Its time to take care of TCell lymphomas











- Slides: 29

It’s time to take care of T-Cell lymphomas October 22 -24, 2006, Bologna, Italy Conventional Treatment of PTCL: The Asian Perspective Kensei Tobinai, MD, Ph. D National Cancer Center Hospital, Tokyo, Japan

Topics of Today 1) Epidemiology of PTCL in Japan 2) JCOG Studies and Allogeneic SCT for ATL 3) CCR 4 Expression in ATL & PTCL-U and a Monoclonal Antibody Treatment 4) Concurrent Chemoradiotherapy of Nasal NK/T-NHL

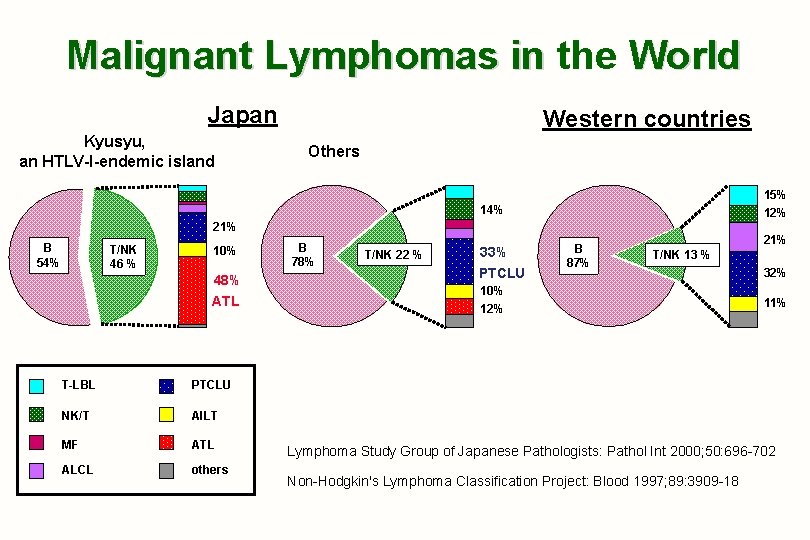
Lymphomas in Japan by WHO Classification 1. 3, 194 cases were reviewed, and consisted of 69% of B-NHL, 25% of T- or NK-NHL, and 4% of Hodgkin lymphoma. 2. Major subtypes of T- or NK-NHL in Japan 1) 7. 5% of adult T-cell leukemia-lymphoma (ATL) 19. 2% in Kyushu, a south-western island 2) 6. 7% of PTCL, unspecified 3) 2. 6% of NK/T-NHL 4) 2. 4% of angioimmunoblastic T-cell lymphoma (AILT) 5) 1. 7% of T-LBL/T-ALL 6) 1. 5% of ALCL Lymphoma Study Group of Japanese Pathologists:Pathol Int 2000; 50: 692 -702

Malignant Lymphomas in the World Japan Kyusyu, an HTLV-I-endemic island Western countries Others 15% 12% 14% 21% B 54% T/NK 46 % 10% 48% ATL T-LBL PTCLU NK/T AILT MF ATL ALCL others B 78% T/NK 22 % 33% PTCLU B 87% 21% T/NK 13 % 10% 12% 32% 11% Lymphoma Study Group of Japanese Pathologists: Pathol Int 2000; 50: 696 -702 Non-Hodgkin's Lymphoma Classification Project: Blood 1997; 89: 3909 -18

Diagnosis of ATL • Acute-type ATL (prototype) has characteristic findings, including flower cells in PB, hypercalcemia and frequent organ involvement (skin, GI-tract, lung, etc. ) • Peripheral T-cell phenotype (CD 4+, CD 8 -, CD 25+) • Presence of antibodies to HTLV-I in serum • Four clinical subtypes;

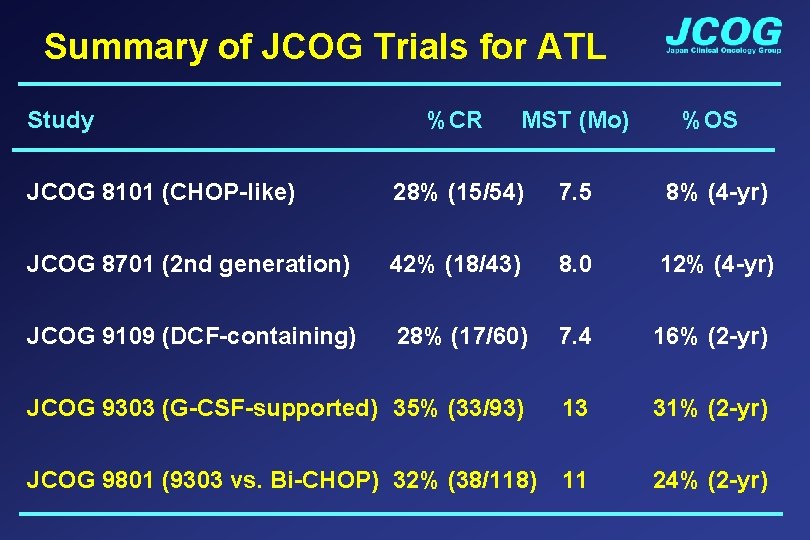
Summary of JCOG Trials for ATL Study %CR MST (Mo) %OS JCOG 8101 (CHOP-like) 28% (15/54) 7. 5 8% (4 -yr) JCOG 8701 (2 nd generation) 42% (18/43) 8. 0 12% (4 -yr) JCOG 9109 (DCF-containing) 28% (17/60) 7. 4 16% (2 -yr) JCOG 9303 (G-CSF-supported) 35% (33/93) 13 31% (2 -yr) JCOG 9801 (9303 vs. Bi-CHOP) 32% (38/118) 11 24% (2 -yr)

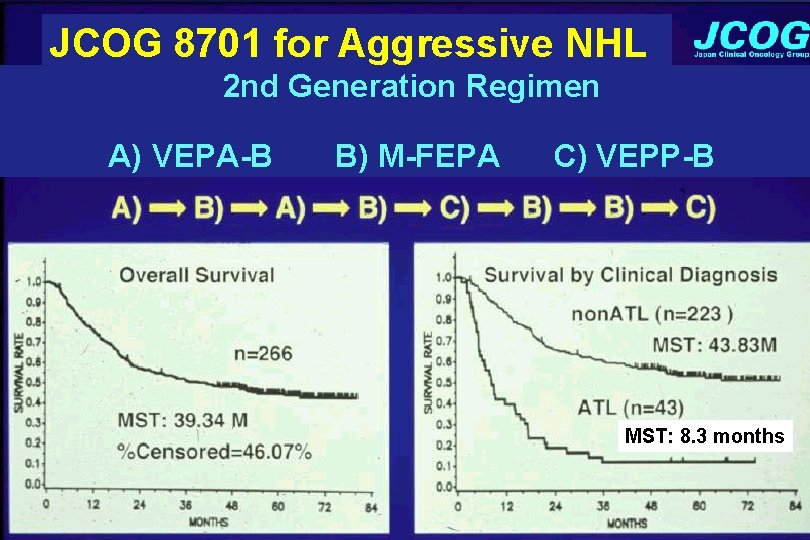
JCOG 8701 for Aggressive NHL 2 nd Generation Regimen A) VEPA-B B) M-FEPA C) VEPP-B MST: 8. 3 months

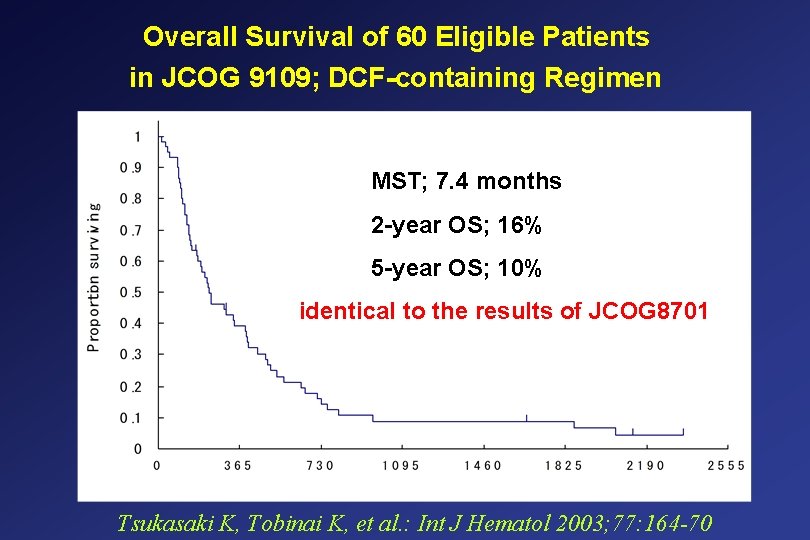
Overall Survival of 60 Eligible Patients in JCOG 9109; DCF-containing Regimen MST; 7. 4 months 2 -year OS; 16% 5 -year OS; 10% identical to the results of JCOG 8701 Tsukasaki K, Tobinai K, et al. : Int J Hematol 2003; 77: 164 -70

JCOG 9303; a G-CSF-supported, multiagent Day 1 VCR 1 mg/m 2 CPA 350 mg/m 2 DXR 40 mg/m 2 PSL 40 mg/m 2 MCNU 60 mg/m 2 VDS 2. 4 mg/m 2 ETP 100 mg/m 2 8 15 - 17 30 CBDCA 250 mg/m 2 IT-MTX+PSL on cycle 1, 3, 5 every 4 weeks for 7 cycles

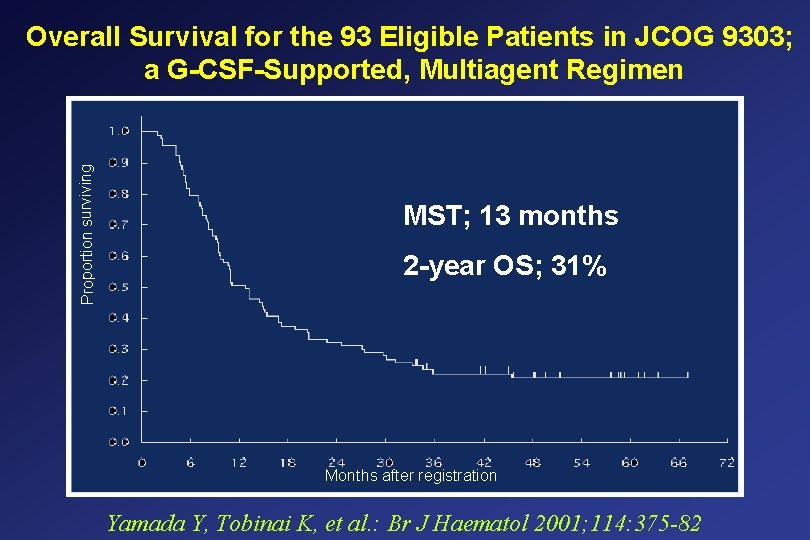
Proportion surviving Overall Survival for the 93 Eligible Patients in JCOG 9303; a G-CSF-Supported, Multiagent Regimen MST; 13 months 2 -year OS; 31% Months after registration Yamada Y, Tobinai K, et al. : Br J Haematol 2001; 114: 375 -82

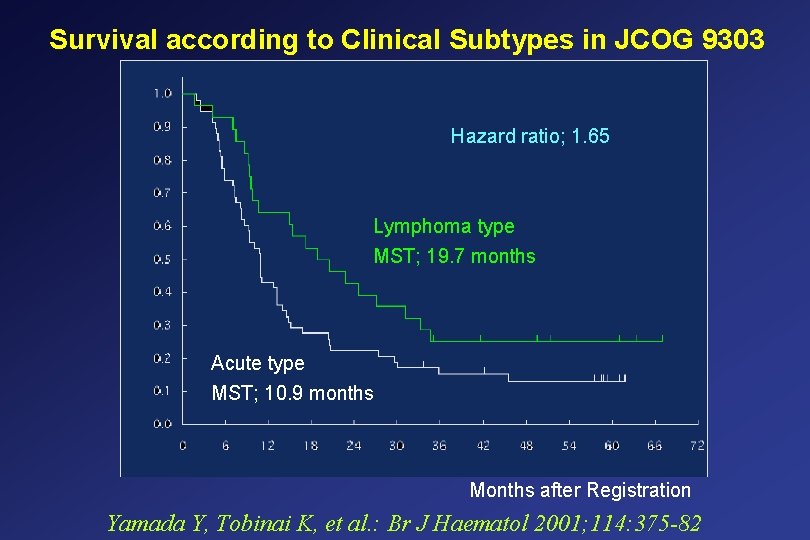
Survival according to Clinical Subtypes in JCOG 9303 Hazard ratio; 1. 65 Lymphoma type MST; 19. 7 months Acute type MST; 10. 9 months Months after Registration Yamada Y, Tobinai K, et al. : Br J Haematol 2001; 114: 375 -82

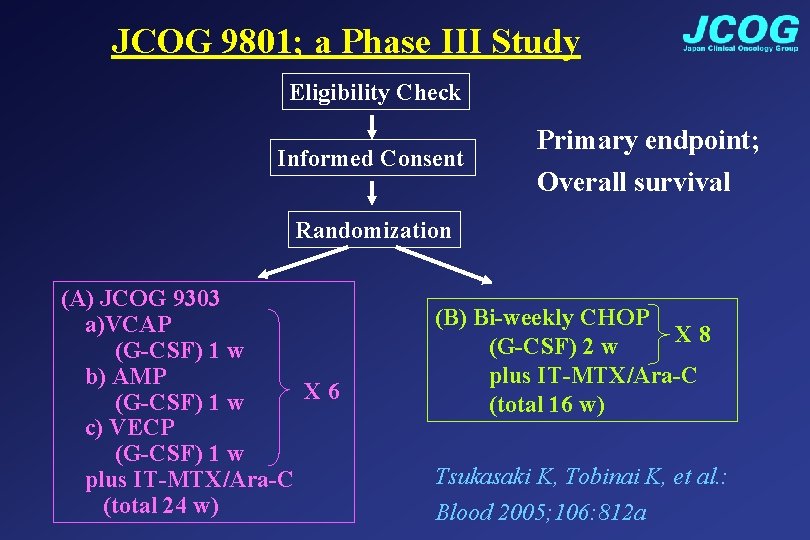
JCOG 9801; a Phase III Study Eligibility Check Informed Consent Primary endpoint; Overall survival Randomization (A) JCOG 9303 a)VCAP (G-CSF) 1 w b) AMP X 6 (G-CSF) 1 w c) VECP (G-CSF) 1 w plus IT-MTX/Ara-C (total 24 w) (B) Bi-weekly CHOP X 8 (G-CSF) 2 w plus IT-MTX/Ara-C (total 16 w) Tsukasaki K, Tobinai K, et al. : Blood 2005; 106: 812 a

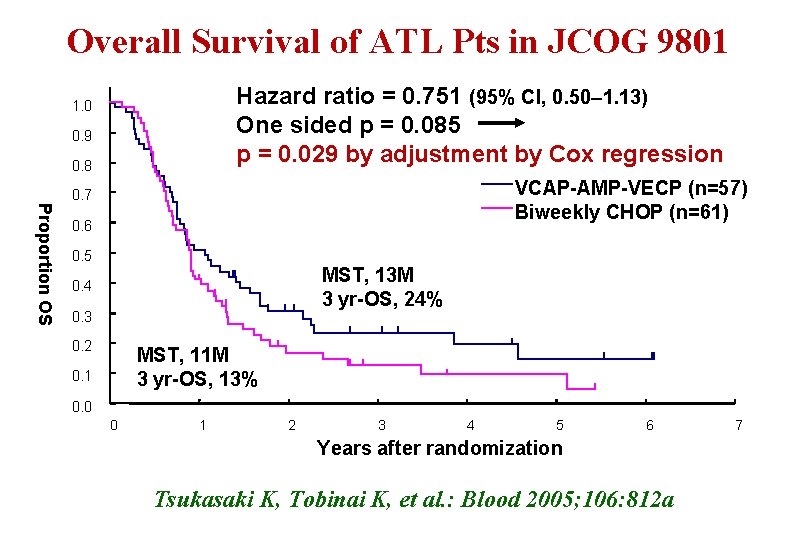
Overall Survival of ATL Pts in JCOG 9801 Hazard ratio = 0. 751 (95% CI, 0. 50– 1. 13) One sided p = 0. 085 p = 0. 029 by adjustment by Cox regression 1. 0 0. 9 0. 8 Proportion OS VCAP-AMP-VECP (n=57) Biweekly CHOP (n=61) 0. 7 0. 6 0. 5 MST, 13 M 3 yr-OS, 24% 0. 4 0. 3 0. 2 MST, 11 M 3 yr-OS, 13% 0. 1 0. 0 0 1 2 3 4 5 6 Years after randomization Tsukasaki K, Tobinai K, et al. : Blood 2005; 106: 812 a 7

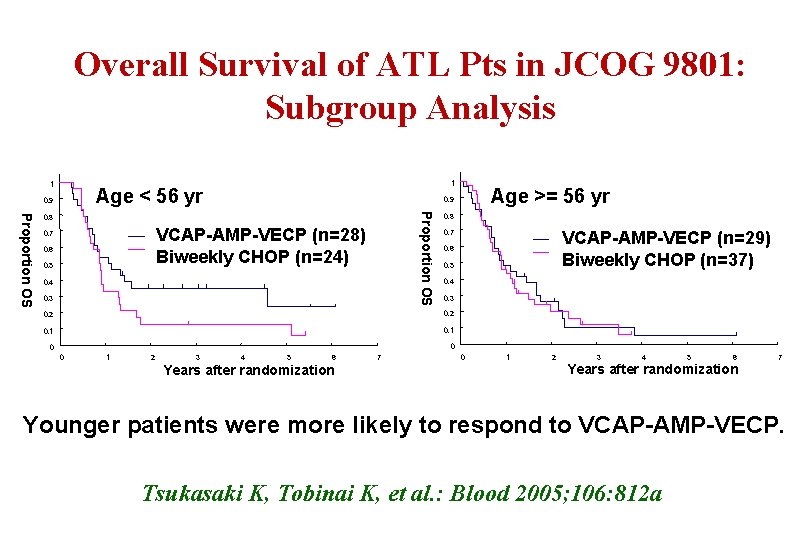
Overall Survival of ATL Pts in JCOG 9801: Subgroup Analysis 1 1 Age < 56 yr 0. 9 Proportion OS 0. 8 VCAP-AMP-VECP (n=28) Biweekly CHOP (n=24) 0. 7 0. 6 0. 5 Age >= 56 yr 0. 9 0. 4 0. 3 0. 8 0. 7 VCAP-AMP-VECP (n=29) Biweekly CHOP (n=37) 0. 6 0. 5 0. 4 0. 3 0. 2 0. 1 0 0 0 1 2 3 4 5 6 Years after randomization 7 0 1 2 3 4 5 6 7 Years after randomization Younger patients were more likely to respond to VCAP-AMP-VECP. Tsukasaki K, Tobinai K, et al. : Blood 2005; 106: 812 a

Conclusions of JCOG 9801 VCAP-AMP-VECP, a G-CSF-supported, dose-intensified multiagent regimen should be a new standard chemotherapy for newly diagnosed aggressive ATL. However, the MST of 13 months is still not satisfactory. Based on the promising results in retrospective analysis of myeloablative allo-SCT, we are planning a phase II study of the all-SCT after induction therapy with VCAP-AMP-VECP for young pts with aggressive ATL. Tsukasaki K, Tobinai K, et al. : Blood 2005; 106: 812 a

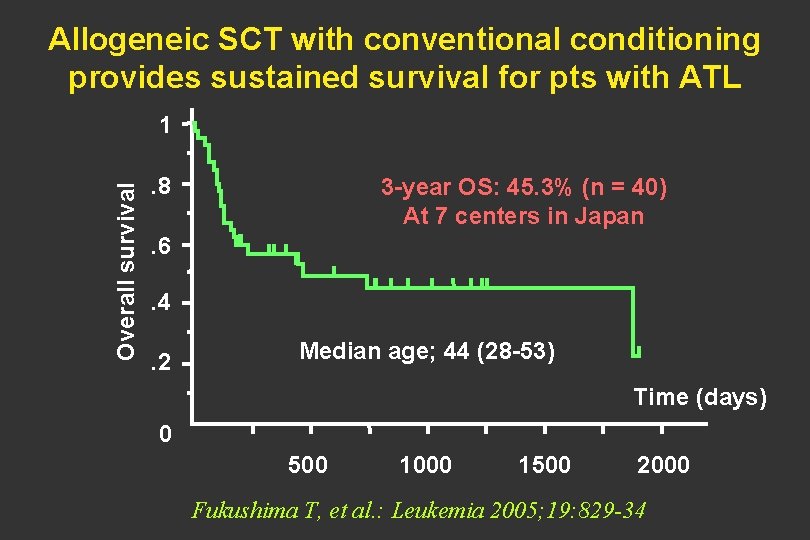
Allogeneic SCT with conventional conditioning provides sustained survival for pts with ATL Overall survival 1. 8 3 -year OS: 45. 3% (n = 40) At 7 centers in Japan . 6. 4. 2 Median age; 44 (28 -53) Time (days) 0 500 1000 1500 2000 Fukushima T, et al. : Leukemia 2005; 19: 829 -34

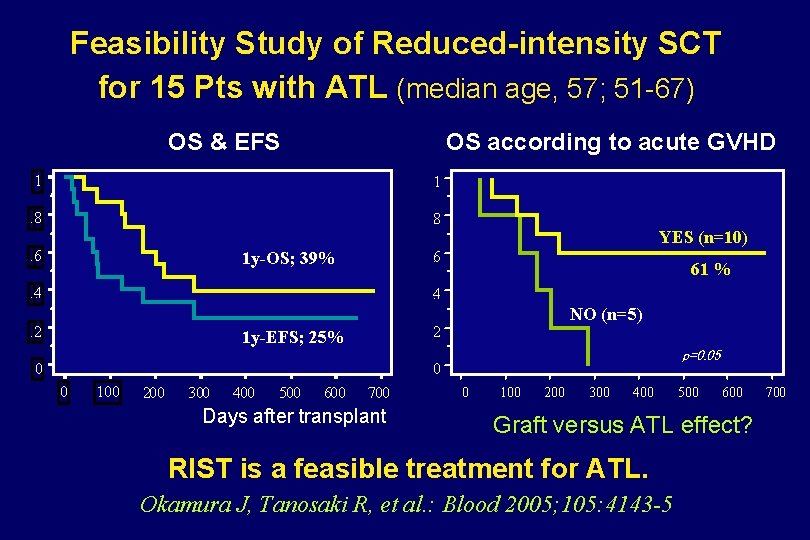
Feasibility Study of Reduced-intensity SCT for 15 Pts with ATL (median age, 57; 51 -67) OS & EFS OS according to acute GVHD 1 1 . 8. . 6. 1 y-OS; 39% YES (n=10) 6. . 4. 61 % 4. . 2. NO (n=5) 2. 1 y-EFS; 25% 0 p=0. 05 0 0 100 200 300 400 500 600 700 Days after transplant 0 100 200 300 400 500 600 Graft versus ATL effect? RIST is a feasible treatment for ATL. Okamura J, Tanosaki R, et al. : Blood 2005; 105: 4143 -5 700

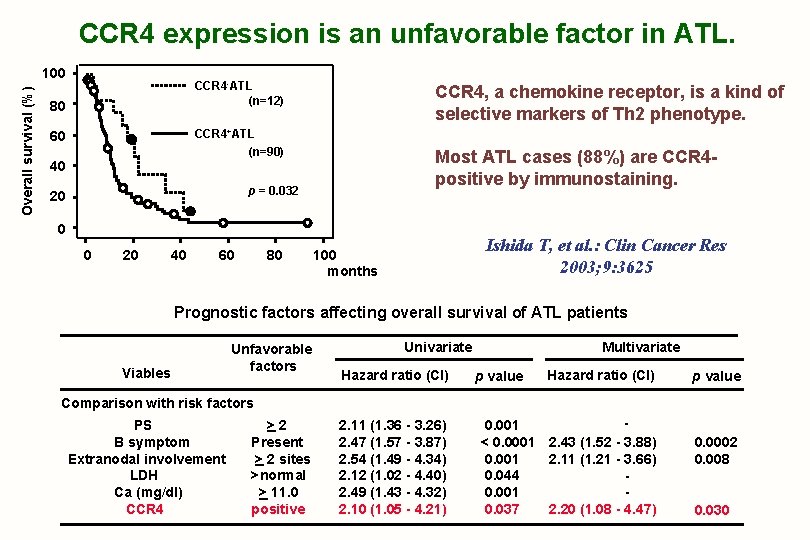
CCR 4 expression is an unfavorable factor in ATL. Overall survival (%) 100 CCR 4 -ATL (n=12) 80 CCR 4, a chemokine receptor, is a kind of selective markers of Th 2 phenotype. CCR 4+ATL 60 (n=90) 40 Most ATL cases (88%) are CCR 4 positive by immunostaining. p = 0. 032 20 0 0 20 40 60 80 Ishida T, et al. : Clin Cancer Res 2003; 9: 3625 100 months Prognostic factors affecting overall survival of ATL patients Viables Unfavorable factors Univariate Hazard ratio (CI) Multivariate p value Hazard ratio (CI) p value Comparison with risk factors PS B symptom Extranodal involvement LDH Ca (mg/dl) CCR 4 >2 Present > 2 sites >normal > 11. 0 positive 2. 11 (1. 36 - 3. 26) 2. 47 (1. 57 - 3. 87) 2. 54 (1. 49 - 4. 34) 2. 12 (1. 02 - 4. 40) 2. 49 (1. 43 - 4. 32) 2. 10 (1. 05 - 4. 21) 0. 001 < 0. 0001 2. 43 (1. 52 - 3. 88) 0. 001 2. 11 (1. 21 - 3. 66) 0. 044 0. 001 0. 037 2. 20 (1. 08 - 4. 47) 0. 0002 0. 008 0. 030

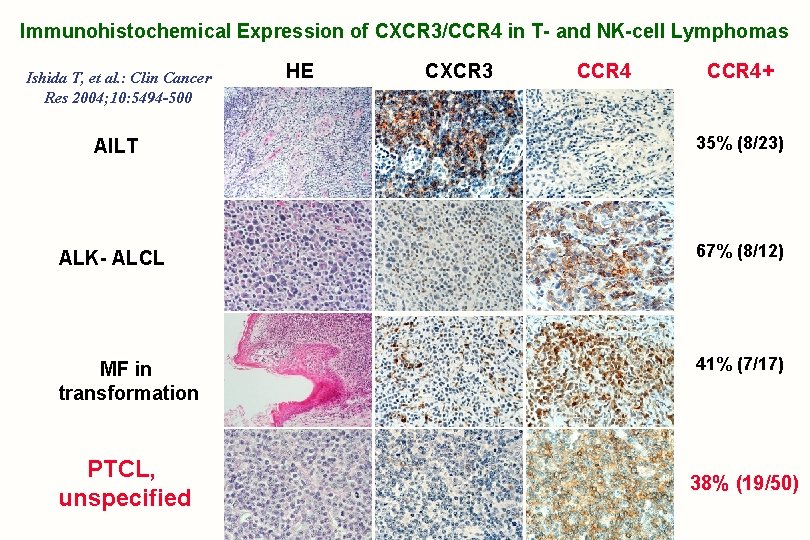
Immunohistochemical Expression of CXCR 3/CCR 4 in T- and NK-cell Lymphomas Ishida T, et al. : Clin Cancer Res 2004; 10: 5494 -500 HE CXCR 3 CCR 4+ AILT 35% (8/23) ALK- ALCL 67% (8/12) MF in transformation 41% (7/17) PTCL, unspecified 38% (19/50)

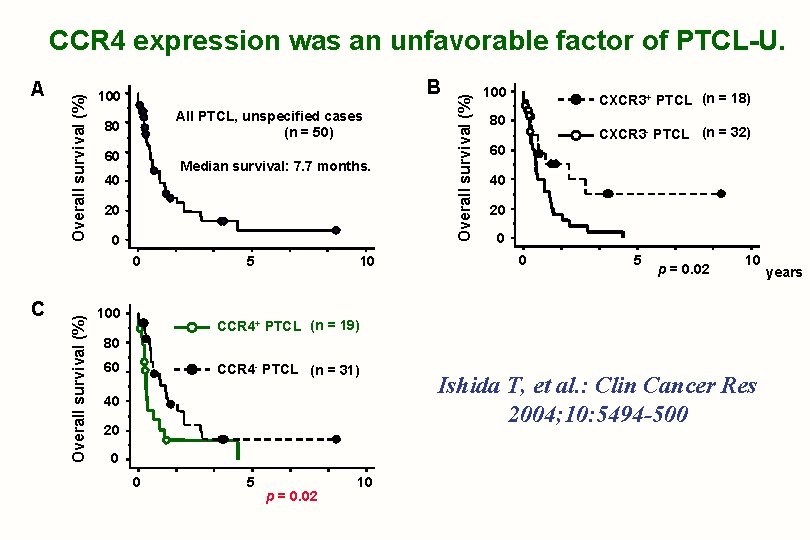
B 100 All PTCL, unspecified cases (n = 50) 80 60 Median survival: 7. 7 months. 40 20 0 C Overall survival (%) 0 100 5 10 Overall survival (%) A Overall survival (%) CCR 4 expression was an unfavorable factor of PTCL-U. 100 CXCR 3+ PTCL (n = 18) 80 CXCR 3 - PTCL (n = 32) 60 40 20 0 0 5 p = 0. 02 10 CCR 4+ PTCL (n = 19) 80 60 CCR 4 - PTCL (n = 31) 40 20 0 0 5 p = 0. 02 10 Ishida T, et al. : Clin Cancer Res 2004; 10: 5494 -500 years

Chemokine Receptors & PTCL 1) Chemokine receptors may be useful not only for further characterization of T- and NK-cell lymphomas, but also in predicting clinical outcomes. 2) Most ATL cases (88%) are CCR 4 -positive by immunostaining. CCR 4 expression is an unfavorable prognostic factor in ATL and PTCL-U. Ishida T, et al. : Clin Cancer Res 2004; 10: 5494 -500

Phase I Study of an ADCC-Enhanced, Humanized Anti-CCR 4 Antibody (KW-0761) in Patients with CCR 4 Positive Peripheral T-Cell Malignancies Pre-clinical studies of KW-0761 showed promising results for its clinical application to T-cell malignancies, especially to ATL. Patient enrolment will be initiated very soon in Japan.

Phase I/II Study of Concurrent Chemoradiotherapy for Untreated Localized Nasal NK/T-NHL (JCOG 0211 -DI) - ~ 36 patients / 3 years - Study design: phase I/II study - Endpoints Phase I primary endpoint; toxicity Phase II primary endpoint; 2 y-OS secondary endpoints; %CR, PFS, etc. Most pts with nasal NK/T-cell lymphoma have localized disease; however, a standard therapy for localized DLBCL such as CHOP followed by RT is unsuccessful in a large fraction of pts.

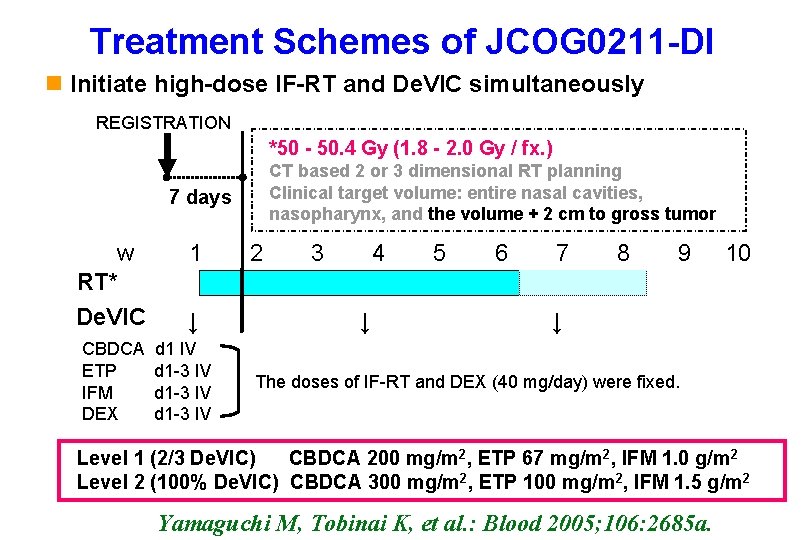
Treatment Schemes of JCOG 0211 -DI n Initiate high-dose IF-RT and De. VIC simultaneously REGISTRATION *50 - 50. 4 Gy (1. 8 - 2. 0 Gy / fx. ) 7 days w RT* De. VIC CBDCA ETP IFM DEX CT based 2 or 3 dimensional RT planning Clinical target volume: entire nasal cavities, nasopharynx, and the volume + 2 cm to gross tumor 1 2 3 4 5 6 7 8 9 ↓ ↓ d 1 IV d 1 -3 IV 10 ↓ The doses of IF-RT and DEX (40 mg/day) were fixed. Level 1 (2/3 De. VIC) CBDCA 200 mg/m 2, ETP 67 mg/m 2, IFM 1. 0 g/m 2 Level 2 (100% De. VIC) CBDCA 300 mg/m 2, ETP 100 mg/m 2, IFM 1. 5 g/m 2 Hematology Division, National Cancer Center Yamaguchi M, Tobinai K, et al. : Blood 2005; 106: 2685 a.

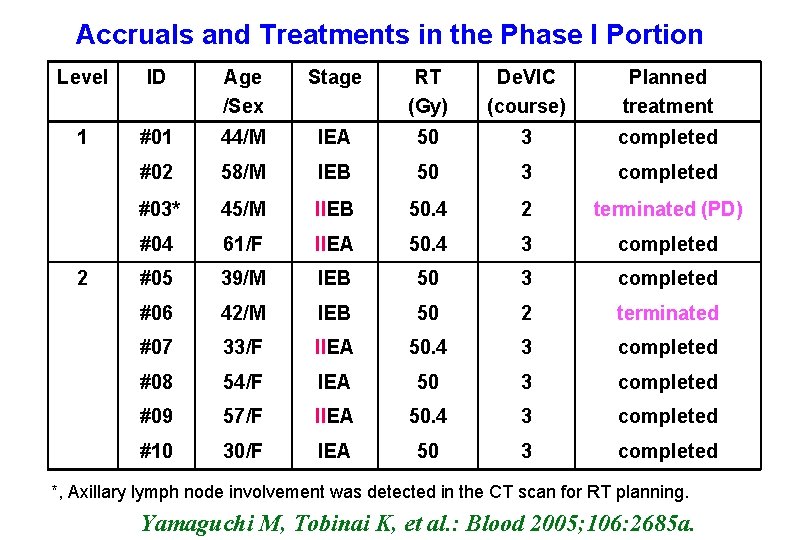
Accruals and Treatments in the Phase I Portion Level ID Age /Sex Stage RT (Gy) De. VIC (course) Planned treatment 1 #01 44/M IEA 50 3 completed #02 58/M IEB 50 3 completed #03* 45/M IIEB 50. 4 2 terminated (PD) #04 61/F IIEA 50. 4 3 completed #05 39/M IEB 50 3 completed #06 42/M IEB 50 2 terminated #07 33/F IIEA 50. 4 3 completed #08 54/F IEA 50 3 completed #09 57/F IIEA 50. 4 3 completed #10 30/F IEA 50 3 completed 2 *, Axillary lymph node involvement was detected in the CT scan for RT planning. Yamaguchi M, Tobinai K, et al. : Blood 2005; 106: 2685 a.

Assessment of DLTs in JCOG 0211 -DI ★ Criteria for DLT (NCI-CTC ver 2. 0) A Grade 4 leukopenia or neutropenia lasting for 5 days or more B Grade 4 thrombocytopenia C Non-hematologic toxicity of grade 3 or more, except for N/V or mucositis related to radiation DLT Level 1 Level 2 0 / 3* 4 / 6 #06 (stage IB) : #07 (stage IIA): #09 (stage IIA): #10 (stage IA) : RD for the phase II Criteria A, B, C (Infection) Criteria A, C (Infection, Weight loss) Criteria B, C (Infection) Criteria C (Keratitis) Yamaguchi M, Tobinai K, et al. : Blood 2005; 106: 2685 a.

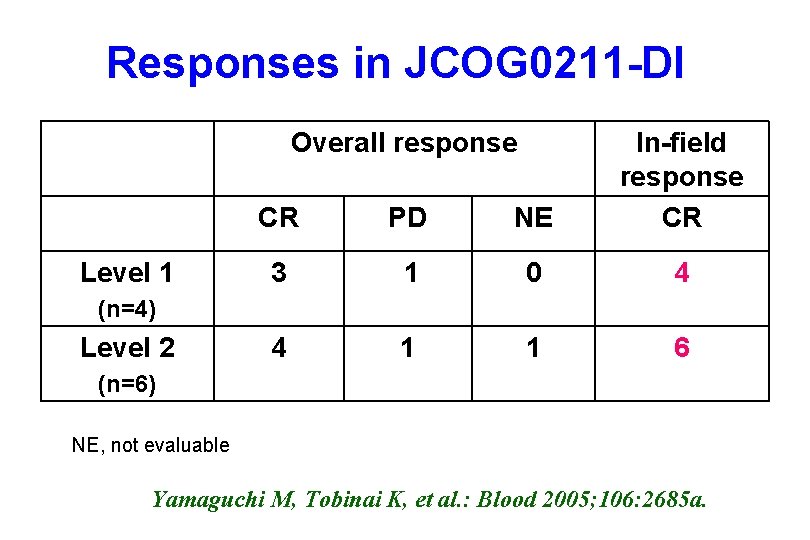
Responses in JCOG 0211 -DI Overall response Level 1 CR PD NE In-field response CR 3 1 0 4 4 1 1 6 (n=4) Level 2 (n=6) NE, not evaluable Yamaguchi M, Tobinai K, et al. : Blood 2005; 106: 2685 a.

Preliminary Conclusions of JCOG 0211 -DI Concurrent chemoradiotherapy using MDR-non-related agents and etoposide appears to be a promising treatment strategy for localized nasal NK/T-cell lymphoma. Feasibility and efficacy of Level 1 (2/3 De. VIC) will be further evaluated in the phase II portion. Patient enrolment into the phase II portion will be completed within a few months. Hematology Division, National Cancer Center Yamaguchi M, Tobinai K, et al. : Blood 2005; 106: 2685 a.

Acknowledgements • JCOG studies for ATL Tsukasaki K, Yamada Y, Fukuda H, Shimoyama M, et al. • Allogeneic SCT for ATL Fukushima T, Okamura J, et al. • CCR 4 expression in ATL & PTCL-U Ishida T, Ueda R, et al. • Phase I/II study for nasal NK/T-NHL Yamaguchi M, Oguchi M, et al.