Chemical Kinetics Chapter 16 Kinetics Reaction Rates Factors











![Rate Laws k = m, n = rate constant order rate = k[NO 2]n Rate Laws k = m, n = rate constant order rate = k[NO 2]n](https://slidetodoc.com/presentation_image_h/e280e39e9024ff90347689401918cc80/image-24.jpg)










![Half-life, first order reactions Integrated law: Half-life: Half of initial reacted [A]t = ½[A]0 Half-life, first order reactions Integrated law: Half-life: Half of initial reacted [A]t = ½[A]0](https://slidetodoc.com/presentation_image_h/e280e39e9024ff90347689401918cc80/image-36.jpg)
![Half-life, second order reactions Integrated law: Half-life: Half of initial reacted [A]t = ½[A]0 Half-life, second order reactions Integrated law: Half-life: Half of initial reacted [A]t = ½[A]0](https://slidetodoc.com/presentation_image_h/e280e39e9024ff90347689401918cc80/image-40.jpg)



![First order ln[A]0 slope = -k ln[A] Plot: ln[A] vs. time First order ln[A]0 slope = -k ln[A] Plot: ln[A] vs. time](https://slidetodoc.com/presentation_image_h/e280e39e9024ff90347689401918cc80/image-44.jpg)
![Second order slope = k 1 [A] Plot: 1 vs. time [A] 1 [A]o Second order slope = k 1 [A] Plot: 1 vs. time [A] 1 [A]o](https://slidetodoc.com/presentation_image_h/e280e39e9024ff90347689401918cc80/image-45.jpg)
![Zero order [A]0 slope = -k [A] Plot: [A] vs. time Zero order [A]0 slope = -k [A] Plot: [A] vs. time](https://slidetodoc.com/presentation_image_h/e280e39e9024ff90347689401918cc80/image-46.jpg)













- Slides: 86

Chemical Kinetics Chapter 16

Kinetics Reaction Rates Factors affecting rate Quantitative rate expressions Determination Factors Models for Rates Reaction Mechanisms Effects of catalysts

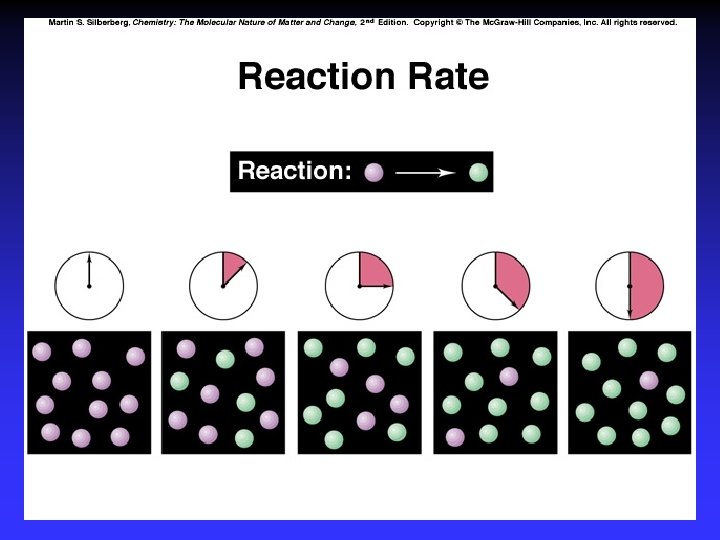
Rates Change in concentration of a reactant or product per unit time



Factors affecting rates Nature of the reactants State of subdivision/surface area Concentration Temperature Catalysts

Reactants Complexity Bond strengths Etc.








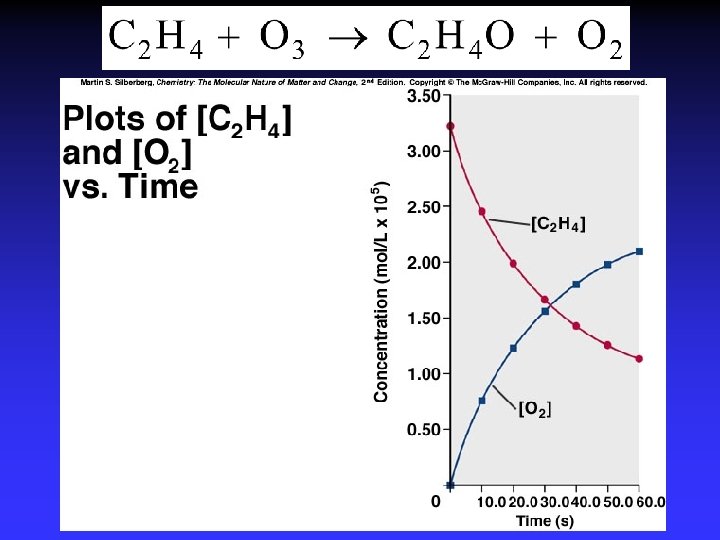
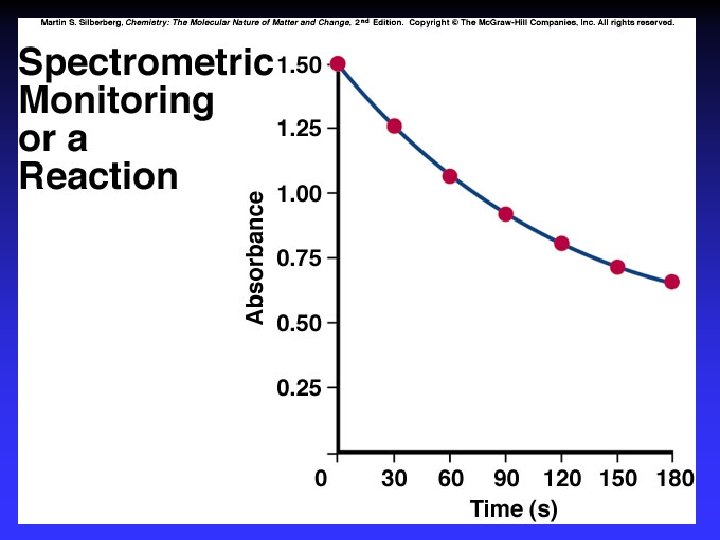
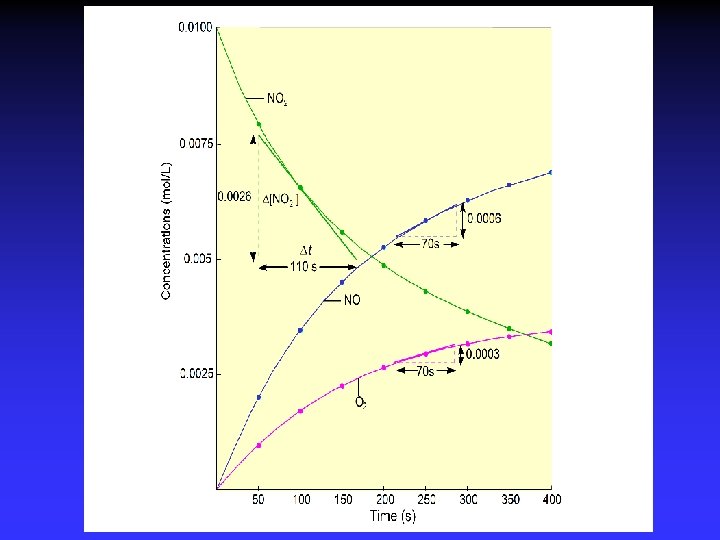
Concentrations as functions of time

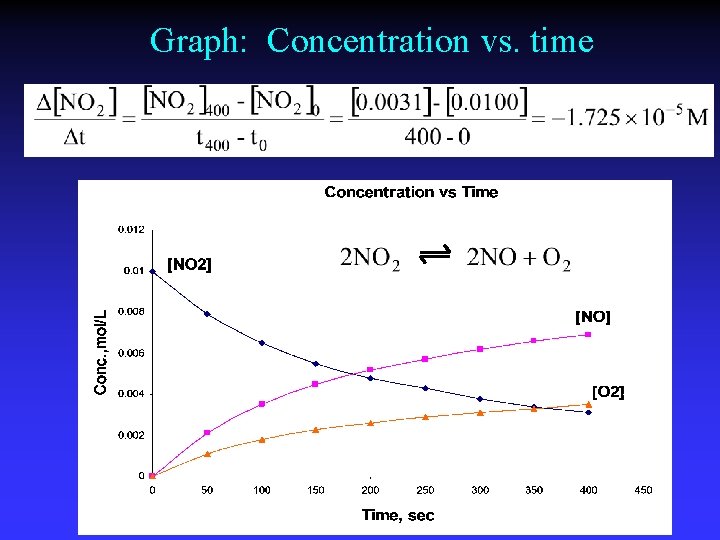
Graph: Concentration vs. time

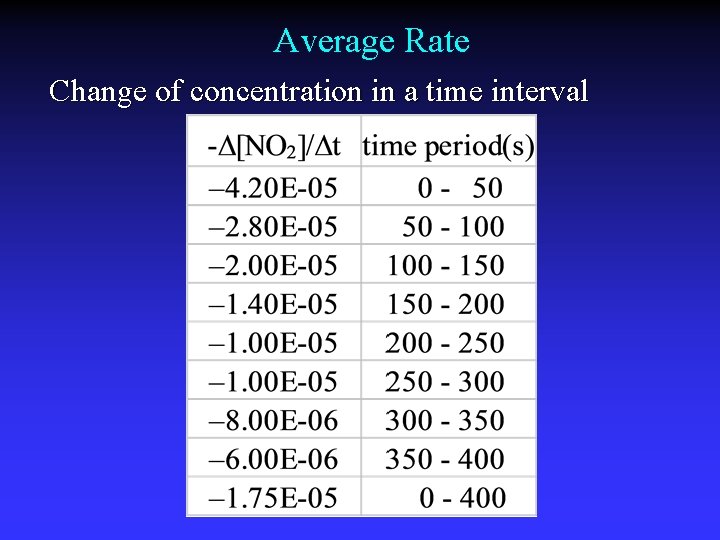
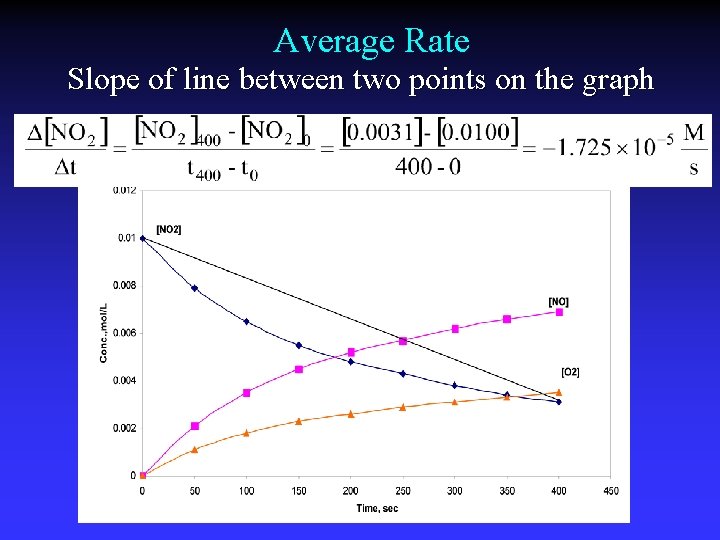
Average Rate Change of concentration in a time interval

Average Rate Slope of line between two points on the graph

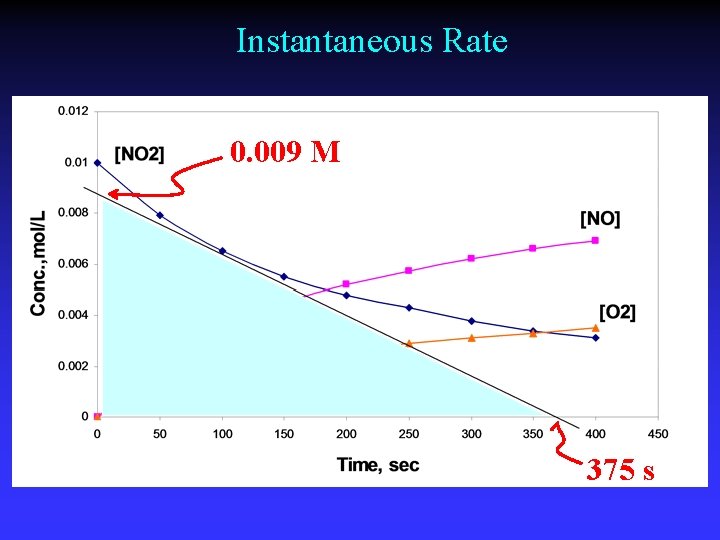
Instantaneous rate Slope of tangent line at a point on the graph

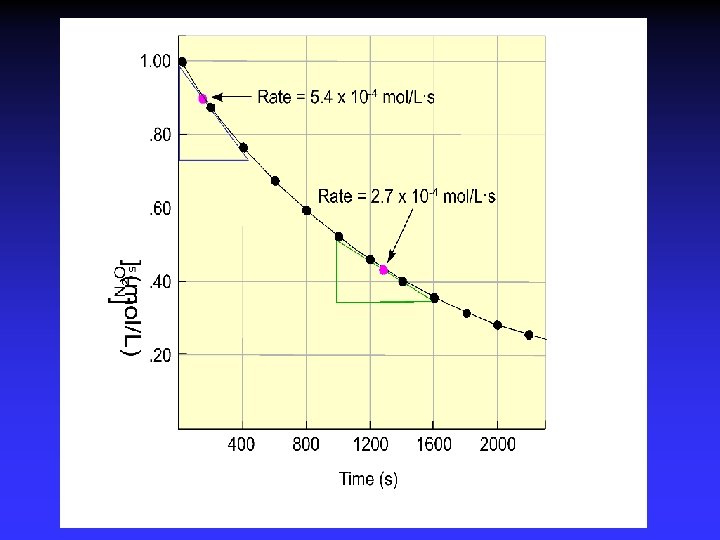
Instantaneous Rate 0. 009 M 375 s


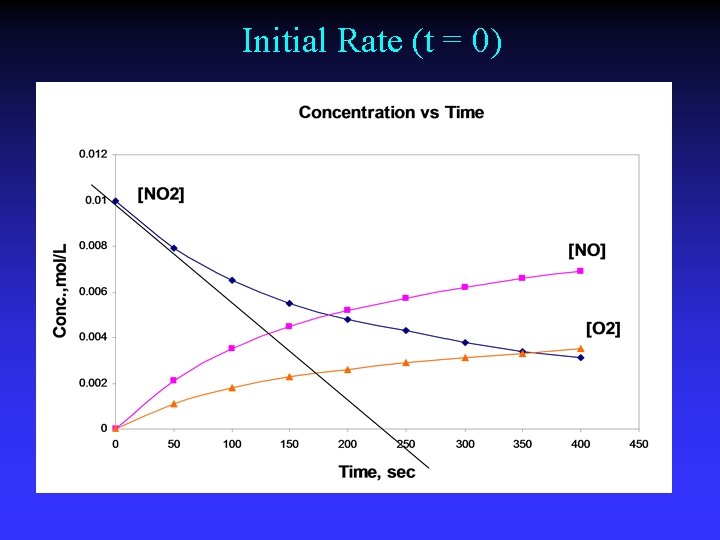
Initial Rate (t = 0)

Initial rate Slope of tangent line at time 0 (y intercept)
![Rate Laws k m n rate constant order rate kNO 2n Rate Laws k = m, n = rate constant order rate = k[NO 2]n](https://slidetodoc.com/presentation_image_h/e280e39e9024ff90347689401918cc80/image-24.jpg)
Rate Laws k = m, n = rate constant order rate = k[NO 2]n

Introduction to Rate Laws Reversible chemical reactions Forward: Backward: Equilibrium:

Introduction Dominant Reaction: Rate Law: k, k’: n: specific rate constant order of reactant can be zero, fractional, or negative

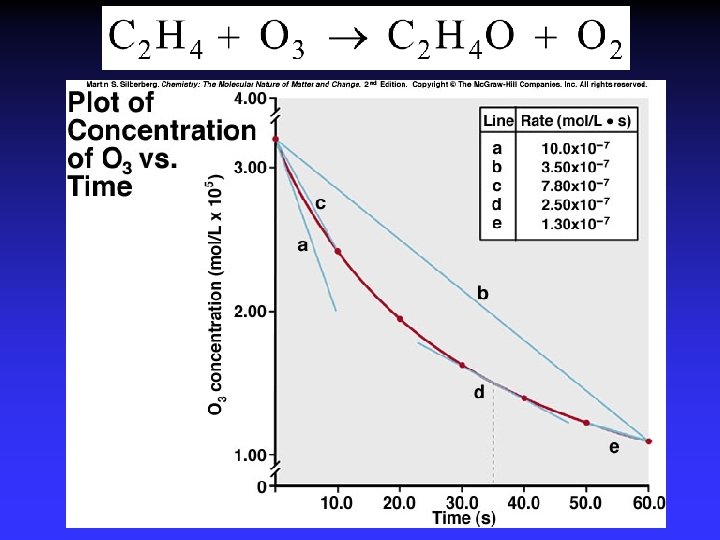
Method of Initial Rates Unknown: k, m, n Initial rate: instantaneous rate just after reaction is initiated

Initial Rates, NO 2 decomposition

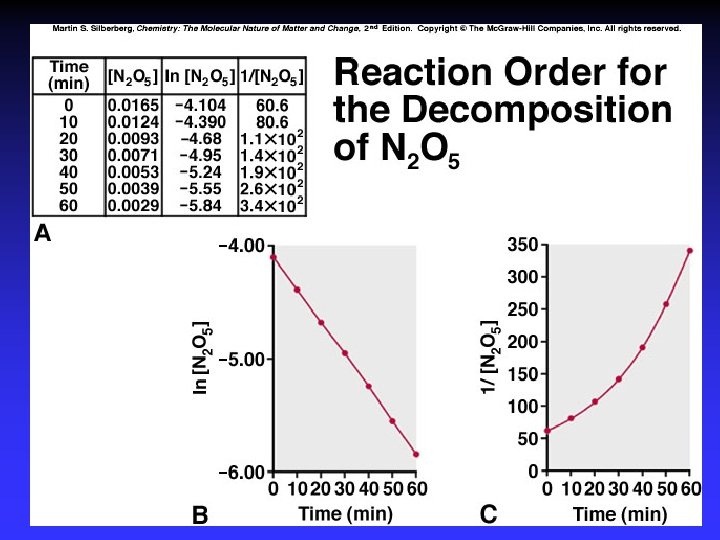
Order of Reaction General: Substituting: Solution:

Rate constant Rate 1 7. 1 x 10 -5 M s-1 k Rate 2 2. 8 x 10 -4 M s-1 k = = -k[0. 01 M]2 0. 71 M-1 s-1 = = -k[0. 02 M]2 0. 70 M-1 s-1

You try

O 2 + 2 NO 2 NO 2

Overall Order Sum: 1 = + 6 2 Overall order of reaction: + 6 3

Types Differential: Rate dependence on concentration Integrated: Concentration dependence on time

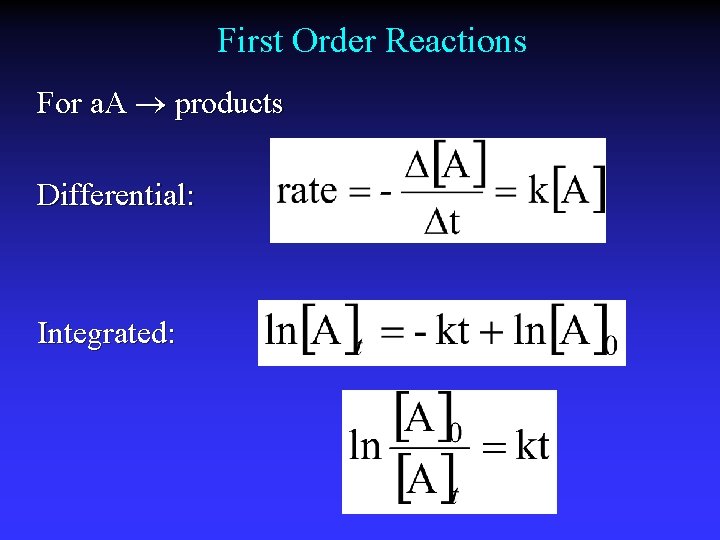
First Order Reactions For a. A products Differential: Integrated:
![Halflife first order reactions Integrated law Halflife Half of initial reacted At ½A0 Half-life, first order reactions Integrated law: Half-life: Half of initial reacted [A]t = ½[A]0](https://slidetodoc.com/presentation_image_h/e280e39e9024ff90347689401918cc80/image-36.jpg)
Half-life, first order reactions Integrated law: Half-life: Half of initial reacted [A]t = ½[A]0 Independent of [A]0



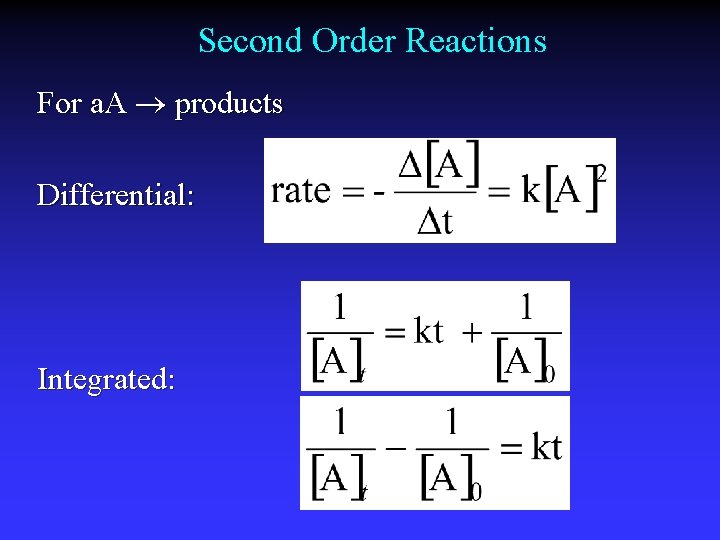
Second Order Reactions For a. A products Differential: Integrated:
![Halflife second order reactions Integrated law Halflife Half of initial reacted At ½A0 Half-life, second order reactions Integrated law: Half-life: Half of initial reacted [A]t = ½[A]0](https://slidetodoc.com/presentation_image_h/e280e39e9024ff90347689401918cc80/image-40.jpg)
Half-life, second order reactions Integrated law: Half-life: Half of initial reacted [A]t = ½[A]0 Inversely proportional to [A]0

Zero Order Reactions For a. A products Differential: Integrated:


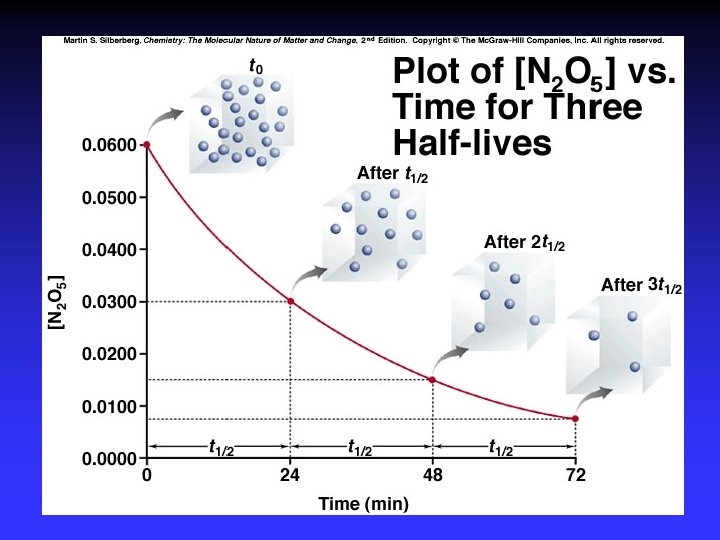
Graphical Method First order Second order Zero order Straight line
![First order lnA0 slope k lnA Plot lnA vs time First order ln[A]0 slope = -k ln[A] Plot: ln[A] vs. time](https://slidetodoc.com/presentation_image_h/e280e39e9024ff90347689401918cc80/image-44.jpg)
First order ln[A]0 slope = -k ln[A] Plot: ln[A] vs. time
![Second order slope k 1 A Plot 1 vs time A 1 Ao Second order slope = k 1 [A] Plot: 1 vs. time [A] 1 [A]o](https://slidetodoc.com/presentation_image_h/e280e39e9024ff90347689401918cc80/image-45.jpg)
Second order slope = k 1 [A] Plot: 1 vs. time [A] 1 [A]o time
![Zero order A0 slope k A Plot A vs time Zero order [A]0 slope = -k [A] Plot: [A] vs. time](https://slidetodoc.com/presentation_image_h/e280e39e9024ff90347689401918cc80/image-46.jpg)
Zero order [A]0 slope = -k [A] Plot: [A] vs. time



Summary Conditions set so dominant forward reaction Differential Rate Laws rate as a function of concentration method of initial rates Integrated Rate Laws concentration as a function of time graphical method Experimental data collection Rate law types can be interconverted

Reaction Mechanism Chemical equation: Summary Mechanism: Series of elementary steps Elementary Steps: Reactions with rate laws from molecularity Molecularity: Number of species that must collide to produce reaction


Reaction Mechanism Proposed elementary steps must satisfy conditions: — reasonable reactions — sum of steps = overall balanced reaction — mechanism rate law = experimental rate law

Intermediates appear in steps — produced in one step — used in subsequent — not in overall equation —

Rate-determining step In a multi-step process: SLOWEST step Determines overall reaction rate “Bottleneck”

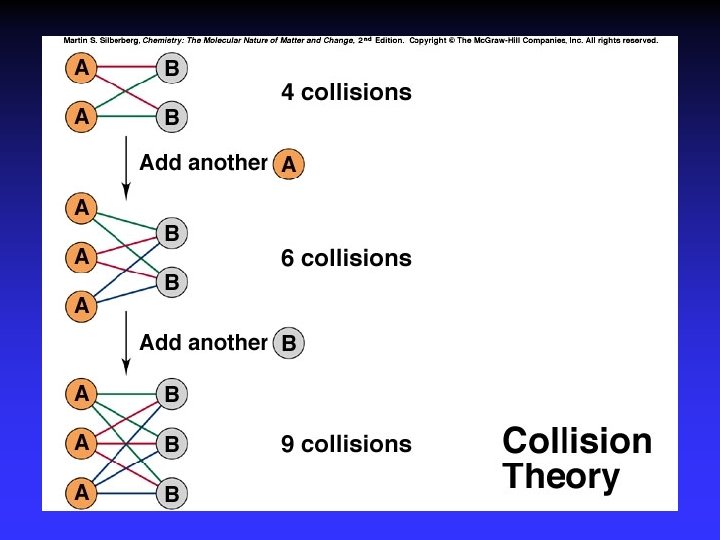
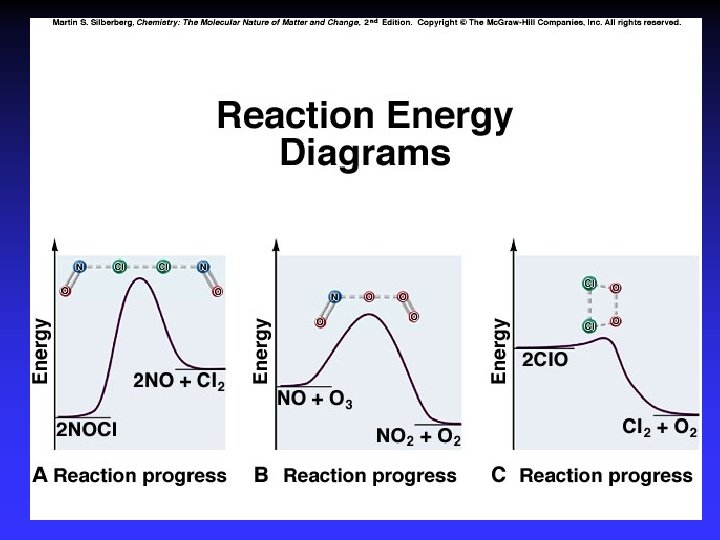
Model for Kinetics Collision Theory rate determined by particle collisions collision frequency and energy Transition State Theory how reactants convert to products

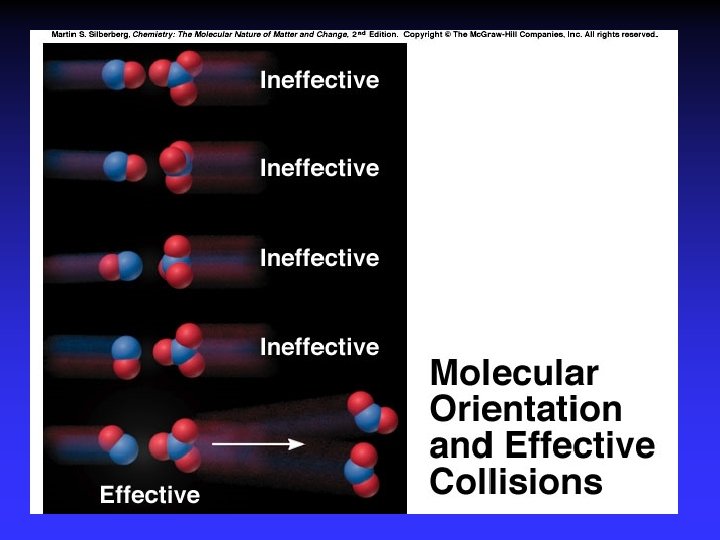
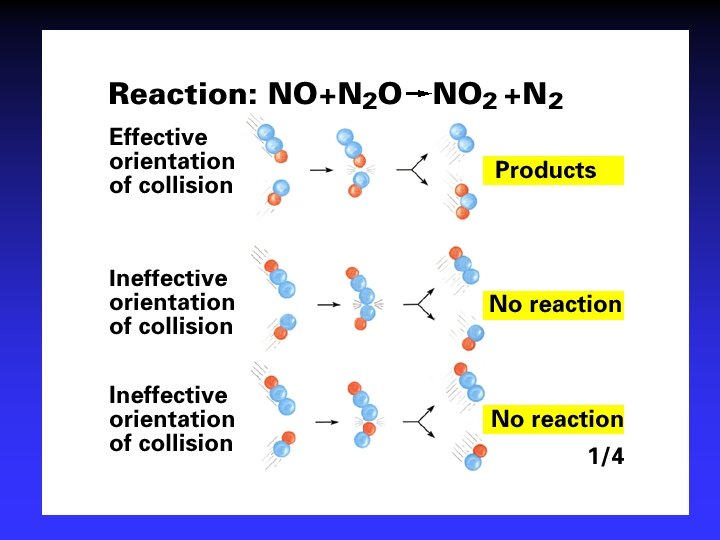
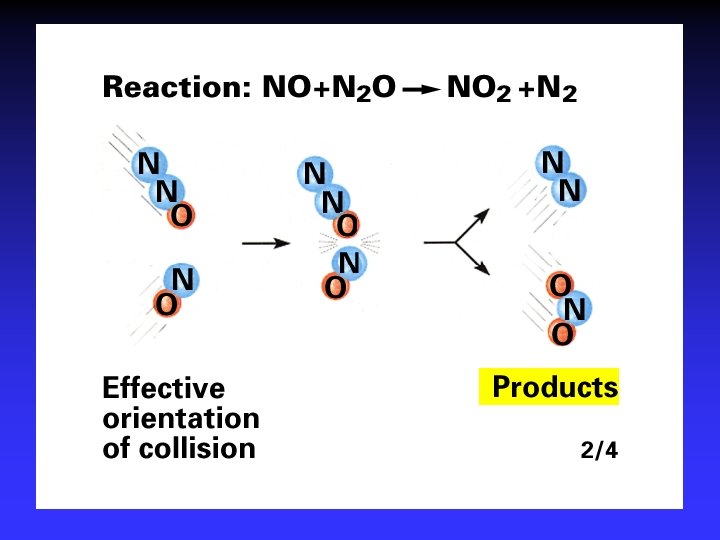
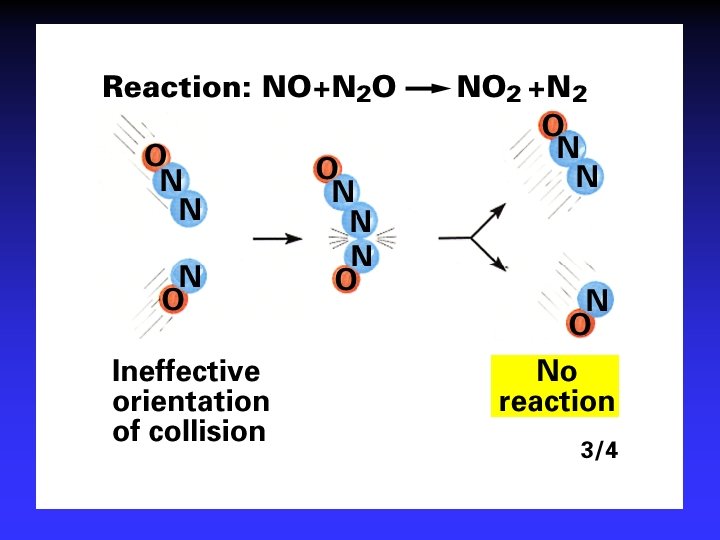
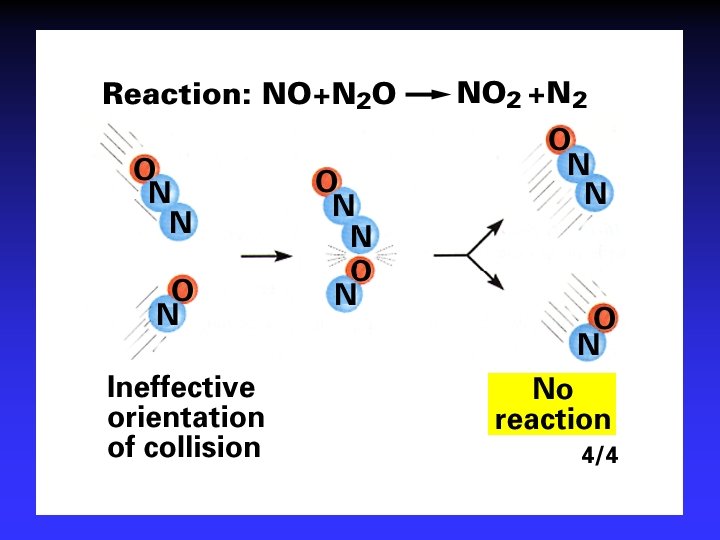
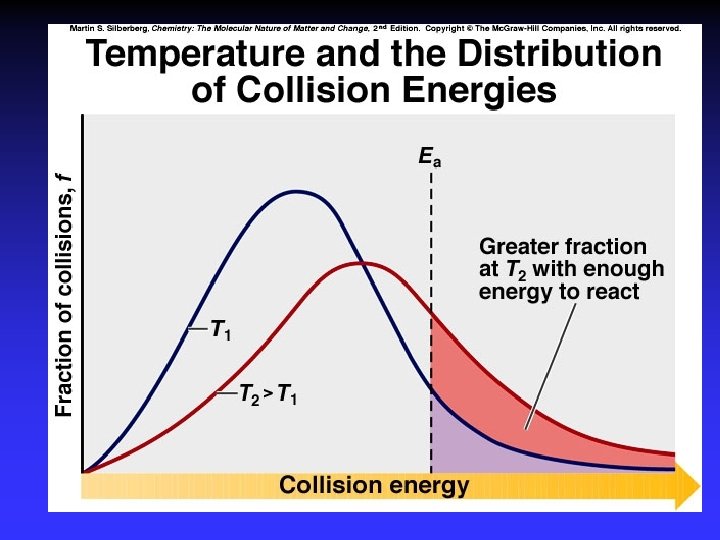
Collision Theory (Bimolecular Collsions) Z: fa : P: no. of bimolecular collisions per second fraction with Ea fraction with correct orientation Ea: activation energy







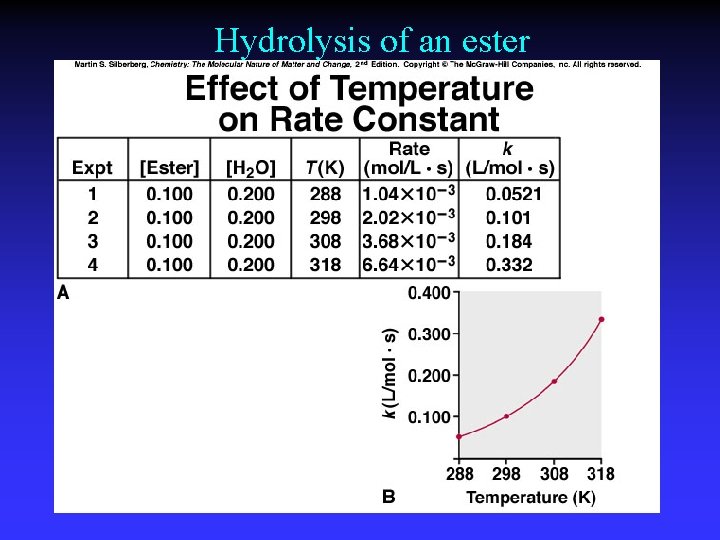
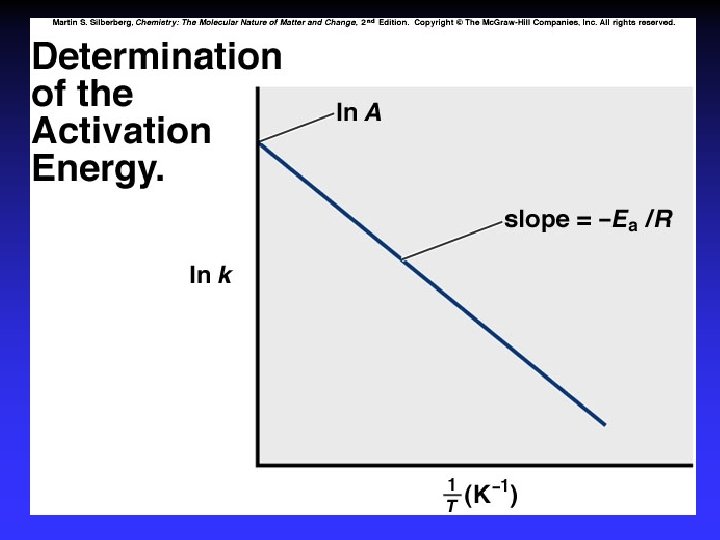
Arrhenius Equation k: E a: T: R: A: rate constant activation energy (minimum required) absolute temperature universal gas constant orientation factor Energy & orientation requirements for reaction

Hydrolysis of an ester




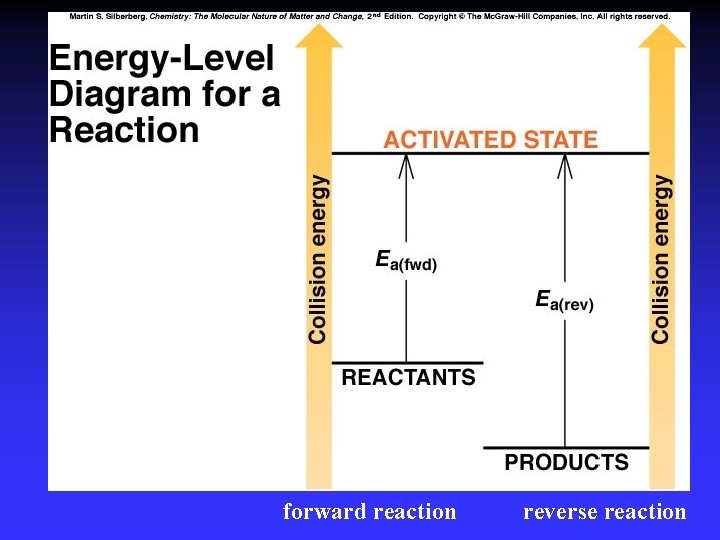
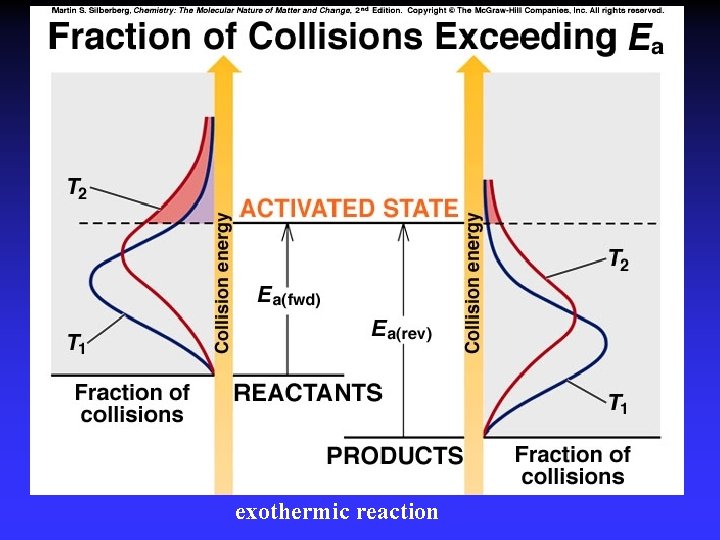
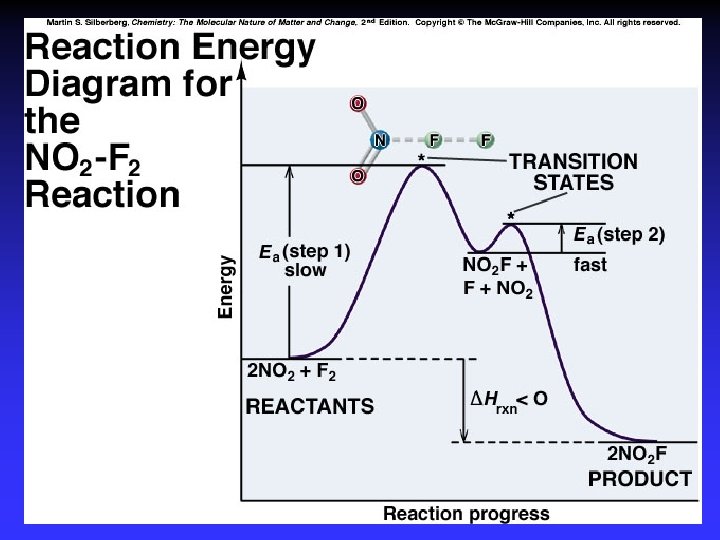
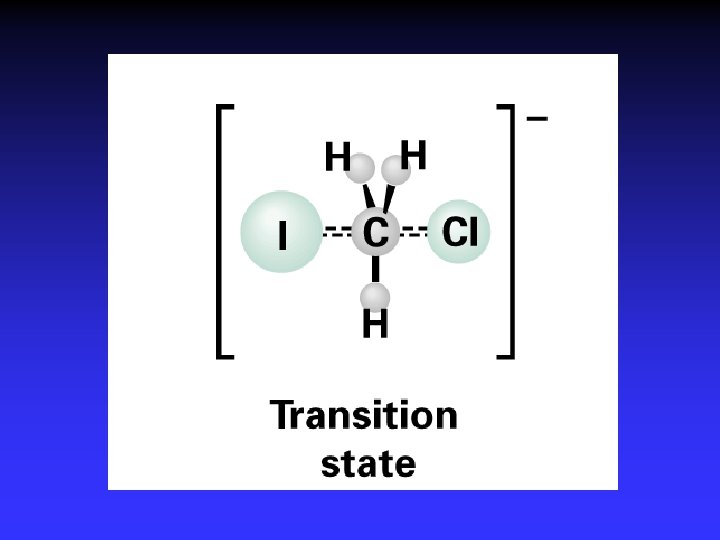
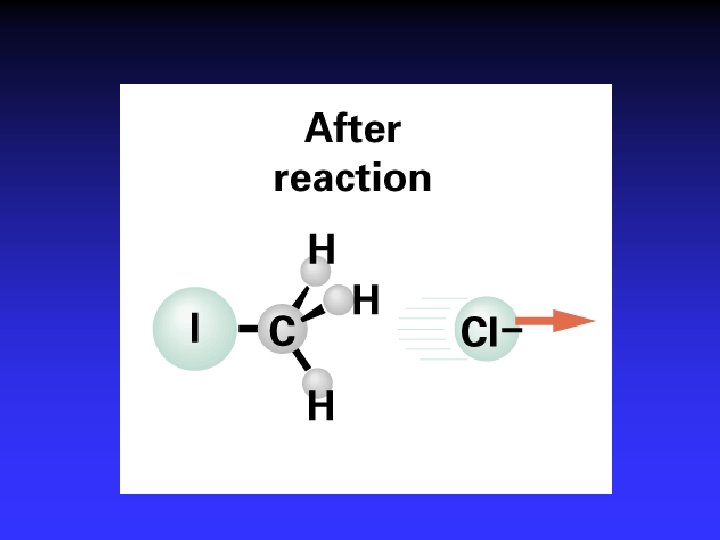
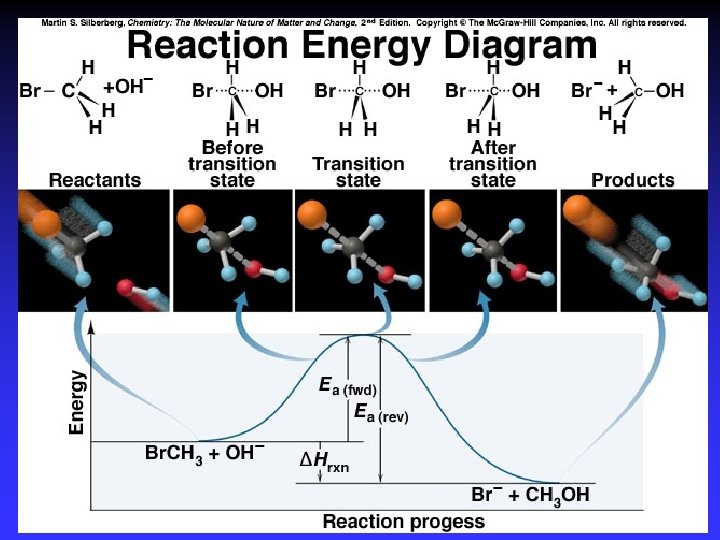
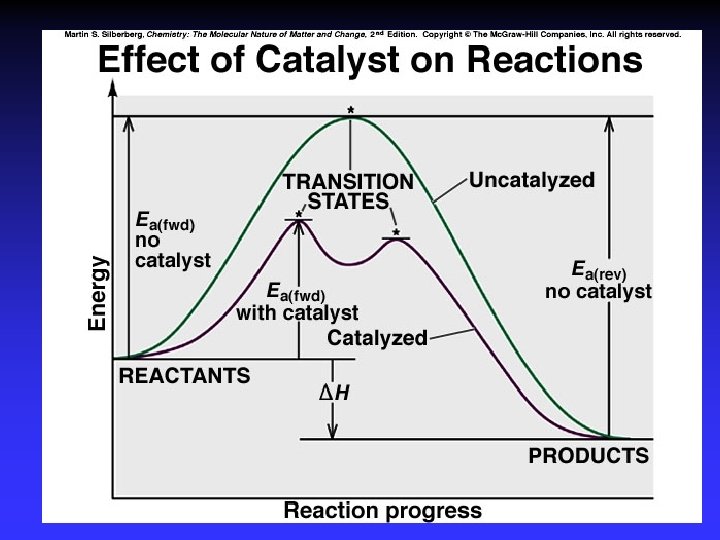
Transition State Theory Ea and internal energy: Bonds breaking and forming Atoms rearranging “Transition State” Unstable intermediate At point of highest energy

forward reaction reverse reaction

exothermic reaction


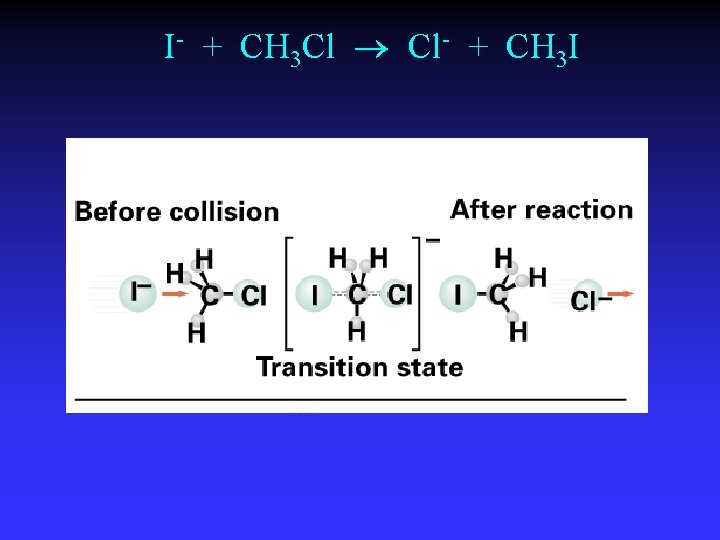
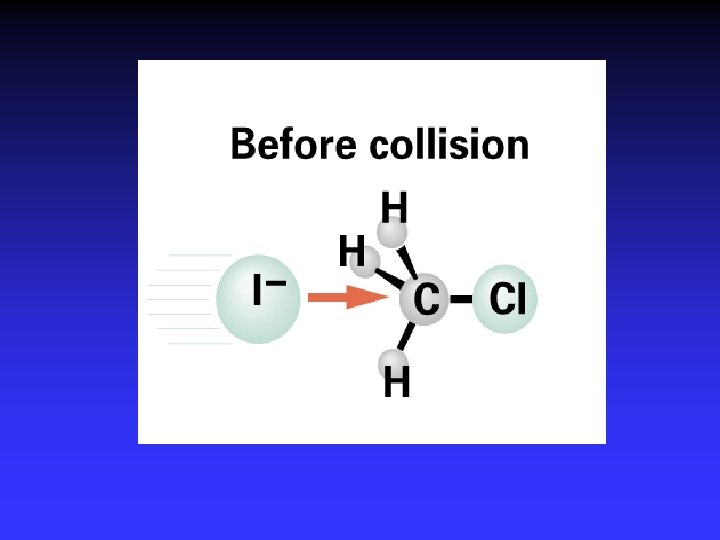
I- + CH 3 Cl Cl- + CH 3 I






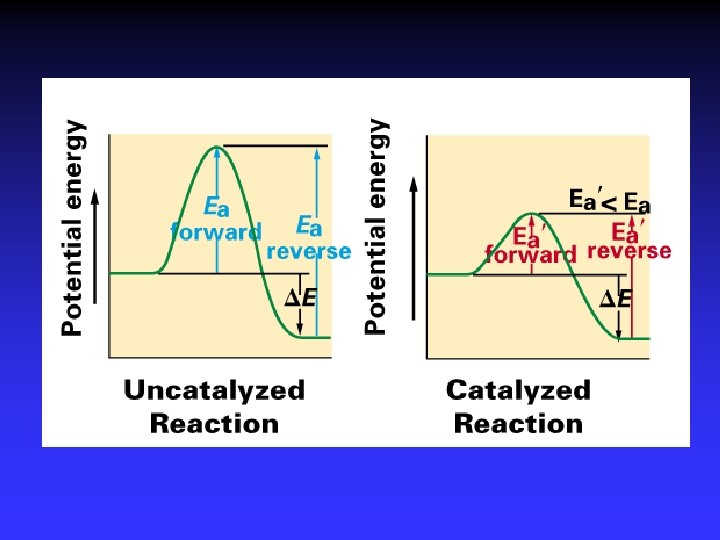
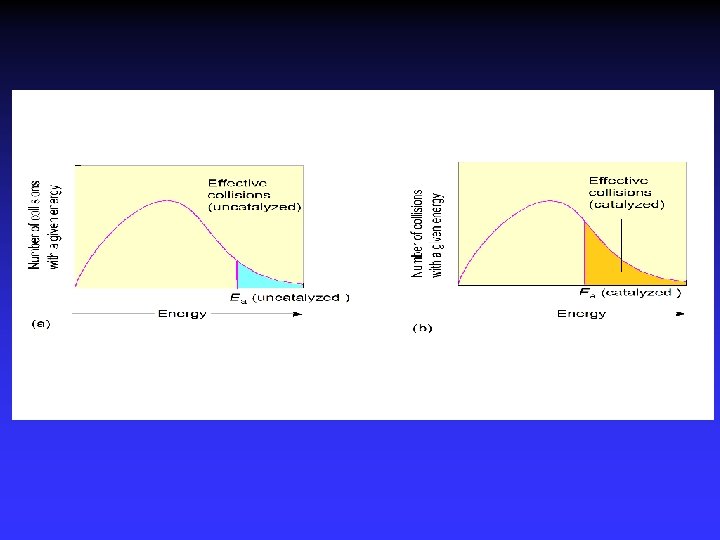
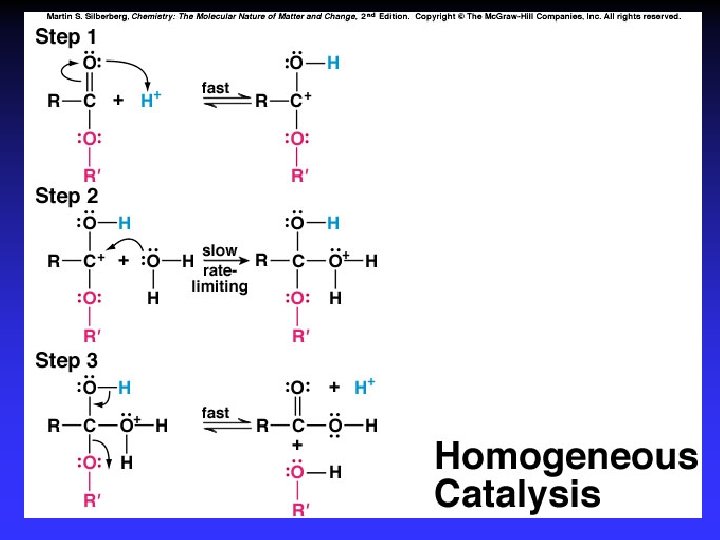
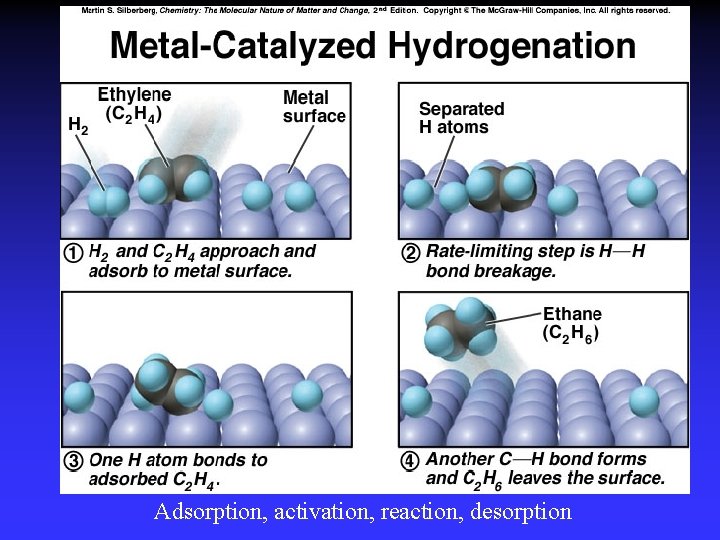
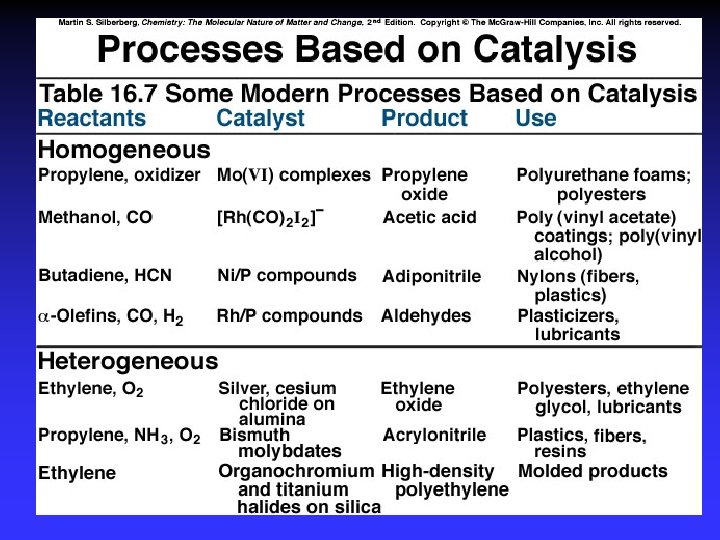
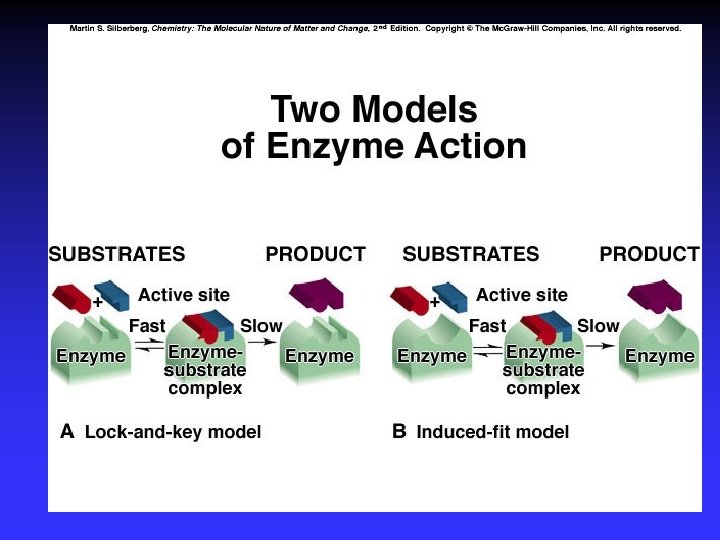
Catalysts Speed reaction Are not consumed Alternative pathway for reaction with lower Ea Types Homogeneous Heterogeneous Enzymes are biological catalysts




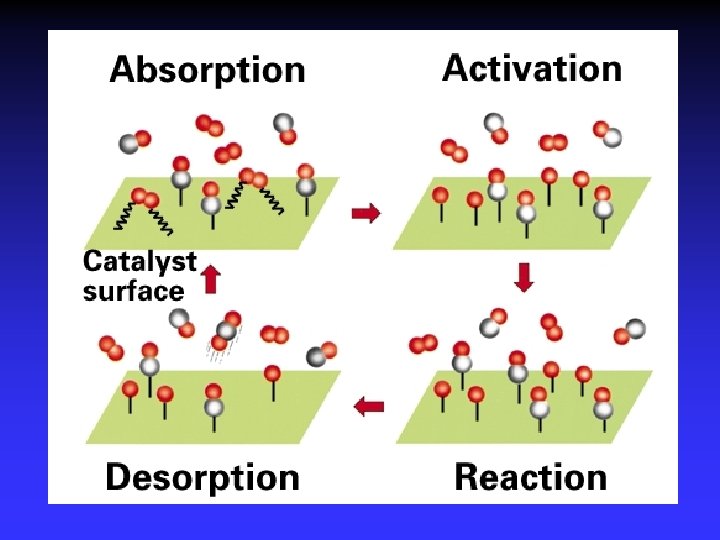
Adsorption, activation, reaction, desorption



