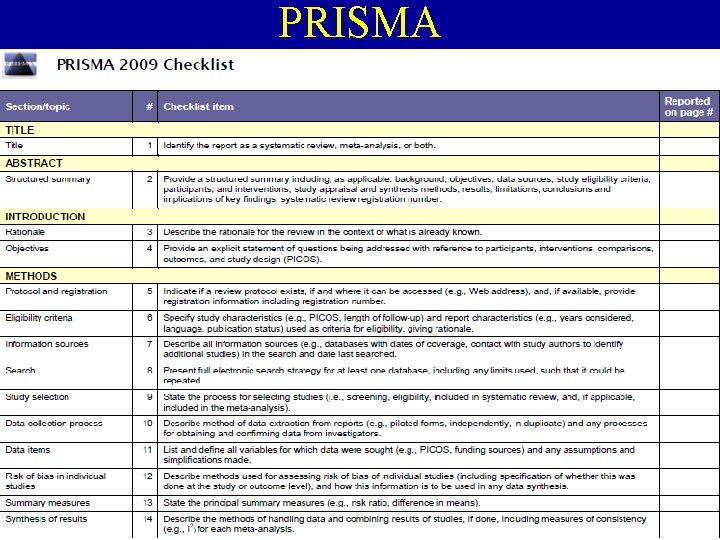
PRISMA
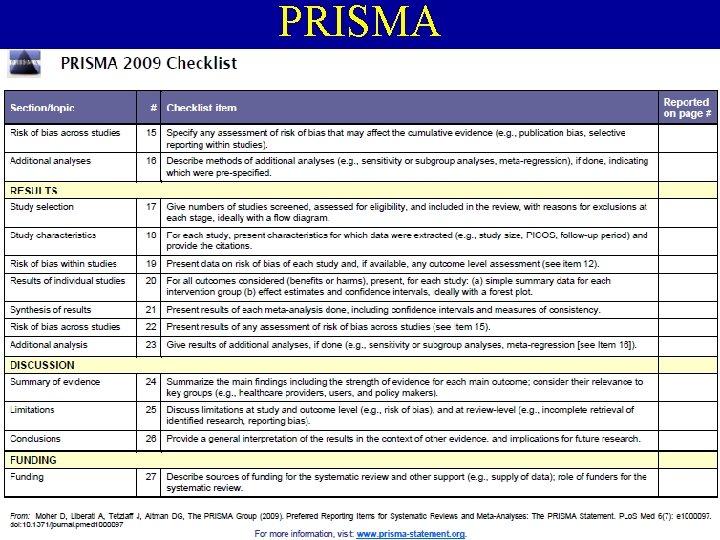
PRISMA

GRRAS • Guidelines for reporting reliability and agreement studies (GRRAS) • Reference: Kottner J, Audigé L, Brorson S, Donner A, Gajeweski BJ, Hróbjartsson A, Robersts C, Shoukri M, Streiner DL. Guidelines for reporting reliability and agreement studies (GRRAS)

Thanks. .

REFERENCES • 1 Mayer D. (2004). Essential Evidence-Based Medicine. Cambridge University Press. • 2. Katz DL. (2001). Clinical Epidemiology &Evidence-Based Medicine. London. Sage Publication. • 3. Appraising Diagnostic Test Studies. http: //wwwusers. york. ac. uk/~mb 55/msc/critappr/diagappr. pdf#page=1&zoom=auto, 0, 842. • 4. Thompson M. Oxford Teaching Tips for Diagnostic Studies CEBM Teaching Course 2012 http: //www. cebm. net/? o=1083. • 5. Lilienfold DE, Stalley PD. (1994). Foundations of Epidemiology. Oxford University Press. • 6. Gordis L Epidemiology. (5. ed. ). Chapter 4: Assessing the Validity and Reliability of Diagnosis and Screening Tests. Pennsylvania. W. B. Saunders Company. • 7. Lilienfold DE, Stalley PD. (1994). Foundations of Epidemiology. Oxford University Press. • 8. Holtz A. (2006). The Medical Science of House. Çev. Karagöz F. (2009). İstanbul. E Yayınları. • 9. Gordis L Epidemiology. (5. ed. ). Chapter 4: Assessing the Validity and Reliability of Diagnosis and Screening Tests. Pennsylvania. W. B. Saunders Company. • 10. Alkafajei A. Improving Clinical Diagnosis. Validity and Reliability. Problem-Based Learning Exercise in Epidemiology. • 11. Zakowski L, Seibert C, Van. Eyck S. Evidence-based Medicine: Answering Questions of Diagnosis. 2004. Clinical Medicine & Research Volume 2, Number 1: 63 -69. • 12. Mathew J. Lecture 5 – Clinical Testing. Understanding the process of diagnosis and clinical testing. EPI-546 Block I. http: //learn. chm. msu. edu/epi/PDF/EPI 546_Lecture_5. pdf • 13. Bernard C. K. Slopes of a Receiver Operating Characteristic Curve and Likelihood Ratios for a Diagnostic Test. American Journal of Epidemiology. 1998, 148: 1127 -32. • 14 A Practical Guide to Biostatistics. Epidemiologyc Tests/19. Chapter four. 76 http: //www. surgicalcriticalcare. net/Statistics/epidemiologic. pdf.
- Slides: 76