Precipitation Reactions Introduction to precipitates Discuss key ideas











- Slides: 12

Precipitation Reactions

Introduction to precipitates Ø Discuss key ideas: Ø Ø Define salts (including polyatomic ions) Soluble insoluble Electrostatic attraction of water Define precipitate - insoluble salt. Ø Activity: Ø Read Science Search 4 p 156 & 157. Ø Do Copy & Complete on p 157 Ø Answer questions: 1, 2, 4 & 8

General Precipitation reactions Ø Can be written as: Soluble Salt + Soluble Salt -> Insoluble Salt + Soluble Salt (Precipitate) Ø That is, two clear solutions mix to form a solution that is either cloudy, or has solid particles in the solution Ø Example Na. Cl + Soluble Ag. NO 3 -> Soluble Ag. Cl Insoluble + Na. NO 3 Soluble

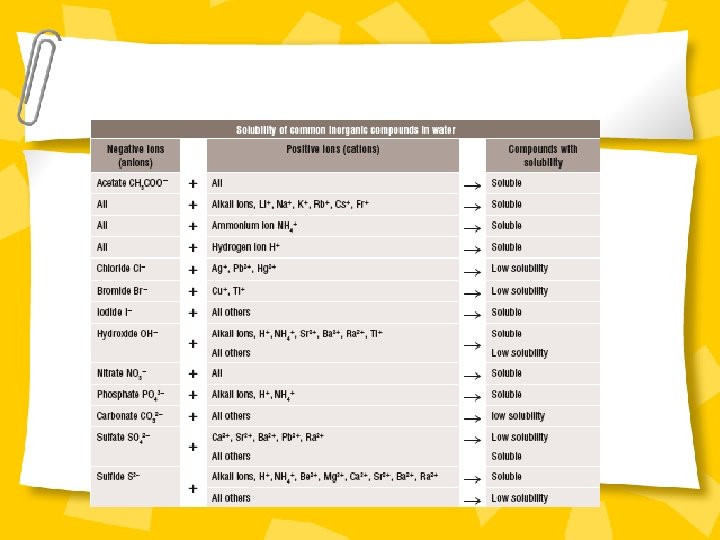
Precipitation Experiment Ø Read handout and complete experiment. Ø Answer question on solubility rules.

Precipitate handout

Precipitate experiment

Questions / discussion • Write your own table of results for the experiment. • Analyse the table of solubility rules to work out what salt has precipitated from solutions • Construct word equations to describe what is happening in each reaction


Checklist Ø Line 17 - 3/5 - not balancing equations or theories and laws Ø Line 18 - 3/5 - Experiment combining solutions. Ø Line 19 - 20/5 - Precipitation

What is a Word Equation? • Chemical Formula (number of elements in molecule): Ag. NO 3 (silver nitrate) • Chemical equation (Formulas to show chemical reactions): Ag. NO 3 + Na. Cl -> Ag. Cl + Na. No 3 • Word equation (Chemical names to show chemical reactions): silver sodium nitrate + chloride -> silver chloride + sodium nitrate

Reactants & Products • Reactants = The elements or molecules used as the input to a chemical reaction. Products = the elements or molecules that are the output of the chemical reaction. • Example: Ag. NO 3 + Na. Cl -> Ag. Cl + Na. No 3 (REACTANTS) (PRODUCTS) • OR: silver sodium nitrate + chloride (REACTANTS) -> silver chloride + sodium nitrate (PRODUCTS)

What is a Word Equation? • Chemical Formula (number of elements in molecule): Ag. NO 3 (silver nitrate) • Chemical equation (Formulas to show chemical reactions): Ag. NO 3 + Na. Cl -> Ag. Cl + Na. No 3 • Word equation: silver sodium silver nitrate + chloride -> Reactants -> sodium chloride + nitrate Products