RAMAN SPECTROSCOPY SOLID STATE SPECTROSCOPY CLASS MISS PIMPIKA














- Slides: 19

RAMAN SPECTROSCOPY SOLID STATE SPECTROSCOPY CLASS MISS PIMPIKA PIMSORN ID: D 5410088

OUTLINE • • INTRODUCTION PRINCIPLE INSTRUMENT APPLICATION

INTRODUCTION Raman spectroscopy • is a spectroscopic technique. • bases on inelastic scattering of monochromatic light, usually from a laser source. (in the visible, nearinfrared, and near-ultraviolet range. ) • can be used to study solid, liquid and gaseous samples. • measures the vibration, rotation and other low frequency transitions in molecules.

INTRODUCTION • Sir Chandrasekhara Ventaka Raman is Indian physicist. • Discovered the “Raman effect”. • Studied extensively in X-ray Diffractions, Acoustics, Optics, Dielectrics and Colloidal solutions. http: //ibnlive. in. com/news/google-doodles-the-raman-effectfor-cv-ramans-125 th-birthday/432660 -11. html

INTRODUCTION Raman effect • is based on molecular deformations in electric field E determined by molecular polarizability α. • occurs when monochromatic light impinges upon a molecule and interacts with the electron cloud and the bonds of this molecule. • requires a changing induced dipole moment. P = αE α is the polarizability of the molecule E is the electronic field

PRINCIPLE • Monochromatic laser light with frequency υ0 excites molecules and transforms them into oscillating dipoles. Such oscillating dipoles emit light of three different frequencies I. Rayleigh scattering (no exchange of energy: incident and scattered photons have the same energy) II. Stokes Raman scattering (atom or molecule absorbs energy: scattered photon has less energy than the incident photon) III. Anti-Stokes Raman scattering (atom or molecule loses energy: scattered photon has more energy than the incident photon)

PRINCIPLE Concentration measurement I = CNI 0 F(T) I - Raman signal intensity C - is a constant, N - the number density, I 0 - the laser intensity, - the Raman cross-section, - the scattering solid angle, - the length of the observed segment of the laser beam, F(T) - a temperature dependent factor determined by the spectral width and resolution of the detection system and the investigated molecule

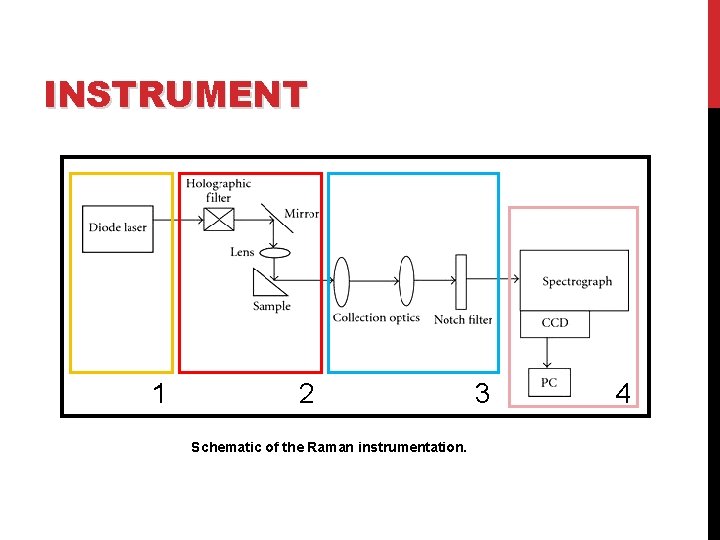
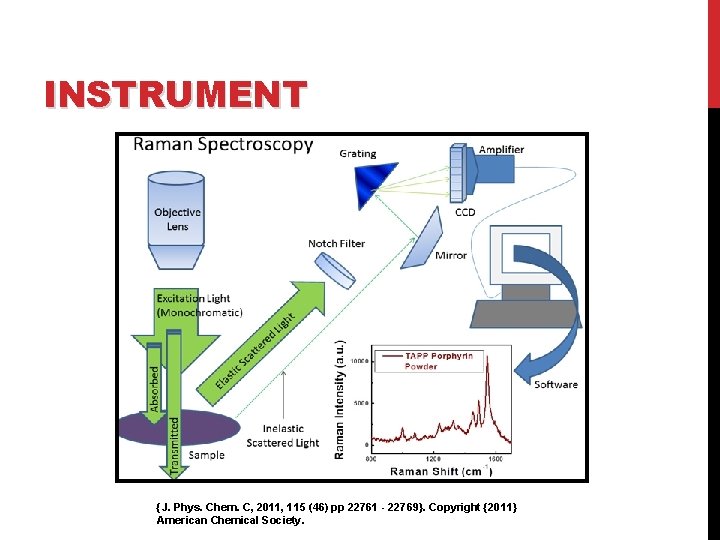
INSTRUMENT A Raman system typically consists of four major components: 1. Excitation source (Laser). 2. Sample illumination system and light collection optics. 3. Wavelength selector (Filter or Spectrophotometer). 4. Detector (Photodiode array, CCD or PMT).

INSTRUMENT 1 2 Schematic of the Raman instrumentation. 3 4

INSTRUMENT {J. Phys. Chem. C, 2011, 115 (46) pp 22761 - 22769}. Copyright {2011} American Chemical Society.

INSTRUMENT

APPLICATION 1. Stimulated Raman Scattering : SRS • Very strong laser pulse with electric field strength > 109 V·cm-1 transforms up to 50% of all laser pulse energy into coherent beam at Stokes frequency - 0 m CH 2 -stretching vibration SRS tissue imaging of fresh mouse skin. Stimulated Raman transitional schemes

APPLICATION 2. Coherent Anti-Stoke Raman : CARS • Instead of the traditional one laser, two very strong collinear lasers irradiate a sample. lipid storage in Caenorhabditis elegans CARS spectra of C. elegans. Thomas Hellerer et al. PNAS 2007; 104: 14658 -14663 Transitional scheme for CARS

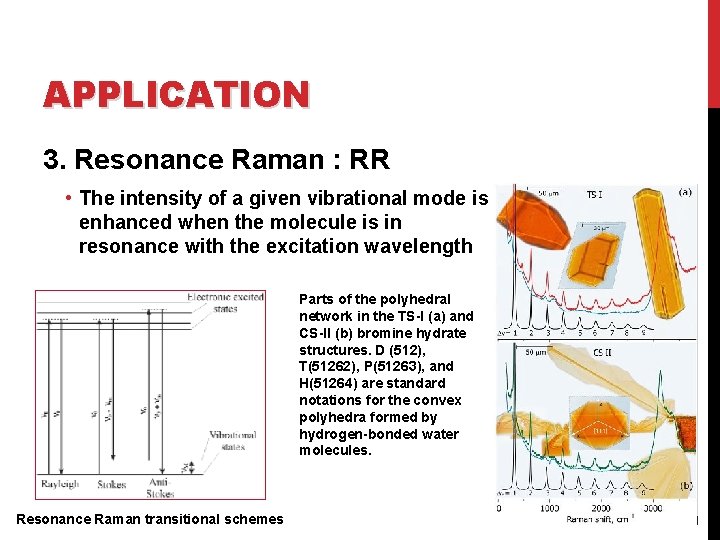
APPLICATION 3. Resonance Raman : RR • The intensity of a given vibrational mode is enhanced when the molecule is in resonance with the excitation wavelength Parts of the polyhedral network in the TS-I (a) and CS-II (b) bromine hydrate structures. D (512), T(51262), P(51263), and H(51264) are standard notations for the convex polyhedra formed by hydrogen-bonded water molecules. Resonance Raman transitional schemes

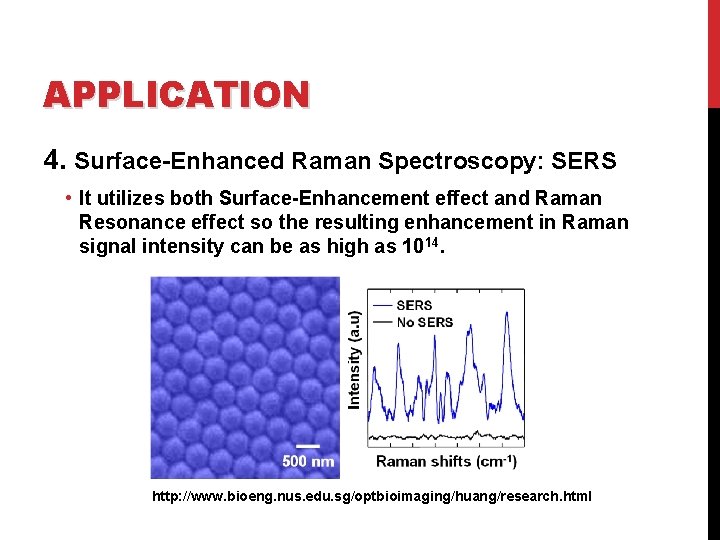
APPLICATION 4. Surface-Enhanced Raman Spectroscopy: SERS • It utilizes both Surface-Enhancement effect and Raman Resonance effect so the resulting enhancement in Raman signal intensity can be as high as 1014. http: //www. bioeng. nus. edu. sg/optbioimaging/huang/research. html

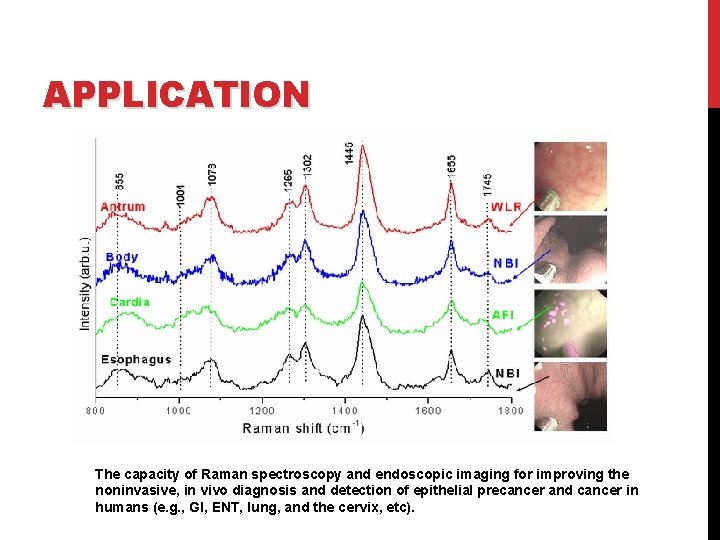
APPLICATION The capacity of Raman spectroscopy and endoscopic imaging for improving the noninvasive, in vivo diagnosis and detection of epithelial precancer and cancer in humans (e. g. , GI, ENT, lung, and the cervix, etc).

ADVANTAGES • Raman experiments do not require any tuneable laser source, any laser with high average power can be used, but since the signal is proportional to -4, short wavelengths are preferred. • Both rotational and vibrational temperatures can be obtained • Atoms, radicals as well as molecules that absorb well down in the VUV region can be measured • Raman are best suited for major species detection, i. e. N 2, O 2, H 2, CO 2, H 2 O, hydrocarbons, normally with a single shot detection limit ~1000 ppm. • The signal is linear in laser intensity, no saturation effects

DISADVANTAGES • The Raman weak scattering signal is very weak, ~1000 times weaker than Rayleigh scattering. • It is very hard to make 2 D-visualization. • The technique is sensitive to background fluorescence and stray light. • It is an incoherent technique, which means that background emission can be a problem. • It requires a laser with high average power. • Trade-off between having a sufficient signal and not damaging the windows.

THANK YOU FOR YOUR ATTENTION