Post it Pollutants are dangerous chemicals in the



















- Slides: 25

Post it Pollutants are dangerous chemicals in the air are called pollutants. Name as many air pollutants as you can from last lesson. Extension: Circle those which could lead to acid rain

Hydrocarbons and fossil fuels To develop an understanding of how air pollutants are produced

Grade criteria Grade A* - Generate combustion reactions using balance symbol equations. Grade A – Summarise combustion reactions using word equations. Grade B – Explain how atoms are conserved during combustion reactions. Grade C – Identify hydrocarbons and fossil fuels by their molecular pictures Grade D –Define the terms ‘hydrocarbon’ and ‘fossil fuels’, giving examples of each

Combustion, fuels and hydrocarbons When a substance burns, it is said to combust. Combustion is a rapid reaction between a substance and oxygen that releases heat and light energy. A fuel is a substance that reacts with oxygen (combusts) to release useful energy. Many fractions obtained from crude oil are used as fuels because they contain hydrocarbons that burn easily and release a large amount of useful energy.

Hydrocarbons – What’s so good about them? • • Contain carbon and hydrogen atoms only Can have different carbon chain lengths Length of the chain determines the use Crude oil is a mixture of lots and lots of different hydrocarbons.

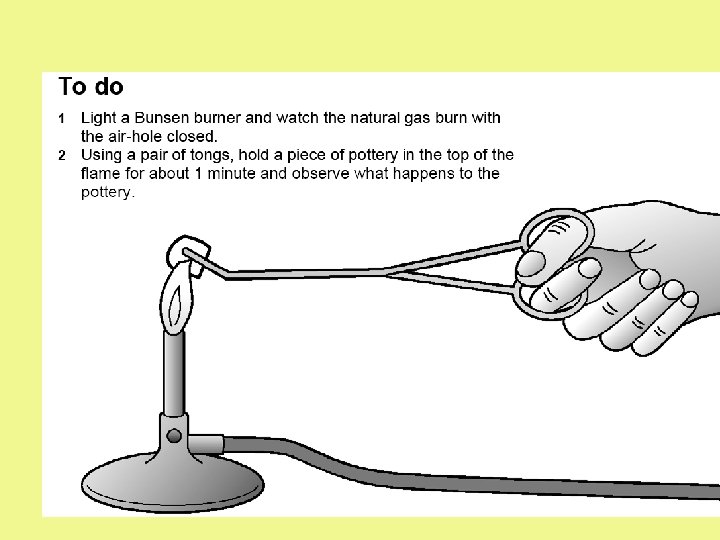
Complete combustion of hydrocarbons Plenty of air is needed to provide enough oxygen for a hydrocarbon fuel to burn completely. The blue flame of a gas hob or a Bunsen burner is an example of complete combustion of a hydrocarbon (in this case, natural gas). What are the products of the complete combustion of a hydrocarbon? carbon hydrocarbon + oxygen + water dioxide


Do you think that combustion is as simple as: carbon hydrocarbon + oxygen + water dioxide

VCOP – What are particulates? Consider your observations, which flame would it have been?

Grade criteria Grade A* - Generate combustion reactions using balance symbol equations. Grade A – Summarise combustion reactions using word equations. Grade B – Explain how atoms are conserved during combustion reactions. Grade C – Identify hydrocarbons and fossil fuels by their molecular pictures Grade D –Define the terms ‘hydrocarbon’ and ‘fossil fuels’, giving examples of each

Plenary: Methane bubbles • What is a hydrocarbon? Name an example • Give a general word equation for combustion • Name a product of incomplete combustion? How is it harmful? • Balance the equation

Homework • Complete pages 20 – 23/24/25

For each answer below write as many questions as you can think of I am the answer. . Hydrocarbon Acid rain Nitrogen Monoxide Combustion Carbon Dioxide + Water Crude oil So I am the Question

Explain why crude oil is an invaluable resource • Know the origins of crude oil • Describe the process to separate crude oil • Recognise the uses of the fractions of crude oil

What genre of chemicals do you get?

Hydrocarbons – What’s so good about them? • • Contain carbon and hydrogen atoms only Can have different carbon chain lengths Length of the chain determines the use Crude oil is a mixture of lots and lots of different hydrocarbons.

Crude Oil

How crude oil was formed • Microscopic plants and animals die and fall to the sea bed • Layers of sand mud form on top • Pressure and high temperature cause oil to form • Oil obtained by drilling

Explain why crude oil is an invaluable resource • Know the origins of crude oil • Describe the process to separate crude oil • Recognise the uses of the fractions of crude oil

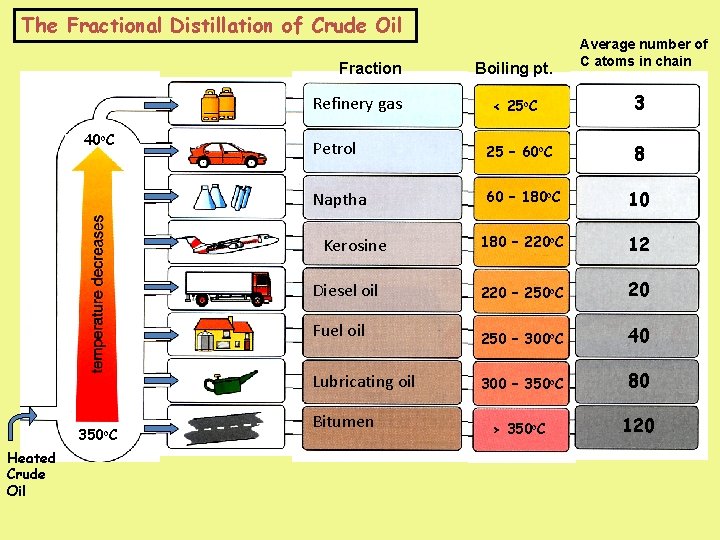
The Fractional Distillation of Crude Oil Fraction Refinery gas 40 o. C 3 25 – 60 o. C Naptha 60 – 180 o. C 10 180 – 220 o. C 12 220 – 250 o. C 20 250 – 300 o. C 40 300 – 350 o. C 80 > 350 o. C 120 Diesel oil Fuel oil Lubricating oil Heated Crude Oil < 25 o. C Petrol Kerosine 350 o. C Boiling pt. Average number of C atoms in chain Bitumen 8

Explain why crude oil is an invaluable resource • Know the origins of crude oil • Describe the process to separate crude oil • Recognise the uses of the fractions of crude oil

The Molymod challenge • Rules Carbon atoms always make 4 bonds • Hydrogen atoms are smaller • Hydrogen atoms can only make one bond Make the molecule methane (1 Carbon to 4 Hydrogens) Make the molecule propane (3 carbons to ? Hydrogens) Make the molecule hexane (? Carbons to 14 Hydrogens)

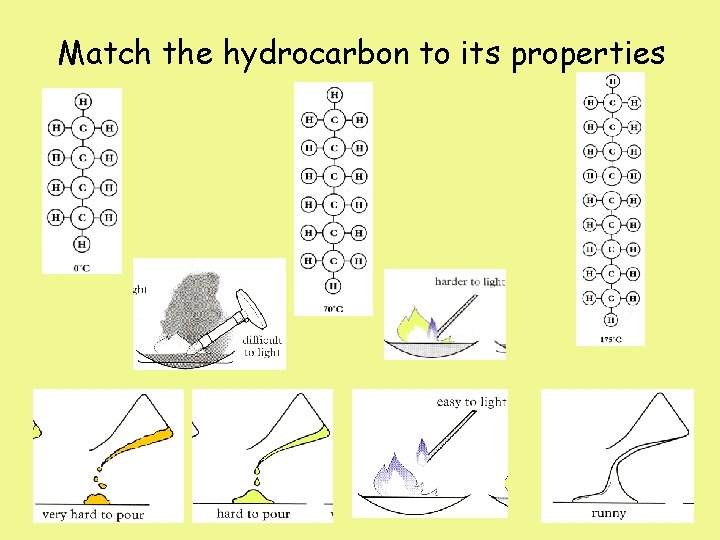
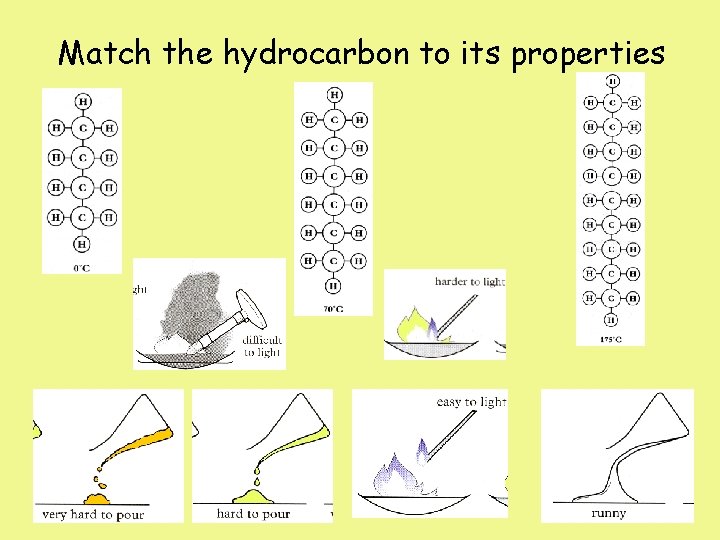
Match the hydrocarbon to its properties

Match the hydrocarbon to its properties

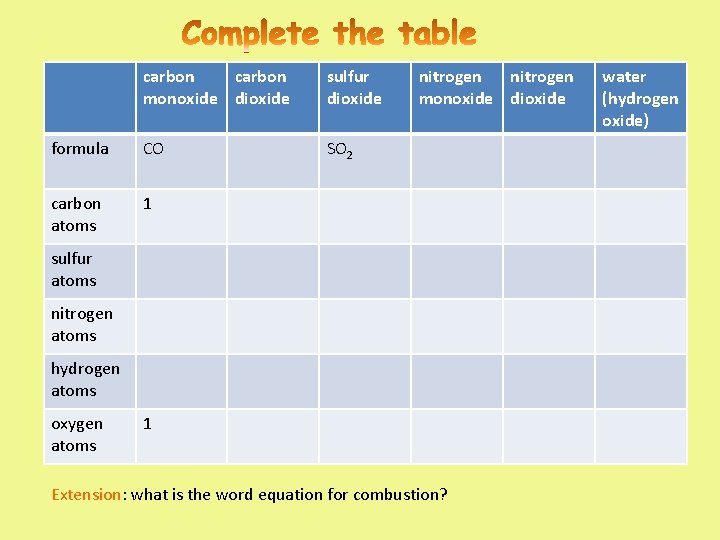
carbon monoxide dioxide sulfur dioxide formula CO SO 2 carbon atoms 1 nitrogen monoxide dioxide sulfur atoms nitrogen atoms hydrogen atoms oxygen atoms 1 Extension: what is the word equation for combustion? water (hydrogen oxide)