Rotational Spectroscopy To a stationary observer a rotating

Rotational Spectroscopy
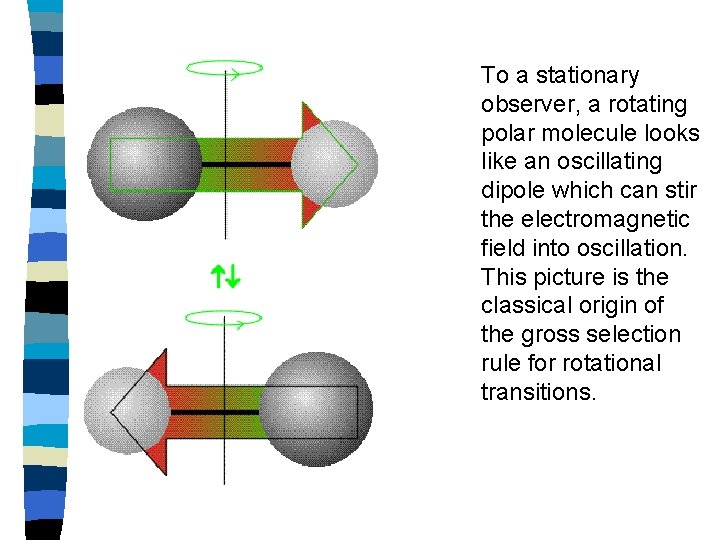
To a stationary observer, a rotating polar molecule looks like an oscillating dipole which can stir the electromagnetic field into oscillation. This picture is the classical origin of the gross selection rule for rotational transitions.
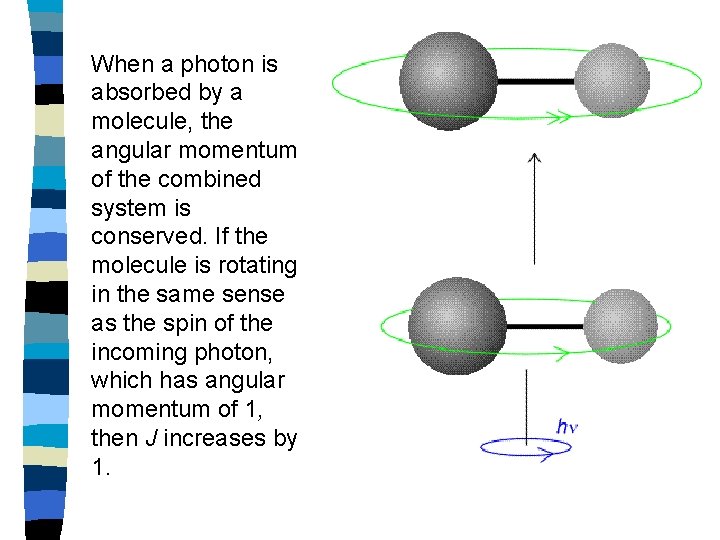
When a photon is absorbed by a molecule, the angular momentum of the combined system is conserved. If the molecule is rotating in the same sense as the spin of the incoming photon, which has angular momentum of 1, then J increases by 1.
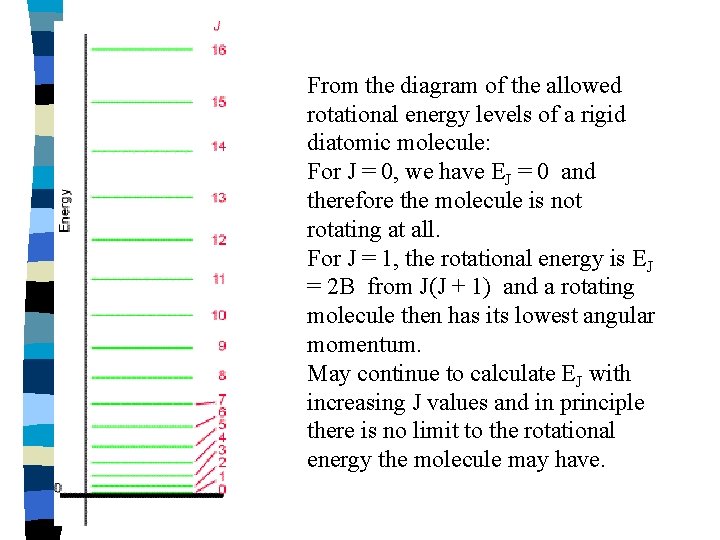
From the diagram of the allowed rotational energy levels of a rigid diatomic molecule: For J = 0, we have EJ = 0 and therefore the molecule is not rotating at all. For J = 1, the rotational energy is EJ = 2 B from J(J + 1) and a rotating molecule then has its lowest angular momentum. May continue to calculate EJ with increasing J values and in principle there is no limit to the rotational energy the molecule may have.

• In practice, reach a point where the centrifugal force of a rapidly rotating diatomic molecule is greater than the strength of the bond, and the molecule is disrupted, but this point is not reached at normal temperatures. Consider energy differences in order to discuss the spectrum. Note that spectroscopists speak of term values, F(J), having units of either frequency (by dividing by h): Or, units of wavenumbers, by dividing by hc: To avoid confusion, I will continue to use tildes to indicate wavenumbers.

Therefore absorption line will appear at 2 B cm-1 Similarly Therefore step-wise raising of the rotational energy results in an absorption spectrum consisting of lines at 2 B, 4 B, 6 B … cm-1 and separation 2 B.

The Non-Rigid Rotor n n The assumption of a rigid bond is only an approximation. The separation between successive lines (and hence the apparent B value) decreases steadily with increasing J n The reason for this decrease may be seen if we calculate internuclear distances from the B values. n The bond length increases with J – this reflects the fact that the more quickly a diatomic molecule rotates, the greater the centrifugal force tending to move the atoms apart.

Two consequences of elasticity n 1) When the bond is elastic, a molecule may have vibrational energy. i. e. , a bond will stretch and compress periodically with a certain fundamental frequency dependent upon the masses of the atoms and the elasticity (or force constant) of the bond. n If the motion is simple harmonic (which is a good approximation) the force constant is given by: Where is the vibrational frequency (cm-1) - The variation of B with J is determined by the force constant – the weaker the bond, the more readily will it distort under centrifugal forces.

2) The quantities r and B vary during a vibration – when these quantities are measured by microwave techniques many hundreds of vibrations occur during a rotation, and hence the measured value is an average. However, from the equation for B we have Since all other quantities are independent of vibration.
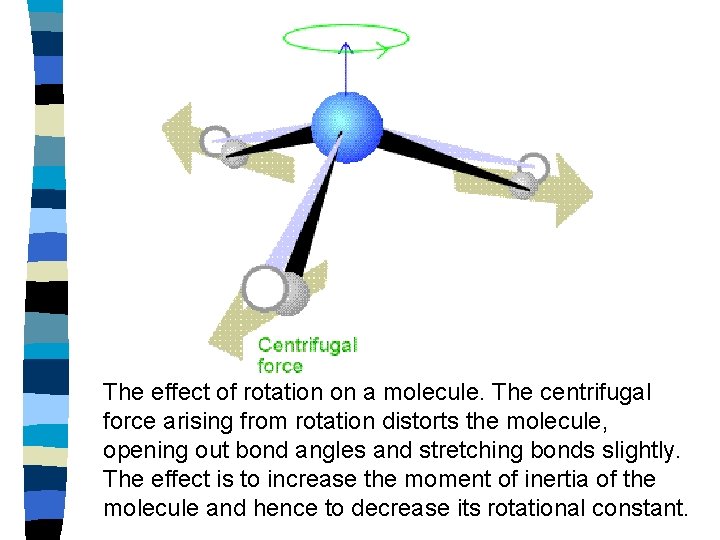
The effect of rotation on a molecule. The centrifugal force arising from rotation distorts the molecule, opening out bond angles and stretching bonds slightly. The effect is to increase the moment of inertia of the molecule and hence to decrease its rotational constant.

The Spectrum of a Non-Rigid Rotator The Schrodinger eqn may be set up for a non-rigid molecule and the rotational energy levels are found to be for a simple harmonic force field: which is a positive quantity

If the force field is anharmonic, the expression becomes Where H, K etc. are small constants dependent on the geometry of the molecule, and negligible compared with D Most spectroscopic data is adequately fitted by eqn for the simple harmonic force field. Solving for h in terms of B, and substituting into D gives:

Note: vibrational frequencies are usually of the order of 103 cm-1 B is of the order 10 cm-1 D is of the order 10 -3 cm-1 i. e. is very small compared with B. For small J, the correction term DJ 2(J + 1)2 is almost negligible For J values of 10 or more, it may become appreciable. Selection rules remain the same J = 1

Knowing D one can (rather inaccurately) calculate the vibrational frequency of a diatomic molecule The force constant is then calculated:

Raman Spectroscopy n Molecular energy levels are explored by examining the frequencies present in radiation scattered by molecules. n Most of the radiation is scattered without change of frequency, giving the Rayleigh line. n About 1 in 107 of the incident photons give up some energy in collision with the sample molecules, emerging with lower energy, giving lower frequency Stokes lines.

n Other incident photons may collect energy from excited sample molecules, emerging as higher frequency anti-Stokes lines. n For Raman spectroscopy to work, lasers are required: 1) frequency shifts in the scattered radiation are small, so highly monochromatic radiation is required in order to observe the shifts. 2) The intensity of the scattered radiation is so low that an intense incident beam is needed. n n

Rotational Raman Spectra Gross selection rule for rotational Raman transitions: molecule must be anisotropically polarizable An electric field applied to a molecule results in its distortion, and the distorted molecule acquires a contribution to its dipole moment (even if it is nonpolar initially). The polarizability may be different when the field is applied (a) parallel or (b) perpendicular to the molecular axis (or, in general, in different directions relative to the molecule); if that is so, then the molecule has an anisotropic polarizability.

The distortion of a molecule in an electric field is determined by its polarizability . In addition to any permanent dipole moment a molecule may have, the molecule acquires an induced dipole moment , if the strength of the field is : An atom is isotropically polarizable. All linear molecules and diatomics (whether homonuclear or heteronuclear) have anisotropic polarizabilities rotationally Raman active allows study of many molecules that are inaccessible to microwave spectroscopy. but some types of rotors both rotationally Raman & microwave inactive
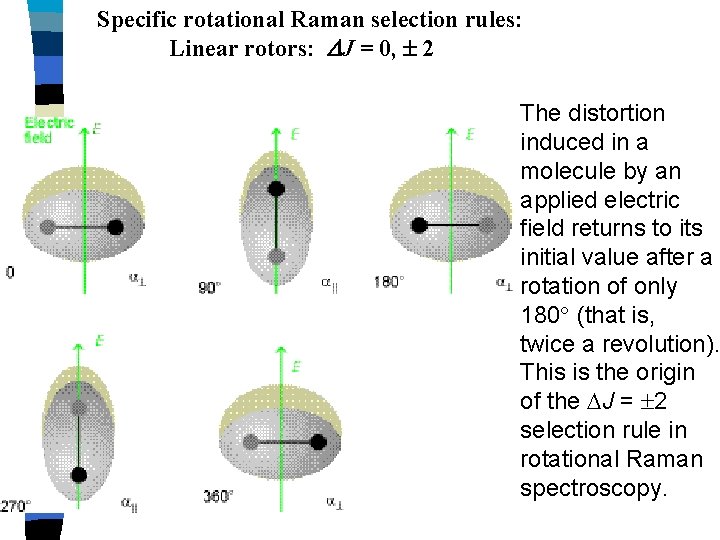
Specific rotational Raman selection rules: Linear rotors: J = 0, 2 The distortion induced in a molecule by an applied electric field returns to its initial value after a rotation of only 180 (that is, twice a revolution). This is the origin of the J = 2 selection rule in rotational Raman spectroscopy.
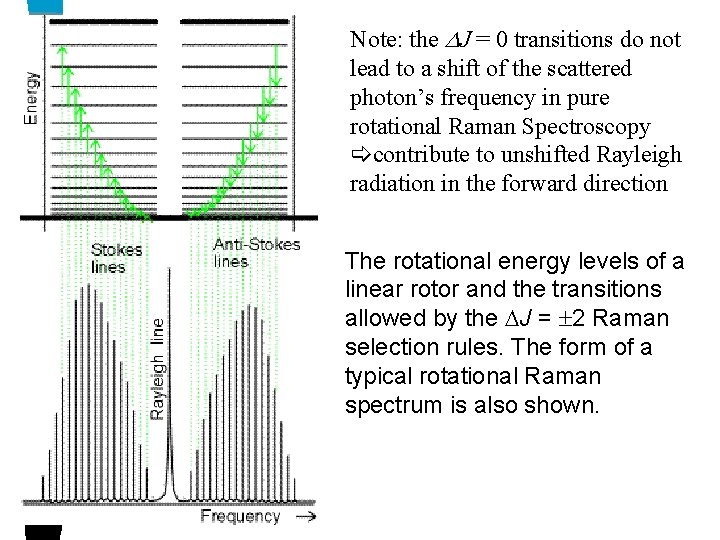
Note: the J = 0 transitions do not lead to a shift of the scattered photon’s frequency in pure rotational Raman Spectroscopy contribute to unshifted Rayleigh radiation in the forward direction The rotational energy levels of a linear rotor and the transitions allowed by the J = 2 Raman selection rules. The form of a typical rotational Raman spectrum is also shown.
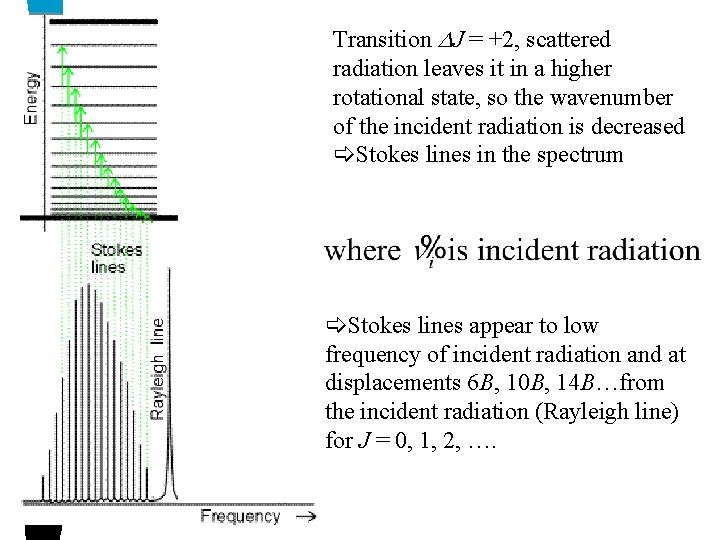
Transition J = +2, scattered radiation leaves it in a higher rotational state, so the wavenumber of the incident radiation is decreased Stokes lines in the spectrum Stokes lines appear to low frequency of incident radiation and at displacements 6 B, 10 B, 14 B…from the incident radiation (Rayleigh line) for J = 0, 1, 2, ….
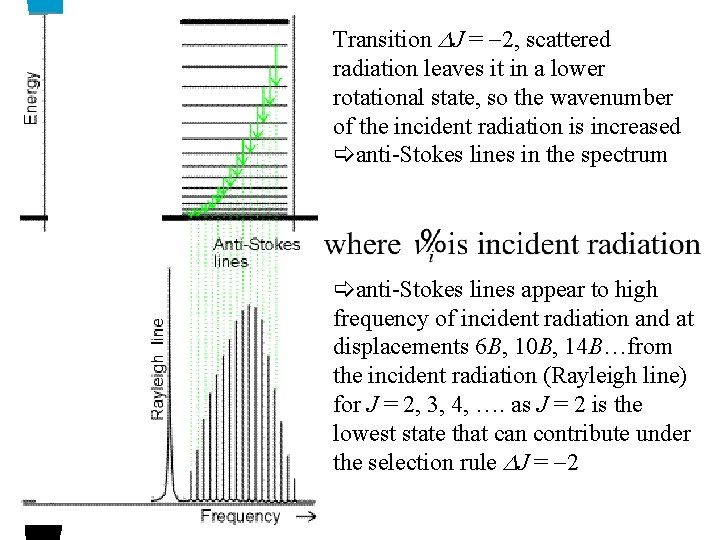
Transition J = -2, scattered radiation leaves it in a lower rotational state, so the wavenumber of the incident radiation is increased anti-Stokes lines in the spectrum anti-Stokes lines appear to high frequency of incident radiation and at displacements 6 B, 10 B, 14 B…from the incident radiation (Rayleigh line) for J = 2, 3, 4, …. as J = 2 is the lowest state that can contribute under the selection rule J = -2

Pure rotation spectra of polyatomics The rotation of a 3 -dimensional body may be quite complex. It is convenient to resolve it into rotational components about 3 mutually perpendicular directions through the centre of gravity – the principal axis of rotation. Therefore a body has three principal moments of inertia, one about each axis, usually designated Ia, Ib, and Ic
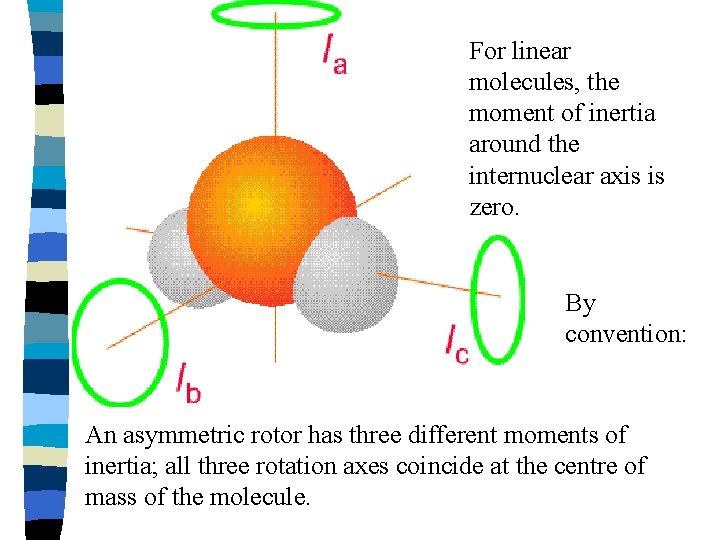
For linear molecules, the moment of inertia around the internuclear axis is zero. By convention: An asymmetric rotor has three different moments of inertia; all three rotation axes coincide at the centre of mass of the molecule.
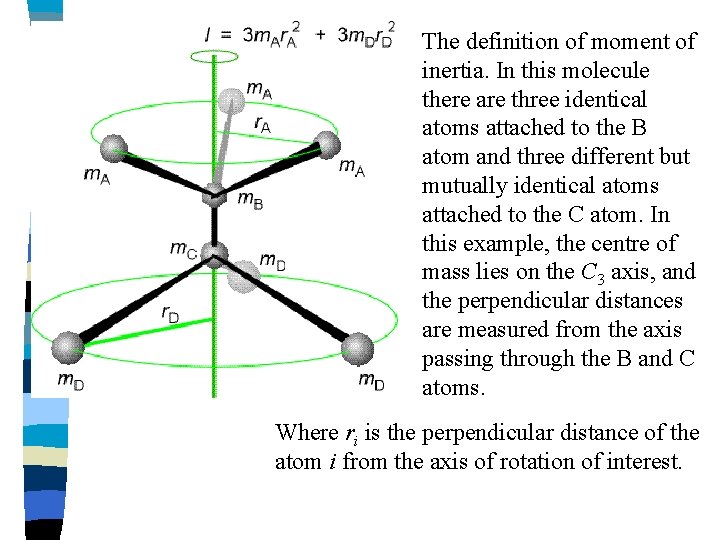
The definition of moment of inertia. In this molecule there are three identical atoms attached to the B atom and three different but mutually identical atoms attached to the C atom. In this example, the centre of mass lies on the C 3 axis, and the perpendicular distances are measured from the axis passing through the B and C atoms. Where ri is the perpendicular distance of the atom i from the axis of rotation of interest.

Molecules may be classified into groups according to the relative values of their three principal moments of inertia: 1) Linear Molecules 2) Symmetric Tops 3) Spherical Tops 4) Asymmetric Tops 1) Linear Molecules (e. g. , HCl, OCS (carbon oxy-sulfide)) 3 directions of rotation: (a) about the bond axis (b) end-over-end rotation in the plane of the screen (c) end-over-end rotation at right angles to the plane (b) and (c) are the same (i. e. , Ib = Ic) (a) must be zero, as the distances of all the atoms to the bond axis must be zero (i. e. , Ia = 0)

2) Symmetric Tops (e. g. , methyl fluoride) (a) about the bond axis (e. g. , C-F bond axis chosen as the main rotational axis since the centre of gravity lies along it is now not negligible because it involves the rotation of 3 comparatively massive H atoms off this axis - like a top (b) end-over-end rotation in the plane of the screen (c) end-over-end rotation at right angles to the plane (b) and (c) are the still the same (i. e. , Ib = Ic) Symmetric Tops 2 subdivisions of this class: • Prolate symmetric top (eg. , methyl fluoride) Ib = Ic > Ia revolution about longer axis • Oblate symmetric top (eg. , boron trifluoride) Ib = Ic < Ia revolution of ellipse about shorter axis

3) Spherical Tops (eg. , methane) when a molecule has all 3 moments of inertia identical However, these molecules have no dipole moment because of their symmetry, rotation alone can produce no dipole change and hence no rotational spectrum is observable. 4) Asymmetric Tops (eg. , water, vinyl chloride) these molecules to which the majority of substances belong, have all 3 moments of inertia different
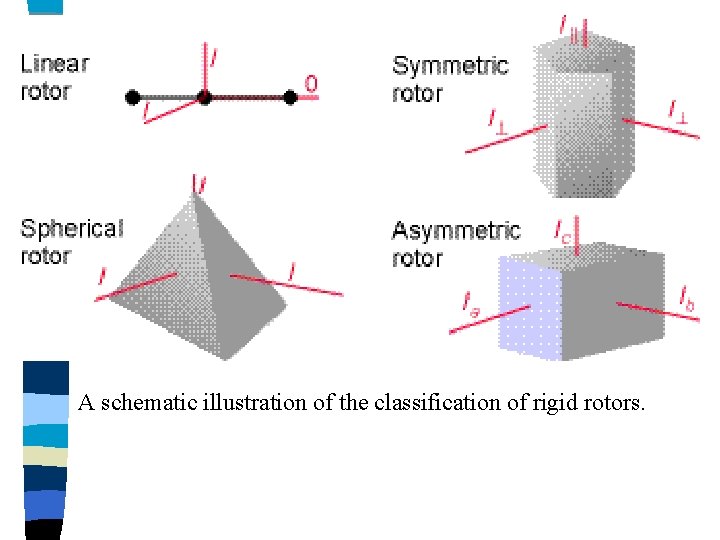
A schematic illustration of the classification of rigid rotors.

Classification based on point group n Linear rotors must, obviously, be C v or D h. n A symmetric rotor must have either a Cn axis with n>2, or an S 4 axis. Point groups include Sn (n ≥ 4), Cnv, Cnh, Dnh (all with n≥ 3) and all Dnd (note that D 2 d has an S 4 axis). n Molecules in the higher symmetry groups, Td , Oh, Ih, are spherical rotors.

Linear Molecules Consider first molecules such as OCS or , where all the atoms lie on a straight line gives rise to particularly simple spectra in the microwave region. Since Ib = Ic , Ia = 0 as for diatomic molecules, the energy levels are given by And the spectrum will show the same 2 B separation modified by the distortion constant. i. e. , discussion on diatomic molecules applies equally to all linear molecules

However, consider following 3 points: 1) Since the moment of inertia for the end-over-end rotation of a polyatomic linear molecule is considerably greater than that of a diatomic molecule, the B value will be much smaller, and the spectral lines more closely spaced. B values for diatomic molecules are ~ 10 cm-1 while for triatomic molecules they can be 1 cm-1 or less, and for larger molecules smaller still.

n n n 2) The molecule must as usual possess a dipole moment if it is to exhibit a rotational spectrum: i. e. , OCS will be microwave active, while CO 2 (OCO) will not. Note also: isotopic substitution does not lead to a dipole moment since the bond lengths and atomic charges are unaltered by the substitution 16 OC 18 O is microwave inactive 3) A non cyclic polyatomic molecule containing N atoms has altogether N-1 individual bond lengths to be determined. in OCS r. CO and r. CS But there is only 1 moment of inertia for the end-overend rotation of OCS, and only this one value can be determined from the spectra.

Symmetric Top Molecules Although the rotational energy levels of this type of molecule are more complicated than those of the linear molecules, because of their symmetry their pure rotational spectra are still relatively simple. e. g. , CH 3 F where Ib = Ic Ia Ia 0 2 directions of rotation in which the molecule can absorb or emit energy: along main symmetry axis (C-F bond) and to it. Need to have 2 quantum numbers to describe the degree of rotation: - one for Ia - one for Ib or Ic It is convenient mathematically to have a quantum number to represent the total angular momentum of the molecule which is the sum of the separate angular momenta about the two different axes

Use J – where J is the total angular momentum (note that in linear molecules J also represents the angular momentum Use K – to represent the angular momentum about the top axis, i. e. , C-F bond (spinning axis) Allowed values for K and J: both must, by conditions of quantum mechanics, be integral or zero. The total angular momentum can be as large as we like – i. e. , (except that a real molecule will be disrupted at very high rotational speeds) Once we have chosen J, however, K is more limited.

e. g. , J = 3 - the rotational energy can be divided in several ways between motion about the main symmetry axis and motion to it. - if all the rotation is about the top axis, then K = 3, since J is total angular momentum (note: K can not be greater than J) -or could have K = 2, 1, 0 in which case the momentum to the axis would have 1, 2, 3 respectively -additionally K can be negative i. e. , imagine positive and negative values of K to correspond with clockwise and anticlockwise rotation about the symmetry axis and can have values of -1, -2, -3 K = J, J – 1, J – 2, …. 0 …. -(J – 1), -J which is a total of 2 J + 1 values altogether
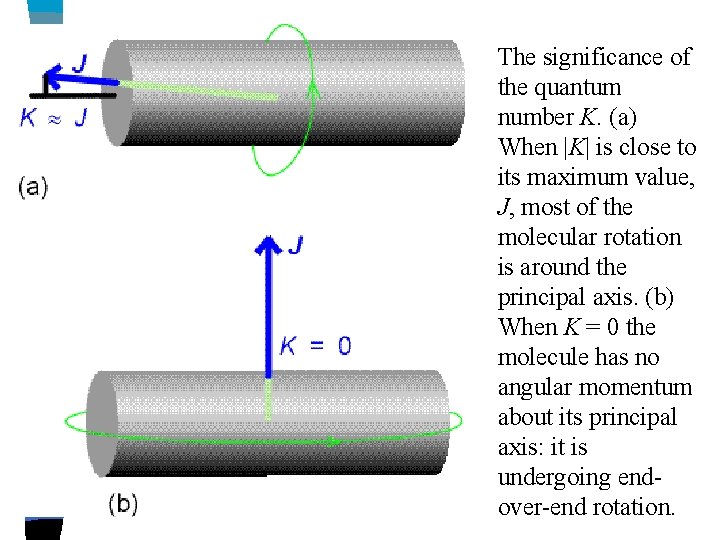
The significance of the quantum number K. (a) When |K| is close to its maximum value, J, most of the molecular rotation is around the principal axis. (b) When K = 0 the molecule has no angular momentum about its principal axis: it is undergoing endover-end rotation.

Consider the rigid symmetric top: i. e. , one in which the bonds are not supposed to stretch under centrifugal forces) Solving Schrodinger equation gives Note: energy depends on K 2 , so it is immaterial whether the top spins clockwise or anticlockwise; the energy is the same for a given angular momentum. for all K > 0, the rotational energy levels are doubly degenerate. - the selection rules for this molecule are:

Applying these to the above equation, the spectrum for rigid symmetric top is given by: Thus the spectrum is independent of K, and hence rotational changes about the symmetry axis do not give rise to a rotational spectrum. - i. e. , rotation about the symmetry axis does not change the dipole moment to the axis (which always remains zero), and hence the rotation cannot interact with radiation. the spectrum is the same as for a linear molecule and only one moment of inertia, i. e. , that for end-over-end rotation can be measured.
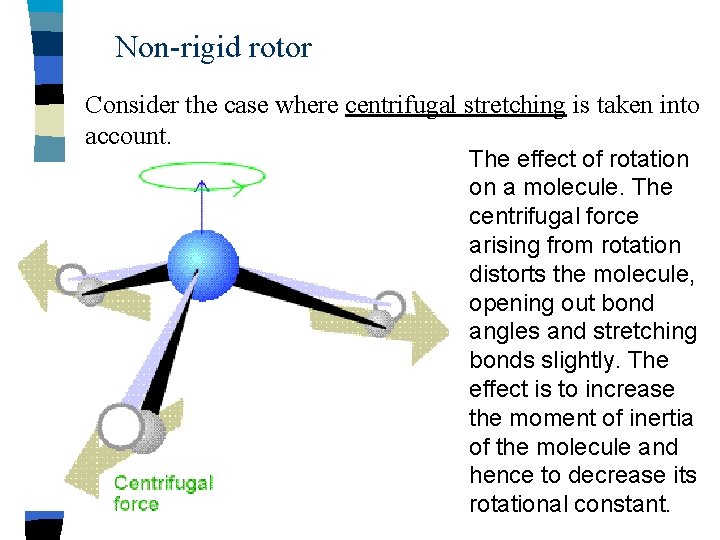
Non-rigid rotor Consider the case where centrifugal stretching is taken into account. The effect of rotation on a molecule. The centrifugal force arising from rotation distorts the molecule, opening out bond angles and stretching bonds slightly. The effect is to increase the moment of inertia of the molecule and hence to decrease its rotational constant.

The energy levels become: The selection rules are unchanged and so the spectrum is: The spectrum will be basically that of a linear molecule (including centrifugal stretching), with an additional term dependent on K 2

It is easy to see why this spectrum now depends on the axial rotation (i. e. , depends on K) although such rotation produces no dipole change. When K = 0 no axial rotation When K > 0 i. e. , K = 1, molecule is rotating about the symmetry axis. axial rotation widens the HCH angles and stretches the C-H bonds. The distorted molecule has a different moment of inertia for end-over-end rotation than the undistorted molecule. Rewriting the above equation gives: The centrifugal distortion constants DJ and DJ, K can be considered as correction terms to the rotational constant B, and hence as perturbing the momentum Ib.

Since each value of J is associated with 2 J + 1 values of K, each line characterized by a certain J value must have 2 J + 1 components. However, since K only appears as K 2 in the equation, there will be only J + 1 different frequencies all those with K > 0 being doubly degenerate. Note: each spectrum yields only one value of B, but the spectra of isotopic molecules can in principle give sufficient information for the calculation of all bond lengths and angles of symmetric top molecules, together with estimates of the force constant of each bond.

Asymmetric Top Molecules Since spherical tops show no microwave spectrum, the only other class of molecules of interest here is the asymmetric top which has all three moments of inertia different. Their rotational energy levels and spectra are very complex each molecule and spectrum must be treated as a separate case and much tedious computation is necessary before structural parameters can be determined. The best method of attack so far has been to consider the asymmetric top as falling somewhere between the oblate and prolate symmetric top, interpolation between the two sets of energy levels of the latter leads to approximation of the energy level – and hence spectrum – of the asymmetric molecule. Methods such as this have been quite successful, and much very precise structural data has been published.

Specific rotational Raman selection rules Linear rotors: J = 0, 2 Symmetric rotors: J = 0, 1, 2; K = 0 Asymmetric rotors: For the latter, K is not a good quantum number, so additional selection rules become too complex. A good quantum number is one which is conserved in the presence of an external interaction.
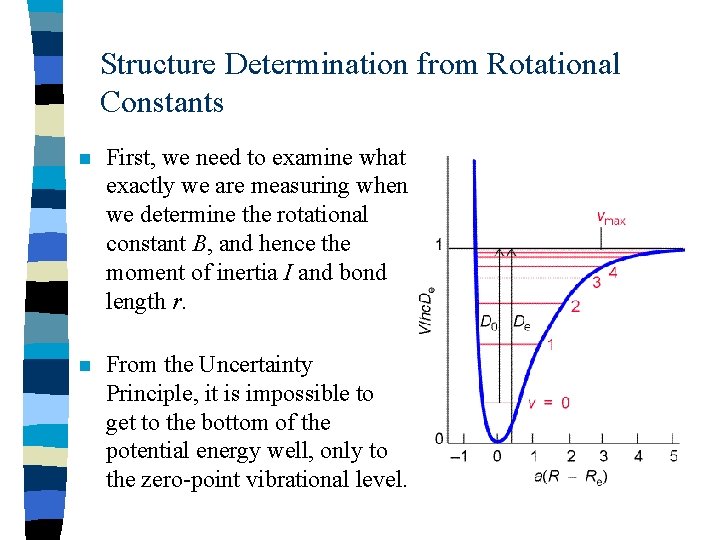
Structure Determination from Rotational Constants n First, we need to examine what exactly we are measuring when we determine the rotational constant B, and hence the moment of inertia I and bond length r. n From the Uncertainty Principle, it is impossible to get to the bottom of the potential energy well, only to the zero-point vibrational level.

A way around the problem n In general, we are measuring B 0. However, it is possible to obtain Be, relating to the unattainable equilibrium position, at least for diatomic molecules. n IF measurements can be made on excited vibrational states, the rotational constants are both slightly vibrationally dependent, so the term values should be written as

Needed equations n Spectral lines are now given by n The vibrational dependence of B is, to a good approximation, given by: where a is a vibration-rotation interaction constant and u is the vibrational quantum number.

Limitations n The vibrational dependence of the centrifugal distortion constant is too small to be of concern. n In order to obtain Be and hence the equilibrium bond length re, Bu must be obtained in at least two vibrational states. n Rotational transitions in excited vibrational states are in general very weak.

Does it really matter? n Unless we require a high degree of accuracy, it does not matter much whether we are determining re or r 0. Molecule 14 N 2 15 N 2 r 0 (Å) 1. 100105 ± 0. 000010 1. 099985 ± 0. 000010 re (Å) 1. 097651 ± 0. 000030 1. 097614 ± 0. 000030 Note that re is independent of isotopic substitution but r 0 is not.
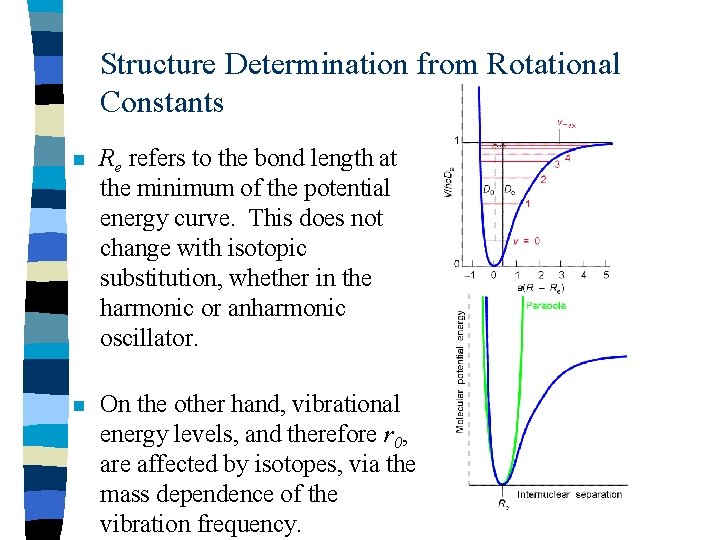
Structure Determination from Rotational Constants n Re refers to the bond length at the minimum of the potential energy curve. This does not change with isotopic substitution, whether in the harmonic or anharmonic oscillator. n On the other hand, vibrational energy levels, and therefore r 0, are affected by isotopes, via the mass dependence of the vibration frequency.
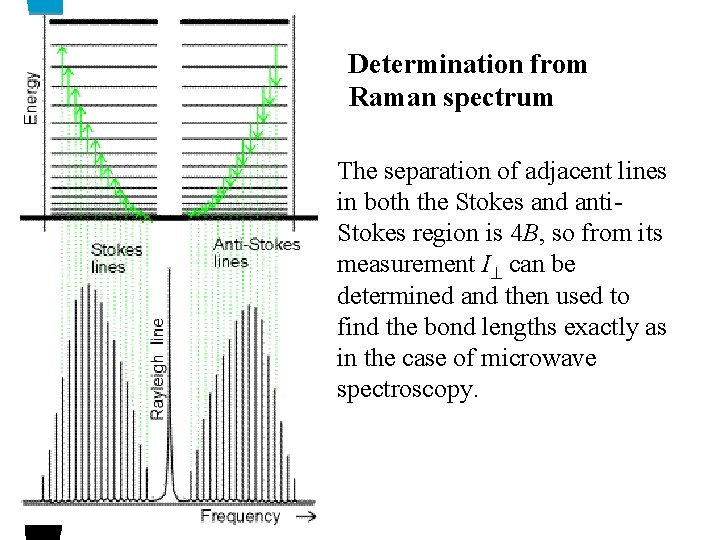
Determination from Raman spectrum The separation of adjacent lines in both the Stokes and anti. Stokes region is 4 B, so from its measurement I can be determined and then used to find the bond lengths exactly as in the case of microwave spectroscopy.

Finding most populated level of rotational states n We can find the most populated level by differentiating the population function wrt J, setting the derivative to zero, finding the maximum, and rounding to the nearest integer. n The population function, after substituting for E, and using B in cm-1, is:

Example problem (populations) n Find the ratio of the population of one of the J = 2 states to that of the J = 0 state for CO, for which the rotational constant is 5. 7898 × 1010 s-1. n Find the ratio of the population of the J = 2 level to the J = 0 level at this temperature. n Find the level with the largest population at this temperature.

Example problem (structures) n From the value of B 0 of 1. 923604 cm-1, obtained from the rotational Raman spectra of 14 N 15 N, calculate the bond length r 0. n Note that this differs from the value given above for 14 N 2, because the zero-point level is lower for the heavier isotopic species. Therefore the midpoint of the zero-point level is at a smaller value of the bond length. We needed at least 5 sig figs in order to see this difference.

Spin Angular Momentum n In addition to orbital angular momentum, there is also angular momentum arising from the motion of the electron about its own axis. This is called spin angular momentum or intrinsic angular momentum, and has no classical analogue. n For spin angular momentum we use the quantum number s. In general, the magnitude is given by

Quantization of Spin n Particles with half-integral spins are called fermions (includes electrons, protons and neutrons). n Particles with integral spins (includes photons and some atomic nuclei) are called bosons. n Atoms or molecules with an even number of fermions are bosons; if they contain an odd number of fermions they are also fermions.

Spin magnetic quantum number n n n For an electron it turns out that only one value of s is allowed, s =1/2. The projection on the zaxis is denoted by the spin magnetic quantum number, ms, which may have 2 s +1 orientations, or two for an electron, ms = 1/2. The magnitude of this component is

Inclusion of Spin in Wavefunction n The spin can be included by multiplying by a spin function. The convention is to use for a clockwise spin function (also denoted by ) and for a counterclockwise spin (also denoted by ). The spin function is thought of as a function of some unspecified spin coordinates, which we do not explicitly represent.

Two-Electron Spin Functions n To be fully correct, electron spin must be included in the wavefunction. Our first impulse might be to write the following four spin functions for twoelectron systems: •

Uncertainty yet again n However, the last two functions are invalid because they distinguish between the electrons. Electrons are identical to one another, and there is no way of experimentally determining which electron has ms = +1/2 and which has ms = -1/2. n The Heisenberg uncertainty principle makes it impossible to follow the path of a particle in quantum mechanics. Therefore, the wavefunction must not distinguish between the two electrons.

Probability Densities for Two Particles n The principle of indistinguishability means the probability density must remain unchanged if the locations of the two particles are interchanged: – With real functions, there are only two ways to satisfy this condition.

The Two Ways n n – In this case the wavefunction is symmetric with respect to interchange. Bosons obey the symmetric case. – In this case the wavefunction is antisymmetric with respect to interchange. Fermions obey the antisymmetric case.

Proper Spin Functions n n The two-electron spin functions are still valid. The other valid spin functions are where C is a normalization constant, in this case equal to 2 - /. These spin functions are combined with symmetric or antisymmetric spatial functions to give an antisymmetric total wavefunction.

Wavefunction must be Antisymmetric n n If the orbitals and are the same, the twoparticle wavefunction is the difference of two identical terms and vanishes. Any physically meaningful two-electron orbital wavefunction cannot contain the same spin orbital more than once in any term.

Pauli Exclusion Principle n Generalizating of our observation for two electrons: In an orbital wavefunction, no two electrons can occupy the same spin orbital. n This requires that if there are two electrons in one spatial orbital, their spins must be paired, i. e. , one electron has spin (ms = ½) and the other electron has spin

Nuclear spin n Exactly analogous to electron spin is nuclear spin, which must also be accounted for. The total wavefunction can be written as where the electronic wavefunction includes the electron spin function, and the nuclear wave function now includes a nuclear spin function in addition to the vibrational and rotational wavefunctions.

Nuclear spin quantum numbers n Nuclei which are fermions (odd total number of protons and neutrons) will be antisymmetric to nuclear exchange, and have nuclear spin quantum number I = n + ½, where n is zero or a positive integer. n Nuclei which are bosons will be symmetric to nuclear exchange, and have nuclear spin quantum number I = n. n The projection on the z-axis is denoted by the nuclear spin magnetic quantum number, MI, which may have 2 I +1 orientations.

Nuclear spin functions n For a diatomic molecule with I = 1/2, the possible nuclear spin functions are: where the first 3 are symmetric (s) to nuclear exchange and the 4 th is antisymmetric (a).

n In general for a homonuclear diatomic molecule there are (2 I+1)(I+1) symmetric and (2 I+1)(I) antisymmetric nuclear spin wave functions, so we have which gives rise to nuclear spin statistical weights.


Symmetry properties n In the simple case of 1 H 2, the electronic and vibrational wavefunctions are symmetric to nuclear exchange, so we need only consider the behaviour of n Since I=1/2 for 1 H, we are dealing with fermions, and thus must be antisymmetric to nuclear exchange.

Rotational symmetry n For even values of the rotational quantum number J, r is symmetric to exchange, and for odd values of J it is antisymmetric. n In order that is always antisymmetric for 1 H the antisymmetric are associated with even 2 ns J states and the symmetric ns are associated with odd J states. These give rise to para- and orthohydrogen, respectively.

The strange case of oxygen n For 16 O, I=0. Since each 16 O is a boson, the total wavefunction must be symmetric to nuclear exchange. n In the case of 16 O 2, which has 2 electrons with unpaired spins in the ground state, e is antisymmetric, and there are no antisymmetric nuclear spin functions (only ns=1). To obtain a symmetric total wavefunction, r must be antisymmetric, so only odd values of J are allowed.
- Slides: 74