University of Ioannina INVESTIGATING THE PROPERTIES OF IRON









- Slides: 21

University of Ioannina INVESTIGATING THE PROPERTIES OF IRON IN INORGANIC POLYMERS WITH 57 Fe MÖSSBAUER SPECTROSCOPY Alexios P. Douvalis Physics Department University of Ioannina-Greece 5 th International Slag Valorisation Symposium

University of Ioannina Outline q Slags and Inorganic Polymers (IP): why Spectroscopy. 57 Fe Mössbauer § Brief introduction and basic parameters. q The state of iron and its properties in synthetic Slags made of binary Fe. Ox-Si. O 2 (Bi) and ternary Fe. Ox-Ca. O-Si. O 2 (Te) oxide systems and the corresponding IPs, as seen by 57 Fe Mössbauer Spectroscopy. § Estimating the valence, atomic environment and structure. § Identifying the iron-bearing phases. § Suggesting paths to explain the role of iron in the IP formation. q Following the evolution of the 57 Fe Mössbauer spectra at different reaction stages of the Slag with the activating Si. O 2/Na 2 O-H 2 O/Na 2 O solution. § Monitoring the oxidation of Fe 2+ to Fe 3+. 5 th International Slag Valorisation Symposium

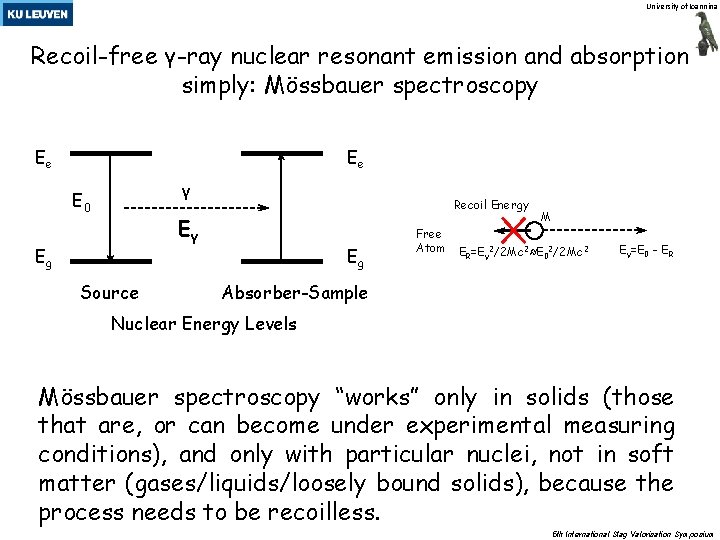
University of Ioannina Recoil-free γ-ray nuclear resonant emission and absorption simply: Mössbauer spectroscopy Ee Ee γ Ε 0 Recoil Energy Eγ Εg Source Εg Free Atom M ER=Eγ 2/2 Mc 2≈E 02/2 Mc 2 Eγ=E 0 - ΕR Absorber-Sample Nuclear Energy Levels Mössbauer spectroscopy “works” only in solids (those that are, or can become under experimental measuring conditions), and only with particular nuclei, not in soft matter (gases/liquids/loosely bound solids), because the process needs to be recoilless. 5 th International Slag Valorisation Symposium

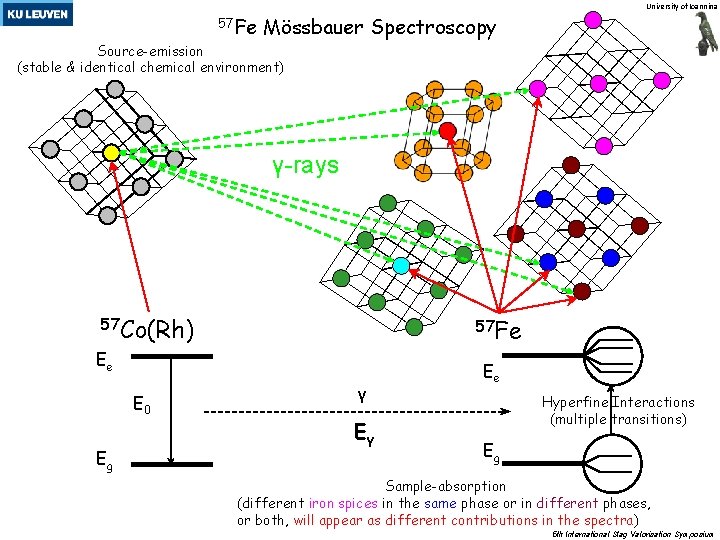
University of Ioannina 57 Fe Mössbauer Spectroscopy Source-emission (stable & identical chemical environment) γ-rays 57 Co(Rh) 57 Fe Ee Ε 0 Εg γ Eγ Ee Hyperfine Interactions (multiple transitions) Εg Sample-absorption (different iron spices in the same phase or in different phases, or both, will appear as different contributions in the spectra) 5 th International Slag Valorisation Symposium

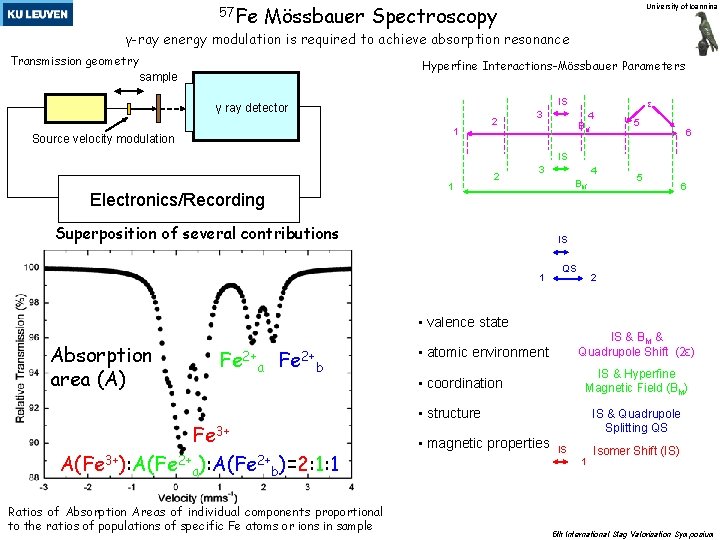
57 Fe Mössbauer Spectroscopy University of Ioannina γ-ray energy modulation is required to achieve absorption resonance Transmission geometry Hyperfine Interactions-Mössbauer Parameters sample IS γ ray detector 1 Source velocity modulation 2 ε 3 4 Bhf 5 6 IS Electronics/Recording 1 2 3 4 Bhf Superposition of several contributions 5 6 IS 1 QS 2 • valence state Absorption area (A) Fe 2+a Fe 2+b Fe 3+ Α(Fe 3+): A(Fe 2+a): A(Fe 2+b)=2: 1: 1 Ratios of Absorption Areas of individual components proportional to the ratios of populations of specific Fe atoms or ions in sample IS & Bhf & Quadrupole Shift (2ε) • atomic environment IS & Hyperfine Magnetic Field (Bhf) • coordination • structure • magnetic properties IS & Quadrupole Splitting QS IS 1 Isomer Shift (IS) 5 th International Slag Valorisation Symposium

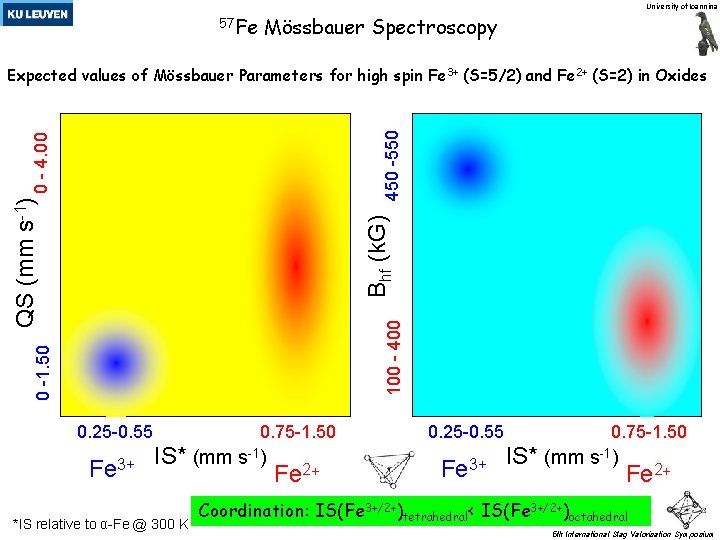
University of Ioannina 57 Fe Mössbauer Spectroscopy 0 -1. 50 100 - 400 Bhf (k. G) QS (mm s-1) 0 - 4. 00 450 -550 Expected values of Mössbauer Parameters for high spin Fe 3+ (S=5/2) and Fe 2+ (S=2) in Oxides 0. 25 -0. 55 Fe 3+ 0. 75 -1. 50 IS* (mm s-1) *IS relative to α-Fe @ 300 K Fe 2+ 0. 25 -0. 55 Fe 3+ 0. 75 -1. 50 IS* (mm s-1) Fe 2+ Coordination: IS(Fe 3+/2+)tetrahedral< IS(Fe 3+/2+)octahedral 5 th International Slag Valorisation Symposium

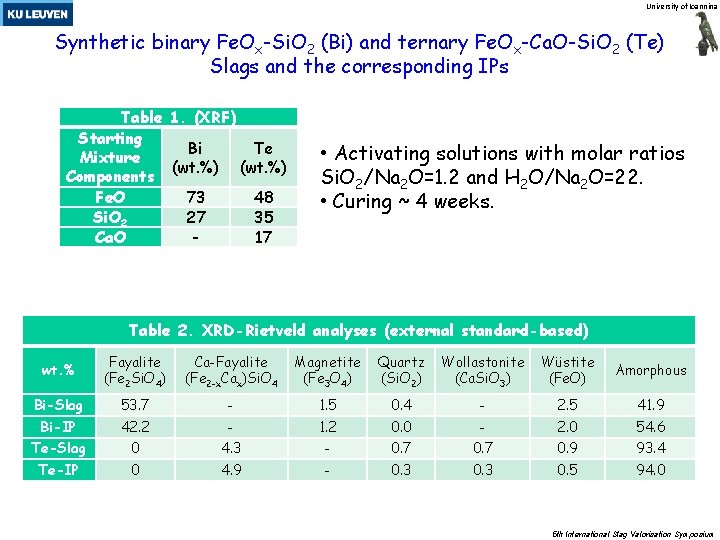
University of Ioannina Synthetic binary Fe. Ox-Si. O 2 (Bi) and ternary Fe. Ox-Ca. O-Si. O 2 (Te) Slags and the corresponding IPs Table 1. (XRF) Starting Bi Te Mixture (wt. %) Components Fe. O 73 48 Si. O 2 27 35 Ca. O 17 • Activating solutions with molar ratios Si. O 2/Na 2 O=1. 2 and H 2 O/Na 2 O=22. • Curing ~ 4 weeks. Table 2. XRD-Rietveld analyses (external standard-based) wt. % Fayalite (Fe 2 Si. O 4) Ca-Fayalite (Fe 2 -x. Cax)Si. O 4 Magnetite (Fe 3 O 4) Quartz (Si. O 2) Wollastonite (Ca. Si. O 3) Wüstite (Fe. O) Amorphous Bi-Slag 53. 7 - 1. 5 0. 4 - 2. 5 41. 9 Bi-IP 42. 2 - 1. 2 0. 0 - 2. 0 54. 6 Te-Slag 0 4. 3 - 0. 7 0. 9 93. 4 Te-IP 0 4. 9 - 0. 3 0. 5 94. 0 5 th International Slag Valorisation Symposium

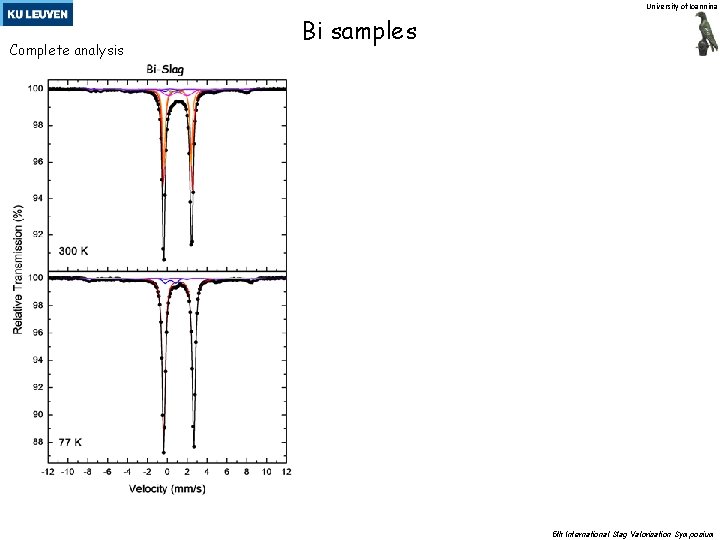
University of Ioannina Complete analysis Bi samples 5 th International Slag Valorisation Symposium

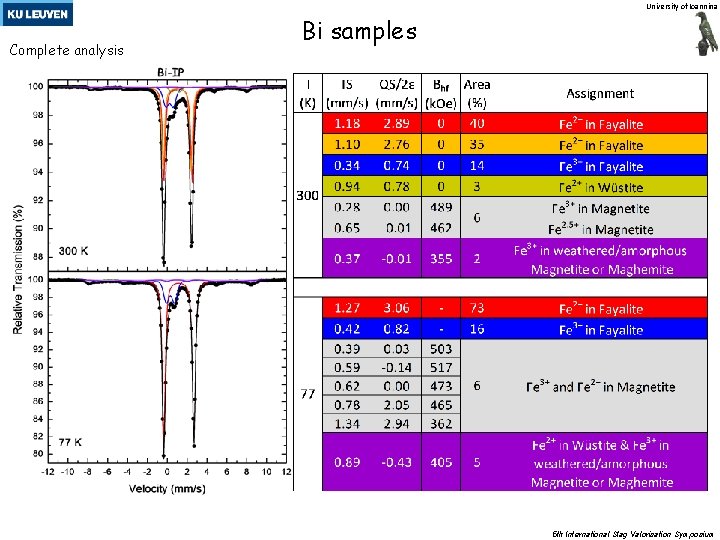
University of Ioannina Complete analysis Bi samples 5 th International Slag Valorisation Symposium

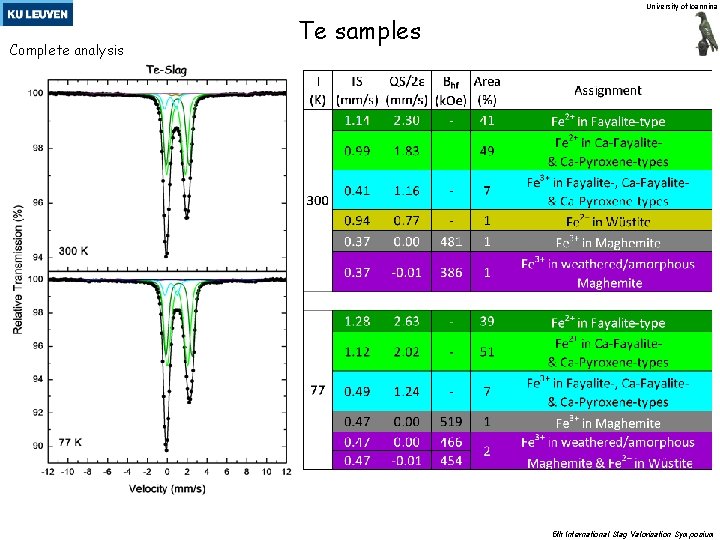
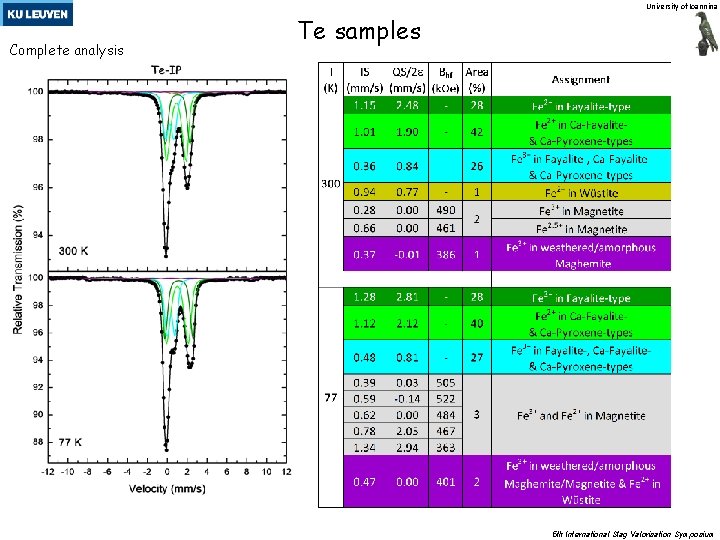
University of Ioannina Complete analysis Te samples 5 th International Slag Valorisation Symposium

University of Ioannina Complete analysis Te samples 5 th International Slag Valorisation Symposium

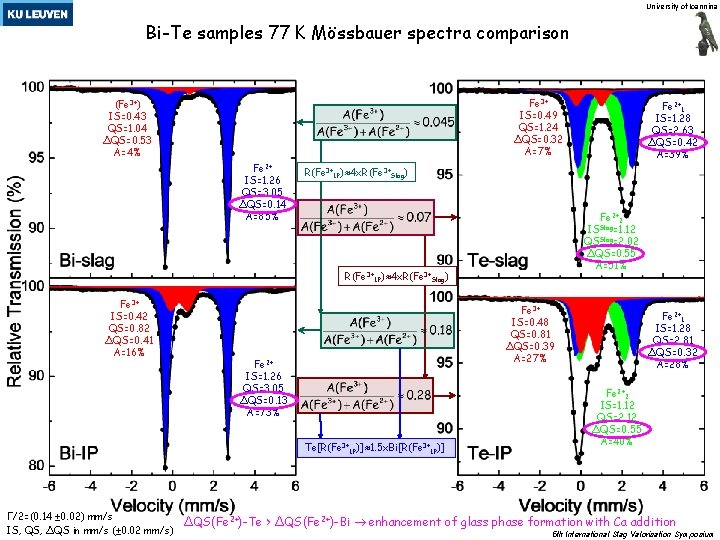
University of Ioannina Bi-Te samples 77 K Mössbauer spectra comparison Fe 3+ IS=0. 49 QS=1. 24 ΔQS=0. 32 A=7% (Fe 3+) IS=0. 43 QS=1. 04 ΔQS=0. 53 A=4% Fe 2+ IS=1. 26 QS=3. 05 ΔQS=0. 14 A=85% R(Fe 3+IP)≈4 x. R(Fe 3+Slag) Fe 2+2 Slag IS =1. 12 QSSlag=2. 02 ΔQS=0. 55 A=51% R(Fe 3+IP)≈4 x. R(Fe 3+Slag) Fe 3+ IS=0. 42 QS=0. 82 ΔQS=0. 41 A=16% Fe 3+ IS=0. 48 QS=0. 81 ΔQS=0. 39 A=27% Fe 2+ IS=1. 26 QS=3. 05 ΔQS=0. 13 A=73% Te[R(Fe 3+IP)]≈1. 5 x. Bi[R(Fe 3+IP)] Γ/2=(0. 14 ± 0. 02) mm/s IS, QS, ΔQS in mm/s (± 0. 02 mm/s) Fe 2+1 IS=1. 28 QS=2. 63 ΔQS=0. 42 A=39% Fe 2+1 IS=1. 28 QS=2. 81 ΔQS=0. 32 A=28% Fe 2+2 IS=1. 12 QS=2. 12 ΔQS=0. 55 A=40% ΔQS(Fe 2+)-Te > ΔQS(Fe 2+)-Bi enhancement of glass phase formation with Ca addition 5 th International Slag Valorisation Symposium

University of Ioannina 57 Fe Bi Mössbauer Spectroscopy Te Increase of the amount of Fe 3+ with Ca addition 5 th International Slag Valorisation Symposium

University of Ioannina 57 Fe Mössbauer Spectroscopy Weighted average values Bi Te Influence of Ca addition to the electronic configuration & coordination of Fe 2+/Fe 3+ 5 th International Slag Valorisation Symposium

University of Ioannina 57 Fe Mössbauer Spectroscopy Weighted average values Bi Te Influence of Ca addition to the electronic configuration & coordination of Fe 2+/Fe 3+ 5 th International Slag Valorisation Symposium

University of Ioannina 57 Fe Mössbauer Spectroscopy Weighted average values Bi Te Enhancement of glass phase formation with Ca addition 5 th International Slag Valorisation Symposium

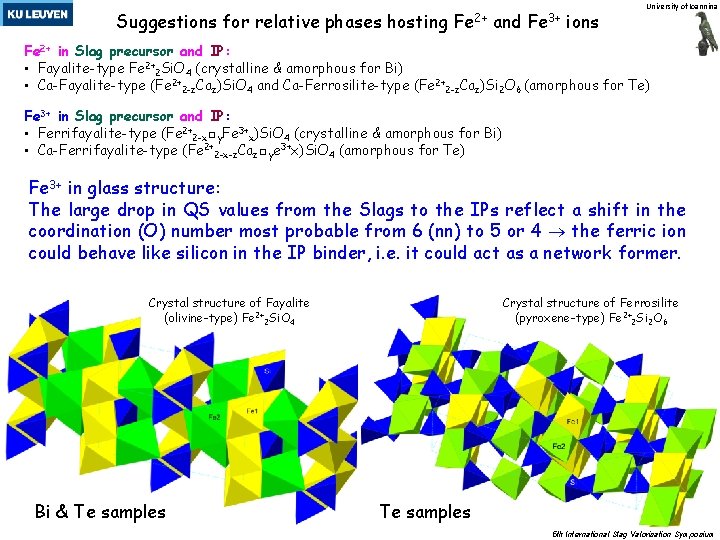
Suggestions for relative phases hosting Fe 2+ and Fe 3+ ions University of Ioannina Fe 2+ in Slag precursor and IP: • Fayalite-type Fe 2+2 Si. O 4 (crystalline & amorphous for Bi) • Ca-Fayalite-type (Fe 2+2 -z. Caz)Si. O 4 and Ca-Ferrosilite-type (Fe 2+2 -z. Caz)Si 2 O 6 (amorphous for Te) Fe 3+ in Slag precursor and IP: • Ferrifayalite-type (Fe 2+2 -x□y. Fe 3+x)Si. O 4 (crystalline & amorphous for Bi) • Ca-Ferrifayalite-type (Fe 2+2 -x-z. Caz□ye 3+x)Si. O 4 (amorphous for Te) Fe 3+ in glass structure: The large drop in QS values from the Slags to the IPs reflect a shift in the coordination (O) number most probable from 6 (nn) to 5 or 4 the ferric ion could behave like silicon in the IP binder, i. e. it could act as a network former. Crystal structure of Fayalite (olivine-type) Fe 2+2 Si. O 4 Bi & Te samples Crystal structure of Ferrosilite (pyroxene-type) Fe 2+2 Si 2 O 6 Te samples 5 th International Slag Valorisation Symposium

University of Ioannina Evolution of the 77 K 57 Fe Mössbauer spectra at different reaction stages 7 w 4 w 1 d 3 d Starting Mixture Components wt% Fe. O Si. O 2 Ca. O Al 2 O 3 Mg. O 47 34 12 5 2 • Activating solutions with molar ratios Si. O 2/Na 2 O=1. 6 and H 2 O/Na 2 O=20. 5 th International Slag Valorisation Symposium

Conclusions University of Ioannina Ø Emphatic appearance of Fe 3+ contributions in the IP samples resulting by oxidation of the Fe 2+ states existing in the Slags after chemical reaction with the activating solutions. Ø The amorphous part of the slags is the most active component of the starting Slag material, while the crystalline part is more resistive to oxidation. Ø The addition of Ca seems to favor the formation of the amorphousglass phase and enhance the presence of Fe 3+ states both in the Slags and, more pronounced, in the IPs. Ø A shift in the Mössbauer parameters of the ferric ions from the slags to the IPs indicates a change in their O-coordination, suggesting that these ions could act as network formers similar to the role of Si. Ø The oxidation of Fe 2+ to Fe 3+ in the curing period of the IPs is fast for the initial 1 -3 days, then slower up to 4 weeks and persists further up to at least 7 weeks. 5 th International Slag Valorisation Symposium

University of Ioannina Collaborators KU LEUVEN • Arne PEYS • Silviana ONISEI • Yiannis PONTIKES 5 th International Slag Valorisation Symposium

University of Ioannina-Greece Mössbauer Spectroscopy & Physics of Materials Laboratory Physics department, University of Ioannina http: //pml. physics. uoi. gr 5 th International Slag Valorisation Symposium