Chemical Kinetics Reaction Rates Chemical Kinetics the study










- Slides: 12

Chemical Kinetics (Reaction Rates) Chemical Kinetics: the study of how fast or slow a reaction takes place

Chemical Reactions occur at different rates! There are slow reactions like a copper roof oxidizing (turning green) over a period of several years. When you have a lot of bonds to break the reaction will be slow. Covalent bonds are hard to break.

There are fast reactions: which require the least number of bonds to be broken. Fast reaction involve ions. Precipitation reactions are also fast. Pb 2+ (aq) + 2 I 1 - (aq) --> Pb. I 2 (s) Neutralization reactions are also fast. H 2 SO 4(aq) + 2 KOH (aq) --> 2 H 2 O (l) + K 2 SO 4 (s)

Rate of Chemical Reactions The rate of a chemical reaction is a ratio of the amount of change in a substance to the amount of time required for that change to take place. a Reactant b Product rate = - 1 D [Reactant] = 1 D [Product] a Dt b Dt Example: Ba. Cl 2 + Na 2 SO 4 D means change Ba. SO 4 + 2 Na. Cl The rate is - 1 D[Ba. Cl 2] = -1 D[Na 2 SO 4] = 1 D[Ba. SO 4 ] = 1 D[Na. Cl] 1 Dt 2 Dt

Mathematical Expression of a Rate Law Suppose you are asked to find the mathematical expression of the rate for the following reaction: C 3 H 8(g) + 5 O 2 (g) --> 3 CO 2 (g) + 4 H 2 O (g) rate law= k [C 3 H 8] x [O 2] 5 k is the rate constant *note that only the reactants are in the expression*

Reaction Process Most chemical reactions take place in a series of steps called reaction mechanisms. Example: Step 1: Cr(CO)6 --> Cr(CO) 5 +CO slow Step 2: Cr(CO)5 + NH 3 --> Cr(CO)5 NH 3 fast Rx. N: Cr(CO)6 + NH 3 --> Cr(CO)5 NH 3 +CO The slowest step determines the rate of the reaction. It is called the rate determining step.

Collision Theory and Activation Energy

The collision theory states that for a product to be made the reactants must collide with enough energy and with the right geometry. If the molecules collide with enough energy, then the atoms will rearrange and form a new product. This is called an effective collision.

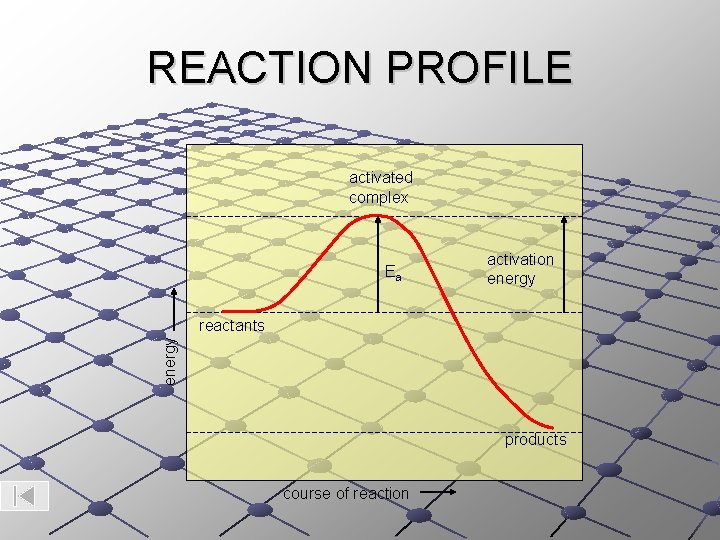
REACTION PROFILE activated complex Ea activation energy reactants products course of reaction

ACTIVATED COMPLEX: is the temporary, unstable arrangement of particles present at the highest point in a chemical reaction profile. ACTIVATION ENERGY (Ea): is the minimum energy with which particles must collide in order for the collision to be effective (resulting in the formation of a NEW substance)

Combustion Reactions A combustion reaction is exothermic. A combustion reaction is a reaction between a fuel and oxygen. If there is hydrogen within the molecules of the fuel, then water will be produced C 8 H 18 + O 2 what are the products? 2 C 8 H 18 + 25 O 2 16 CO 2 + 18 H 2 O

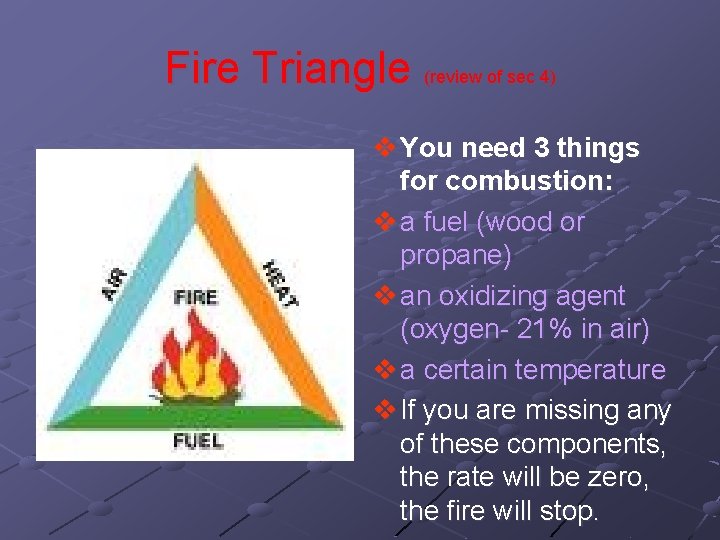
Fire Triangle (review of sec 4) v You need 3 things for combustion: v a fuel (wood or propane) v an oxidizing agent (oxygen- 21% in air) v a certain temperature v If you are missing any of these components, the rate will be zero, the fire will stop.