GRAVIMETRY prof Viktor Kanick Analytick chemie I 1























![Factors influencing solubility n example: Ag. Cl, solub. compl. [Ag. Cl 2]-, [Ag. Cl Factors influencing solubility n example: Ag. Cl, solub. compl. [Ag. Cl 2]-, [Ag. Cl](https://slidetodoc.com/presentation_image/5bb8aa2970f19973128f37c97778fa2b/image-26.jpg)



































- Slides: 68

GRAVIMETRY prof Viktor Kanický, Analytická chemie I 1

Gravimetry n n n Basic method, classical Constituent to be determined is transferred from a given, precise amount of a sample into a defined chemical individuum, whose mass determined by weighing is a measure of a content of the constituent in the sample Analytical weighing balances (scales) in principle isosceles lever + pendulum q q sensitivity accuracy prof Viktor Kanický, Analytická chemie I 2

Weighing q 1) accuracy – the same length of both scale beams (Gauss method of double weighing) Buoyancy force (reduction of weighing at vacuum) mx – real mass z – weight of the weight sx, sz – specific mass of mx , z σ – specific mass of air Σ = 0, 0012 g. cm-3, sz = 8, 4 g. cm-3 (brass) → for z = 1 0, 5 g of subst. ≈ buoyancy 0, 08 mg n prof Viktor Kanický, Analytická chemie I 3

Weighing n Other influencing factors: q q q air humidity: adsorption of H 2 O, time factor finger prints: tweezers, laboratory tongs hygroscopic substances: ground-glass weighing bottles electric charge: powdered non-conductive substances on dry glass weights (relative calibration – calbration standards) weighing procedure: to lock balances, opening… no overloding !!! prof Viktor Kanický, Analytická chemie I 4

Weighing q 2) sensitivity l – scale beam G – beam weight d - distance Analytical balances Capacity: The larges weight the balance is capable of weighing Accuracy: the extent to which a given measurement agrees with the standard value for the measurement. (i. e. given weight is 100. 000 g, weighed 100. 002 g, accuracy is 2 mg. q Calibration: to determine, check or rectify the graduations of balance. It is the comparison between the output of a scale or balance against a standard value. q Reproducibility: ability of scales to return to the same numeric result with repeated application of the same weight. (hysteresis) prof Viktor Kanický, Analytická chemie I 5

Weighing n n Hysteresis: property of load cells, and other weighing systems dependant on elastic materials, such as spring balances, resulting in different indication at the same load, depending upon the direction of approach to that load, i. e. whether it is approached by increasing the load or decreasing the load. Hysteresis error refers to the condition of repeatedly weighing the same object, but obtaining different readings on the numeric readout. Drift: is a progressive (continuously upward or continuously downward) change in the number displayed on the digital readout. The weight readings does not stabilize, or unstable readings with no weight applied. All analytical balances show some uncertainty. prof Viktor Kanický, Analytická chemie I 6

Weighing n n Precision: The extent to which a given set of measurements of the sample agree with their mean. Amount of agreement between repeated measurements of the same quantity. Also know as repeatability. A scale can be extremely precise, but not necessarily be accurate. Repeatability: refers to an instrument’s ability to consistently deliver the same weight reading for a given object, and to return to a zero reading after each weighing cycle. Test this by repeatedly weighing the same object. Count (digit): The smallest increment of weight which the digital display resolves. Also called "division. ” Divisions: The amount of increments a scale offers. prof Viktor Kanický, Analytická chemie I 7

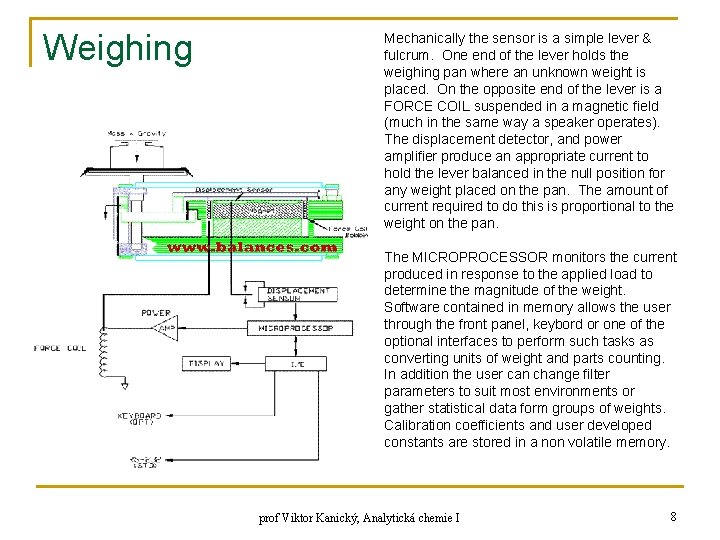
Weighing Mechanically the sensor is a simple lever & fulcrum. One end of the lever holds the weighing pan where an unknown weight is placed. On the opposite end of the lever is a FORCE COIL suspended in a magnetic field (much in the same way a speaker operates). The displacement detector, and power amplifier produce an appropriate current to hold the lever balanced in the null position for any weight placed on the pan. The amount of current required to do this is proportional to the weight on the pan. The MICROPROCESSOR monitors the current produced in response to the applied load to determine the magnitude of the weight. Software contained in memory allows the user through the front panel, keybord or one of the optional interfaces to perform such tasks as converting units of weight and parts counting. In addition the user can change filter parameters to suit most environments or gather statistical data form groups of weights. Calibration coefficients and user developed constants are stored in a non volatile memory. prof Viktor Kanický, Analytická chemie I 8

Precipitation n n classical separation technique for gravimetry preparation of pure compounds q q q insolubility of precipitate x loss (< 0, 1 mg) solubility of precipitate ≈ concentration of saturated solution above the precipitate Low soluble salts ≈ strong electrolytes ≈ complete dissociation ← unit activity of the solid phase is included in the constant = solubility product (thermodynamic value) prof Viktor Kanický, Analytická chemie I 9

Calculation of solubility of pure compounds uni-univalent electrolyte q It is valid in distilled water in the absence of other ions prof Viktor Kanický, Analytická chemie I 10

Calculation of solubility of pure compounds n n example: How many grams of Ag. Cl is contained in 1 l of saturated solution of Ag. Cl, M (Ag. Cl) = 143, 32 g. mol-1 example: What is the solubility of Ag 2 Cr. O 4 in H 2 O? prof Viktor Kanický, Analytická chemie I 11

Solubility n Factors influencing solubility q q q q own ions p. H side reactions complexation temperature solvent particle size ionic strength } prof Viktor Kanický, Analytická chemie I 12

Factors influencing solubility n 1) influence of own ions – excess of precipitatnt q M + B - a) precipitant excess: b) precipitant excess: q n generally example: by precipitating of Ag+ by excess of → washing the precipitate by diluted solution of precipitant, no water x large excess → (soluble) complexes formation → dissolution prof Viktor Kanický, Analytická chemie I 13

Factors influencing solubility n example: calculate concentration of SO 42 - necessary for quantitative precipitation of Ba. SO 4 (M (Ba. SO 4) = 233, 43 g. mol-1; Ks = 1, 08. 10 -10) n Condition: m (Ba. SO 4) in solution < 10 -4 g; V = 300 cm 3 prof Viktor Kanický, Analytická chemie I 14

Factors influencing solubility n 2) p. H and complex formation q side equilibria → soluble complexes (hydroxocomplexes of cations, protonization of anions) q solid phase dissolution q acids and bases increase solubility of precipitate prof Viktor Kanický, Analytická chemie I 15

Factors influencing solubility q q conditional solubility product c (M) c (B) α – side reaction coefficient prof Viktor Kanický, Analytická chemie I 16

Factors influencing solubility n example: diprotic acid: prof Viktor Kanický, Analytická chemie I 17

Factors influencing solubility n example: what is solubility of Ca. F 2 in 0, 01 M – HCl? prof Viktor Kanický, Analytická chemie I 18

Factors influencing solubility n example: calculate solubility of Ag. I v 0, 01 M NH 3 Solubility increases 40 x. prof Viktor Kanický, Analytická chemie I 19

Factors influencing solubility n example: calculate molar solubility of Ba. CO 3 at p. H = 6 and I = 0, 1 (p. Ks = 8, 09; p. K 1 = 6, 15; p. K 2 = 9, 99) prof Viktor Kanický, Analytická chemie I 20

Factors influencing solubility q Dissolution of Ba. CO 3 in H 2 O, influenced p. H, iteration 1) hydrolysis neglected → 2) we find p. H at hydrolysis of CO 32 a) hydrolytic constant b) we neglect for calculation of p. H, because prof Viktor Kanický, Analytická chemie I 21

Factors influencing solubility c) p. H of weak acid: 3) we find cond. solub. product. Ks´ and c (Ba. CO 3) 4) we repeat calc. p. H according 2 c) with c (CO 32 -) = 1, 48. 10 -4 we get p. H = 9, 90, Ks´ = 10 -7, 74, c (Ba. CO 3) = 1, 35. 10 -4 M further approximation yields c (Ba. CO 3) = 1, 32. 10 -4 M prof Viktor Kanický, Analytická chemie I 22

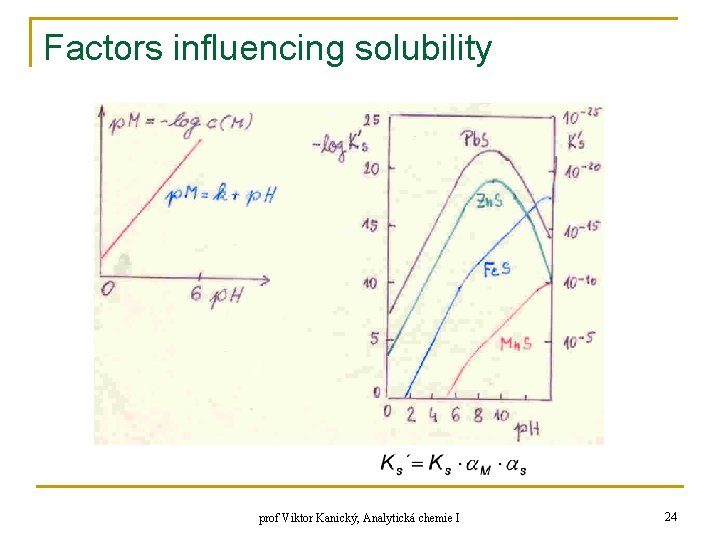
Factors influencing solubility q q q dependence of solubility of sulfides on p. H – principle of „sulfane“ separation of cations sulfide MS: boundary condition: p. H ≤ 6 – simplification → x (S) comprises only 1 term: K 1, K 2 – dissoc. const. H 2 S a HS-, hydrolysis of M is neglected prof Viktor Kanický, Analytická chemie I 23

Factors influencing solubility prof Viktor Kanický, Analytická chemie I 24

Factors influencing solubility q Influence of formation of complexes with own ions M – cation; B – anion, ligand; MB – weak soluble comp. soluble complexes (MB)r – soluble, non-dissociated portion of MB βMB – stability constant calculation of solubility MB in excess of precipitant prof Viktor Kanický, Analytická chemie I 25
![Factors influencing solubility n example Ag Cl solub compl Ag Cl 2 Ag Cl Factors influencing solubility n example: Ag. Cl, solub. compl. [Ag. Cl 2]-, [Ag. Cl](https://slidetodoc.com/presentation_image/5bb8aa2970f19973128f37c97778fa2b/image-26.jpg)
Factors influencing solubility n example: Ag. Cl, solub. compl. [Ag. Cl 2]-, [Ag. Cl 3]2 -, [Ag. Cl 4]3 Ks, β 1 β 2 β 3 β 4 prof Viktor Kanický, Analytická chemie I 26

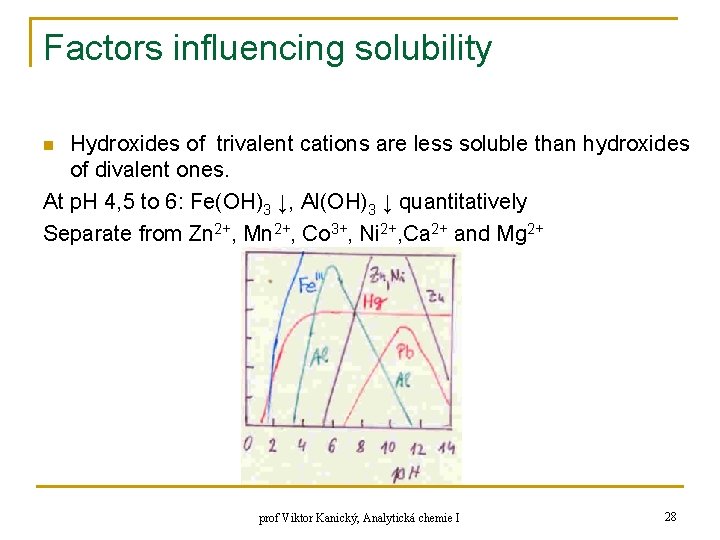
Factors influencing solubility q Dependence of solubility of hydroxides on p. H prof Viktor Kanický, Analytická chemie I 27

Factors influencing solubility Hydroxides of trivalent cations are less soluble than hydroxides of divalent ones. At p. H 4, 5 to 6: Fe(OH)3 ↓, Al(OH)3 ↓ quantitatively Separate from Zn 2+, Mn 2+, Co 3+, Ni 2+, Ca 2+ and Mg 2+ n prof Viktor Kanický, Analytická chemie I 28

Factors influencing solubility n 3) ionic strength (influence of third ions) q indifferent electrolyte 1) 2) 3) q solubility increases with increasing concentration of third ions prof Viktor Kanický, Analytická chemie I 29

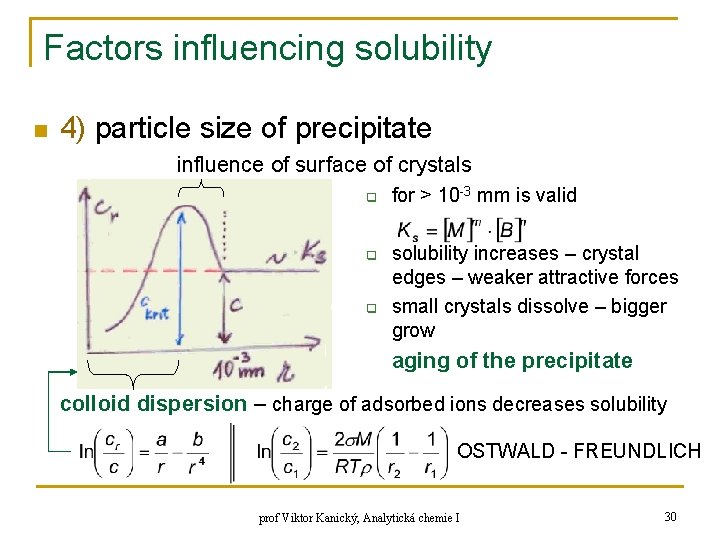
Factors influencing solubility n 4) particle size of precipitate influence of surface of crystals q q q for > 10 -3 mm is valid solubility increases – crystal edges – weaker attractive forces small crystals dissolve – bigger grow aging of the precipitate colloid dispersion – charge of adsorbed ions decreases solubility OSTWALD - FREUNDLICH prof Viktor Kanický, Analytická chemie I 30

Factors influencing solubility n 5) influence of solvent q q q Organic solvents decrease solubility of inorangic substances example: Ca. SO 4 in 50% Et. OH quantitatively precipitates Li. Cl soluble in amylalcohol, neither KCl a Na. Cl Influence increases with charge of ions prof Viktor Kanický, Analytická chemie I 31

Properties of precipitates n depend on : - chemical composition - properties and composition of medium in which precipitation occurs - method of precipitating n precipitate types: - colloidal (sulfur) - gel-like (Fe(OH)3) - lumpy (Ag. Cl) - crystalline (convenient, better filterable, more pure than amorphous): § § n fine (Ba. SO 4) coarse(Pb. Cl 2) requirements: filterability, easy drying, ignition, defined composition prof Viktor Kanický, Analytická chemie I 32

Properties of precipitates n mechanism: n big particles grow at the expense of small 1) formation of oversaturated solution (metastable) 2) formation of crystallization centers (cores, primary) 3) growth of paticles (aging) a) rate of formation of precipitate (Weimarn) c´ - instantaneous concentration of oversaturated solution c - solubility prof Viktor Kanický, Analytická chemie I 33

Properties of precipitates b) mean particle size depends on original concentration of solution (Weimarn) n n More soluble substances – bigger particles t – time – aging c) mean particle size increases with time of contact of precipitate with original solution. prof Viktor Kanický, Analytická chemie I 34

Properties of precipitates n aging: n n n less perferct → more perfect crystals Metastable modifications→ stable (aragonit → kalcit) change of number of crystal water moleules dehydratation (hydrat. oxidy Fe, Ti, Sn, Al, Zr, Th) polymeration (Co. S, Ni. S) prof Viktor Kanický, Analytická chemie I 35

Properties of precipitates q Colloidal dispersions (10 -5 – 10 -7 cm): Brown motion Tyndall effect q RTG → crystalline character n n q q large specific surface (S/V) adsorption ability existence of colloidal dispersions: repulsive elastic forces electric double layer → micelles example: change of sign of charge in isoelectric point x point of equivalence prof Viktor Kanický, Analytická chemie I 36

Properties of precipitates n coagulation q q q charges of the same sign on micelles x coagulation electrolyte excess disturbs a double-layer→ micelles coagulate coagulation increases with discharge of salt in washing solution (Al. Cl 3 >> NH 4 Cl, 1000 x) x ammonia salts preferred–removable at heating dispersion stability – H 2 O molecules binding → n n n lyophobic colloids – unstable (As 2 S 3, S, Au, Ag. X) lyophilic cololoid – stable (starch, gels, proteins, Al 2 O 3. x. H 2 O, Si. O 2. x. H 2 O) peptization – the opposite of coagulation q at filtration and washing with water – removing of electrolyte – unwanted effect! – therefore we wash with electrolyte soloution prof Viktor Kanický, Analytická chemie I 37

Contamination of precipitates q q n co-precipitation post-precippitation (induced preciptation) coprecipitation adsorption occlusion inclusion mixed crystals a) adsorption: depends on – conc. of adsorbed substances n - properties of adsorbed substances - properties of precipitate Paneth-Fajans: most adsorbed own ions and forming low soluble substances prof Viktor Kanický, Analytická chemie I 38

Coprecipitation q q q example: Ba. SO 4 in excess of Ba 2+: Br- < Cl. O 3 - < NO 3 Ba. SO 4 in excess of SO 42 -: Na+ < K+ < Ca 2+ < Pb 2+ easier adsorbed ions with higher charge Freundlich adsorption isotherm (T=const. ); k, n – const. ; x – amount /1 g precip. q q relatively highest adsorption of impurities occurs at lowest conc. of impurities contamination of precipitate is proportional to surface → coagulated colloidal dispersion x coarse crystalline precipitate prof Viktor Kanický, Analytická chemie I 39

Coprecipitation b) occlusion: mechanical stripping of extraneous components of solution at precipitating and growth of crystals around the impurity ≈ concentration of solution and ≈ rate of precipitation c) inclusion: mechanical closure of parent solution at crystal growth prof Viktor Kanický, Analytická chemie I 40

Coprecipitation d) mixed crystals: isomorphic substitution of ions at ∆rion < 10 -15 % and at the same crystal group = solid solutions preferred ions with the same electric charge numbers D – partition koef. ; c 1, c 2 – koncentration of isomorphic const. ; t – precip. ; r-solution it is not possible to purify by repeated precipitating with the same precipitant!!! D less depends on T(K), precip. rate, conc. prof Viktor Kanický, Analytická chemie I 41

Coprecipitation q types of contamination prof Viktor Kanický, Analytická chemie I 42

Postprecipitation n 1) 2) n postprecipitation– induced originally pure precipitate: after some time from oversaturated solution of Mg. Ox precipitates Mg. Ox it is therefore advisable to keep concentration ratios, and Ca. Ox soon be filtered off, dtto sulfides Klathrates: inside benzen prof Viktor Kanický, Analytická chemie I 43

Amorphous precipitates n Amorphous precipitates – by coagulating of colloids → gels q q n large specific surface → significant sorption coagulation by excess of electrolyte – source of contamination filtration – immediately after precip. to wash with electrolyte solution (peptization!!!) Minimizing of coprecipitation – by a suitable procedure prof Viktor Kanický, Analytická chemie I 44

Principles of precipitation n n precipitation from hot solutions – better formation of crystal lattice without third ions – contaminating ions; to cool before filtration in case of more soluble precipitates (Mg. NH 4 PO 4) precipitation from sufficiently diluted solutions; repeated precipitation (2 x) (hydroxides); diminuition of coprecipitation of cations on precip. A+B- by precipitating with B- and vice versa precipitating agent is added slowly at agitation x local increase of concentration; formation of coarse precipitate with small surface allow to settle before filtration– less occlusion x less postprecipitation! thorough washing (hot water, electrolyte), colloids!! Too much soluble substances by alcohol prof Viktor Kanický, Analytická chemie I 45

Precipitation procedure 1) solution of substance to be precipiated is adjusted according to instruction (p. H, temperature) and precipiptated with clear solution of preciptitating reagent 2) precipitation is carried out in 250 -400 ml beakers, sample volume is adjusted to ca 100 -200 ml 3) precipitation solution is added slowly from burette or pipette + stirring with glass rod 4) after the precipitate is settled the test for complete precipitation is performed prof Viktor Kanický, Analytická chemie I 46

Precipitation procedure n Precipitating from homogeneous medium: q q q precipitatin reagent occurs gradually and continuously by chemical reaction decomposition, hydrolysis example: precipitation of sulfides by thioacetamide (at hot, hydrolysis) sulfide precipitates better coagulate and less adsorb prof Viktor Kanický, Analytická chemie I 47

Precipitation procedure q example: precipitation of hydroxides (M 3+, M 4+) separation from M 2+ binding H+ prof Viktor Kanický, Analytická chemie I 48

Filtration n rate of filtration P – filtration area r – pore radius p 1 -p 2 hydrostatic pressure differecnce between both sides of filtration medium l – effective length of capillaries η – dynamic viskosity V – capillary volume t – time q filtration acceleration: n n increase P (folded filter, frit) p 1 -p 2 (exhausting, longer filer funnel stem) decreased η (by warming) prof Viktor Kanický, Analytická chemie I 49

Filtration n besides mechanical effect influenced by: n n n q q hydration adhesion adsorption elektrokinetic effect character of filtered substance hydration – swelling of lyophilic filtration material → filtration retardation (retained even smaller particles) adhesion – liquid in capillaries flows slower along capillary walls prof Viktor Kanický, Analytická chemie I 50

Filtration q q adsorption – precipitate particles adhere on capillary walls elektrokinetic effekct – potential difference between capillary wall and liquid → n n q negative charge of fitr. paper retains cations and positively charged particles of the precipitate paper retains negative particles after washing with HCl character of filtered substance – it creates another filtration layer – different capillarity n Gels – filter clogging, therefore larger filter porosity is required and filtration with exhausting is necessary (p 1 -p 2) prof Viktor Kanický, Analytická chemie I 51

Filtration n n quantitative paper filters (ash-free) pure cellulose, 0, 01 mg of ash Different colour codes n n n „red ribbon“ – medium fast filtration (amorphous Fe 2 O 3) „yellow ribbon“ (colloides, sulfides) „green ribbon“ – slow filtration, fine precipitates „blue ribbon“ – slow filtration, very fine crystalline precipitate, Ba. SO 4 „black ribbon“ - fast filtration, coarse precipitates „white ribbon“ – medium fast filtration, standard filter for many applications prof Viktor Kanický, Analytická chemie I 52

Filtration n > 10% solutions of acids or bases damages paper filters can not be used for filtration of substances that are reduced at ashing filtration crucibles q q q crystalline precipitates glass crucibles – only drying (to 200°C) porcelain crucibles – ignition (to 1200°C) prof Viktor Kanický, Analytická chemie I 53

Washing of precipitate n removal of residues of parent solution – decantation is efficient – washing in beaker after precipitation n using smaller volumes more times V 1 – filtrate volume retained by precipitate V 2 – washing volume cn – concentration of third ions after n-times washing prof Viktor Kanický, Analytická chemie I 54

Washing of precipitate n precipitate – ionex – it is necessary to substitute third ions by adsorption of ions of a washing solution electrolyte n crystalline precipitates – washing by solution of salt with common ion with precipitate n colloidal precipitates – washing by solution of electrolyte protecting from peptisation n ammonium salts – easily removable by ignition n water at last, alcohol for more soluble precipitates prof Viktor Kanický, Analytická chemie I 55

Drying n a) drying in open atmosphere n removal of excess moisture until equilibrium with the pressure of water vapor in the air at a given temperature q Freundlich adsorption isotherm y = a. xb – monomolecular layer of water a) non-hygroscopic substances b) hygroscopic substance (more layers of H 2 O) ad b) can not be dried in open atmosphere → it is necessary to decrease ambient pressure and increase temp. ; vacuum drying q Water vapor pressure of surface bound water is lower nižší, therefore 105 -110°C is needed prof Viktor Kanický, Analytická chemie I 56

Drying n b) in dessicator – dessicants n n chemically (P 2 O 5) By adsorption (silik. ) P 2 O 5 mg/l H 2 O in vapor 2. 10 -5 Mg(Cl. O 4)2 Ba. O KOH silica gel 5. 10 -4 (anhydron) – for elemental analysis 7. 10 -4 2. 10 -3 3. 10 -2 – retains water vapor up to 40 % of its mass addition of Co. Cl 2 – anhydrous blue → wet pink, regeneration 180 -200°C prof Viktor Kanický, Analytická chemie I 57

Drying q molecular sieves – silicates Al, Ca, K, Na q gas adsorption n n acid (CO 2, SO 2) ≈ Ca. O, KOH, Na. OH alkaline (NH 3) ≈ P 2 O 5, H 2 SO 4 q alcohol vapors ≈ Ca. Cl 2 q benzine, chloroform, ether ≈ paraffin prof Viktor Kanický, Analytická chemie I 58

Drying n c) at elevated temperature q q dryer oven to 180°C, ± 5°C control, 105 -110°C, also: loss of crystallization water volatilization: NH 3, CO 2, SO 2, ammonium salts at 150°C, volatile chlorides filtration paper can withstand 150°C drying to constant wight– if a precipitate is dried directly to the form suitable for weighing – repeat the drying and weighing until the weight change (0, 2 mg) prof Viktor Kanický, Analytická chemie I 59

Ignition n n transfer of precipitate to ignited state porcelain crucible + triangle with ceramic rollers (Pt- crucible) temperatures: Bunsen burner - porcel. 700 -800°C - Pt 850 -1000°C Teclu + 100°C x Buns. Mecker + 200°C x Buns. prof Viktor Kanický, Analytická chemie I 60

Principles and procedures of ignition 1) 2) drying and ignition of empty crucible to constant weight slowly (non-luminous flame), tongs filter into the crucible: a) b) 3) to dry at 70°C in drying oven or over burner to incinerate (charring, air, no fire!!!) ignition (500 -1000°C) – burner, muffle furnace; after cooling down to 100 -200°C → into dessicator; to constant weight (± 0, 2 mg) prof Viktor Kanický, Analytická chemie I 61

Types of gravimetric determinations a) without precipitation reagent – salts ignition, heavy metal salts with anions of volatile acids (NO 3 -, CO 32 -, SO 42 -) → Cu. O, Zn. O, Fe 2 O 3, Al 2 O 3, Cr 2 O 3, Bi 2 O 3, Sb 2 O 3, Sn. O 2, Ti. O 2, Zr. O 2) b) addition of precip. reagent : H 2 S, HCl, H 2 SO 4, NH 4 OH, Na 2 HPO 4, Ag. NO 3, Ba. Cl 2 oragnic reagents : 8 -hydroxyquinoline, anthranilic acid, oxalic acid prof Viktor Kanický, Analytická chemie I 62

Types of gravimetric determinations c) precipitation from homogeneous solution for precipitation of hydroxides (urea, carbonyldiamide, carbamide, diaminmethanal): for precipitation of oxalates, phosphates and sulphates – hydrolysis of esters: for precipitation of sulphides: thioacetamide acetamide prof Viktor Kanický, Analytická chemie I 63

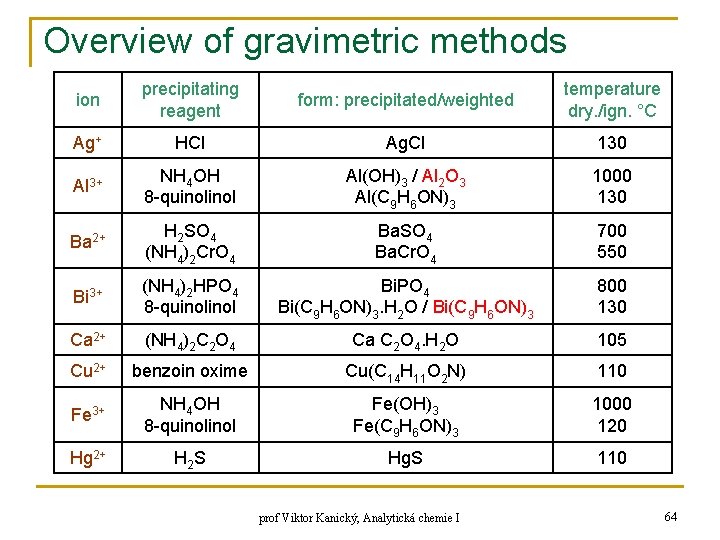
Overview of gravimetric methods ion precipitating reagent form: precipitated/weighted temperature dry. /ign. °C Ag+ HCl Ag. Cl 130 Al 3+ NH 4 OH 8 -quinol Al(OH)3 / Al 2 O 3 Al(C 9 H 6 ON)3 1000 130 Ba 2+ H 2 SO 4 (NH 4)2 Cr. O 4 Ba. SO 4 Ba. Cr. O 4 700 550 Bi 3+ (NH 4)2 HPO 4 8 -quinol Bi. PO 4 Bi(C 9 H 6 ON)3. H 2 O / Bi(C 9 H 6 ON)3 800 130 Ca 2+ (NH 4)2 C 2 O 4 Ca C 2 O 4. H 2 O 105 Cu 2+ benzoin oxime Cu(C 14 H 11 O 2 N) 110 Fe 3+ NH 4 OH 8 -quinol Fe(OH)3 Fe(C 9 H 6 ON)3 1000 120 Hg 2+ H 2 S Hg. S 110 prof Viktor Kanický, Analytická chemie I 64

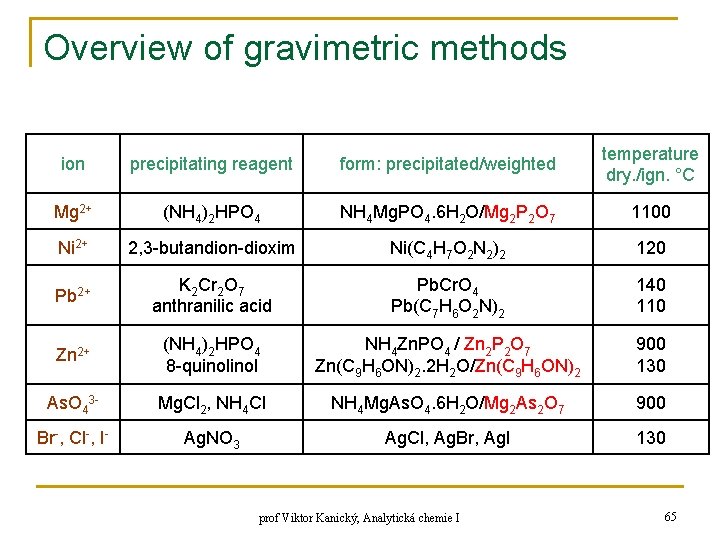
Overview of gravimetric methods ion precipitating reagent form: precipitated/weighted temperature dry. /ign. °C Mg 2+ (NH 4)2 HPO 4 NH 4 Mg. PO 4. 6 H 2 O/Mg 2 P 2 O 7 1100 Ni 2+ 2, 3 -butandion-dioxim Ni(C 4 H 7 O 2 N 2)2 120 Pb 2+ K 2 Cr 2 O 7 anthranilic acid Pb. Cr. O 4 Pb(C 7 H 6 O 2 N)2 140 110 Zn 2+ (NH 4)2 HPO 4 8 -quinol NH 4 Zn. PO 4 / Zn 2 P 2 O 7 Zn(C 9 H 6 ON)2. 2 H 2 O/Zn(C 9 H 6 ON)2 900 130 As. O 43 - Mg. Cl 2, NH 4 Cl NH 4 Mg. As. O 4. 6 H 2 O/Mg 2 As 2 O 7 900 Br-, Cl-, I- Ag. NO 3 Ag. Cl, Ag. Br, Ag. I 130 prof Viktor Kanický, Analytická chemie I 65

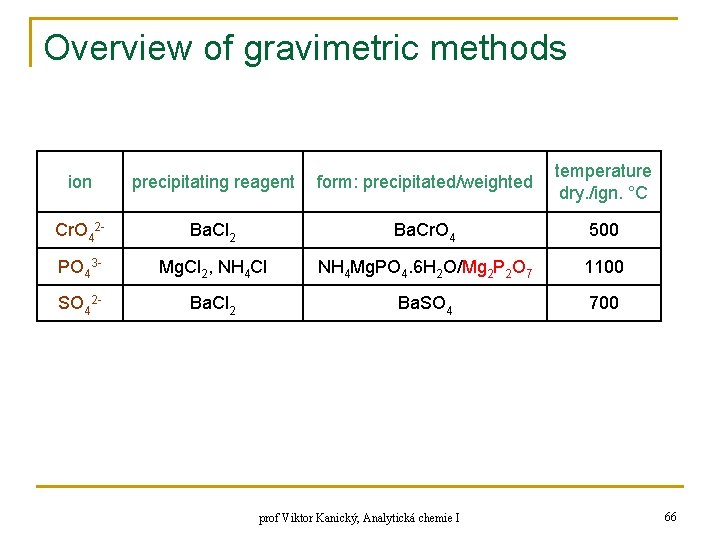
Overview of gravimetric methods ion precipitating reagent form: precipitated/weighted temperature dry. /ign. °C Cr. O 42 - Ba. Cl 2 Ba. Cr. O 4 500 PO 43 - Mg. Cl 2, NH 4 Cl NH 4 Mg. PO 4. 6 H 2 O/Mg 2 P 2 O 7 1100 SO 42 - Ba. Cl 2 Ba. SO 4 700 prof Viktor Kanický, Analytická chemie I 66

Calculation of gravimetric analysis n gravimetric factor– what is < 1, the error is smaller q ex. a (g) sample → b (g) Ag. Cl, chlorides content is prof Viktor Kanický, Analytická chemie I 67

Calculation of gravimetric analysis n indirect determination of K+ and Na+ - mixture of chlorides prof Viktor Kanický, Analytická chemie I 68