ISOTOPES AND IMPURITIES IN WATER USED FOR WATER





- Slides: 14

ISOTOPES AND IMPURITIES IN WATER USED FOR WATER TRIPLE POINT CELLS EUROMET project 732 WP 3: Water Triple Point Cells 1 NMi - the Art of Measurement

WP 3: participants and contributors Participants: • • • Andrea Peruzzi (NMi VSL) Steffen Rudtsch (PTB) Miruna Dobre (SMD) Davor Zvidic (DZM-LPM) Aliye Kartal Dogan (UME) External Contributors: • Harro Meijer (CIO Univ. Groningen) • Detlev Schiel and Reinhard Jaehrling (PTB Braunschweig) • Ken Hill (NRC Ottawa) 2 NMi - the Art of Measurement

Summary • Water triple point cells manufactured at NMi VSL Isotopes: • • • Basic definitions to describe isotopic content of water Isotopic analysis of water samples Isotopic corrections Fractionation effects during fabrication process Temperature differences between the manufactured cells before and after applying the isotopic corrections Impurities: • • ICPMS analysis at PTB Overall Maximum Estimate (OME) method 3 NMi - the Art of Measurement

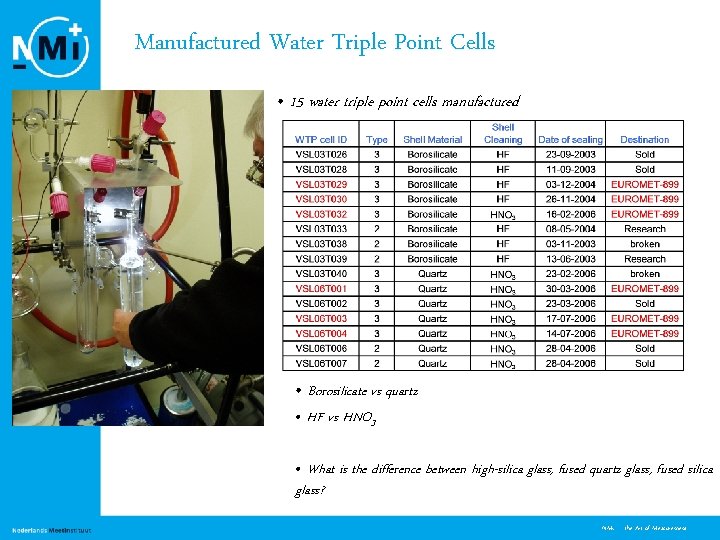
Manufactured Water Triple Point Cells • 15 water triple point cells manufactured • Borosilicate vs quartz • HF vs HNO 3 • What is the difference between high-silica glass, fused quartz glass, fused silica glass? 4 NMi - the Art of Measurement

Expressing the isotopic composition • Natural water is a mixture of the following 4 isotopologues: 1 H 16 O 1 H 17 O 1 H 18 O 2 2 2 • The triple point temperature of any sample of natural water depends on the relative concentrations of the 4 isotopologues • Ttp = 273. 16 K only for Vienna Standard Mean Ocean Water (VSMOW) : • In practice, the water contained in a WTPC always deviates from VSMOW. • The isotopic composition of a sample of natural water is described in terms of its departure from VSMOW (delta-values): 2 H 16 O 2 5 NMi - the Art of Measurement

Isotopic content of WTPCs • Samples taken at different stages while fabricating the cell Receiver water • Isotopic analysis of samples performed by CIO (Univ. Groningen) • 18 O and 2 H measured for each sample Starting water Bulb water u( u( = 0. 08 permil (k=1) 2 H) = 0. 8 permil (K=1) 18 O) 6 NMi - the Art of Measurement

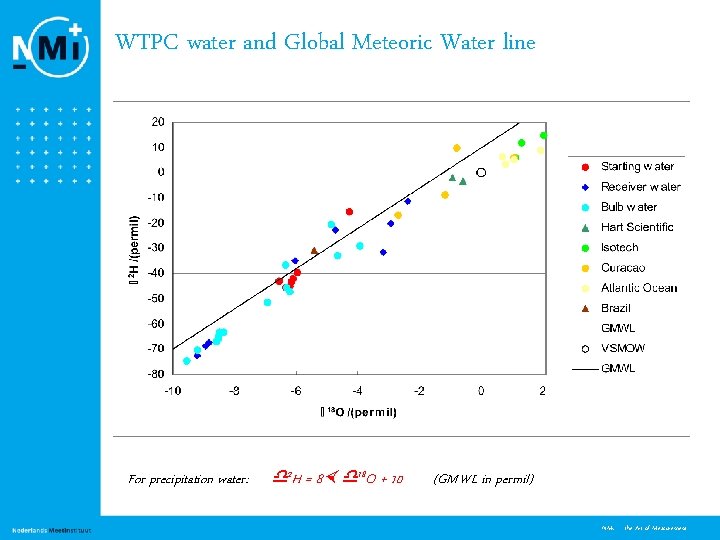
WTPC water and Global Meteoric Water line For precipitation water: 2 H = 8 18 O + 10 (GMWL in permil) 7 NMi - the Art of Measurement

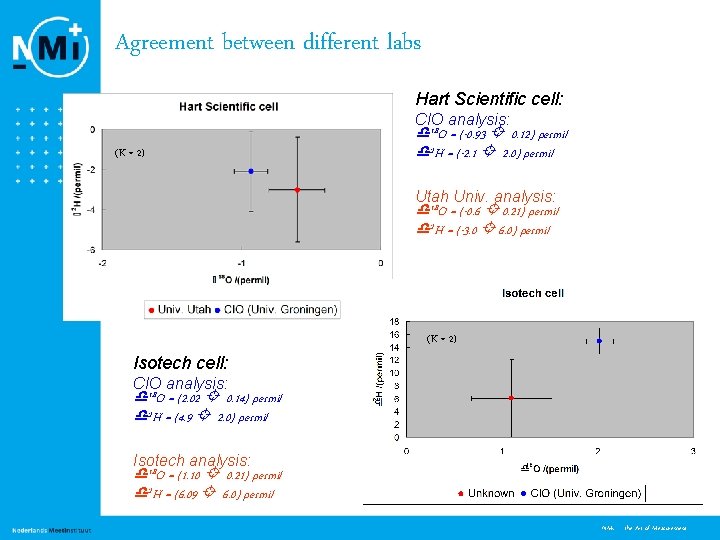
Agreement between different labs Hart Scientific cell: CIO analysis: (K = 2) 18 O = (-0. 93 0. 12) permil 2 H = (-2. 1 2. 0) permil Utah Univ. analysis: 18 O = (-0. 6 0. 21) permil 2 H = (-3. 0 6. 0) permil (K = 2) Isotech cell: CIO analysis: 18 O = (2. 02 0. 14) permil 2 H = (4. 9 2. 0) permil Isotech analysis: 18 O = (1. 10 0. 21) permil 2 H = (6. 09 6. 0) permil 8 NMi - the Art of Measurement

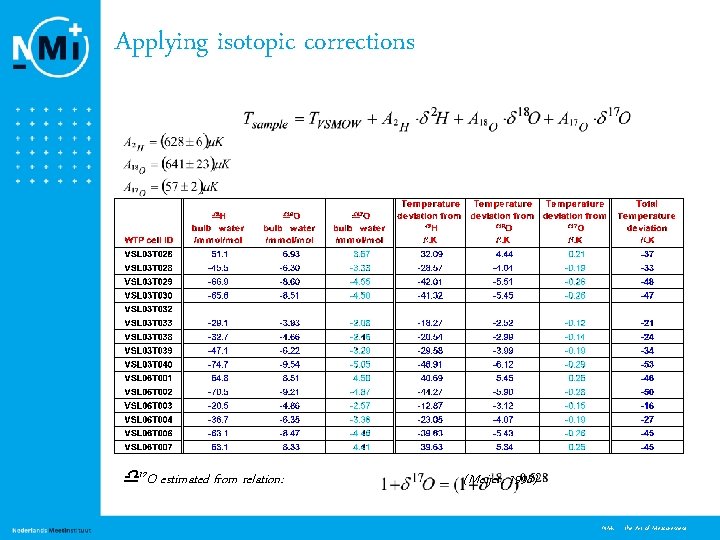
Applying isotopic corrections 17 O estimated from relation: (Meijer, 1998) 9 NMi - the Art of Measurement

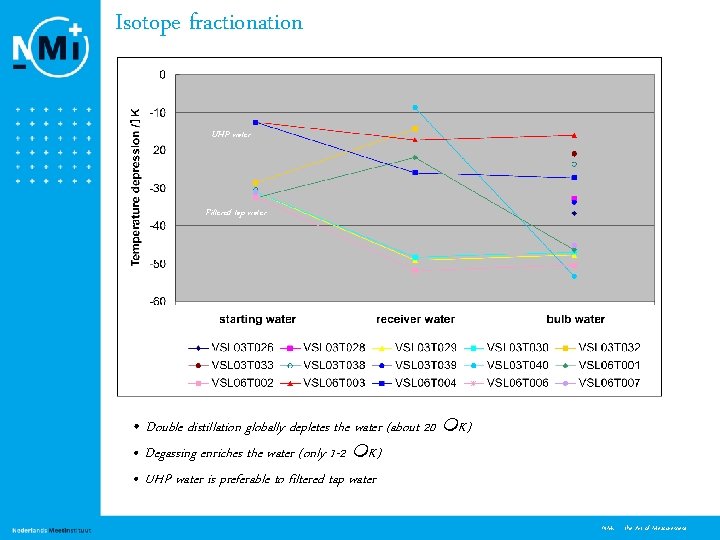
Isotope fractionation UHP water Filtered tap water • Double distillation globally depletes the water (about 20 K) • Degassing enriches the water (only 1 -2 K) • UHP water is preferable to filtered tap water 10 NMi - the Art of Measurement

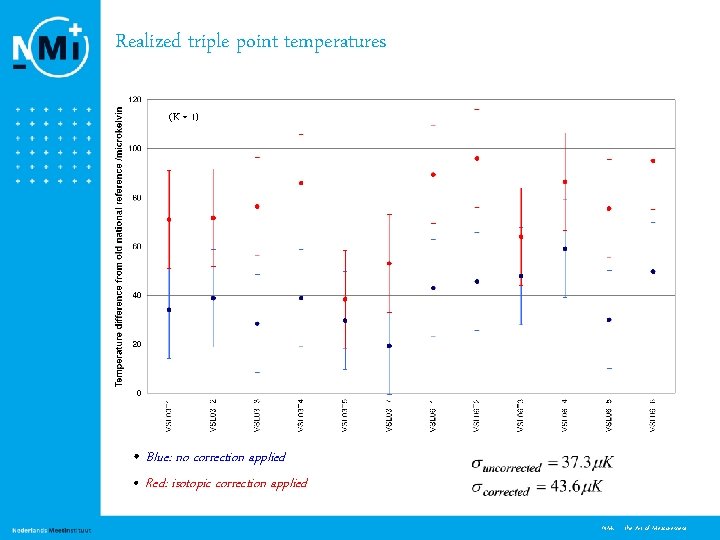
Realized triple point temperatures (K = 1) • Blue: no correction applied • Red: isotopic correction applied 11 NMi - the Art of Measurement

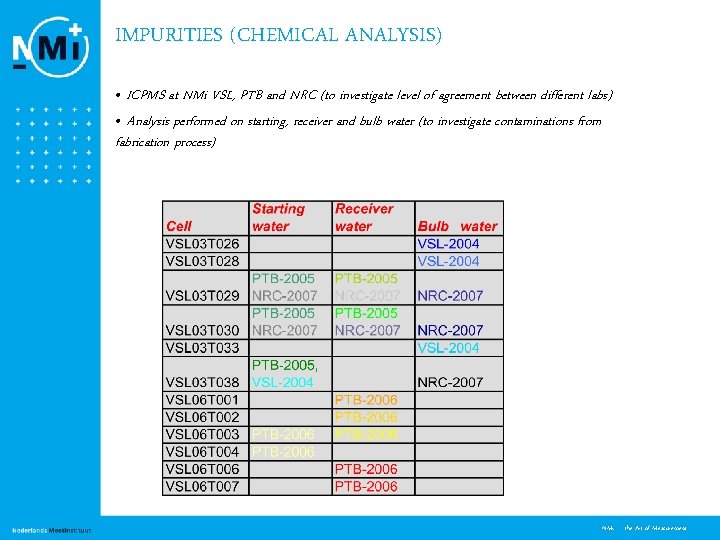
IMPURITIES (CHEMICAL ANALYSIS) • ICPMS at NMi VSL, PTB and NRC (to investigate level of agreement between different labs) • Analysis performed on starting, receiver and bulb water (to investigate contaminations from fabrication process) 12 NMi - the Art of Measurement

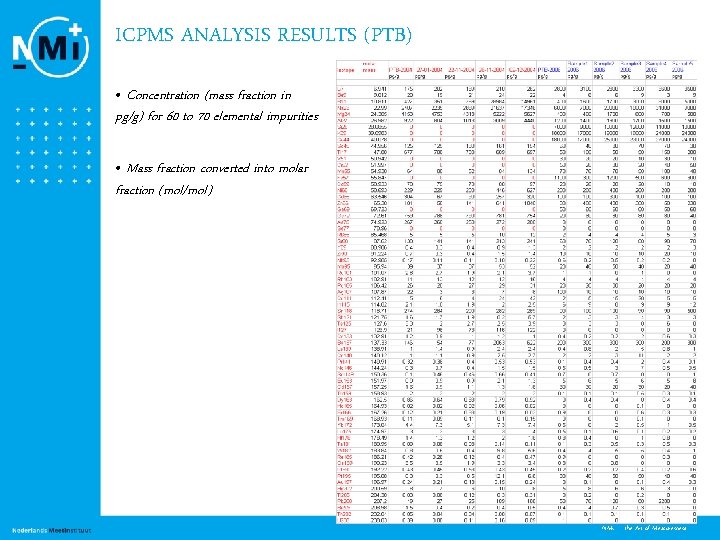
ICPMS ANALYSIS RESULTS (PTB) • Concentration (mass fraction in pg/g) for 60 to 70 elemental impurities • Mass fraction converted into molar fraction (mol/mol) 13 NMi - the Art of Measurement

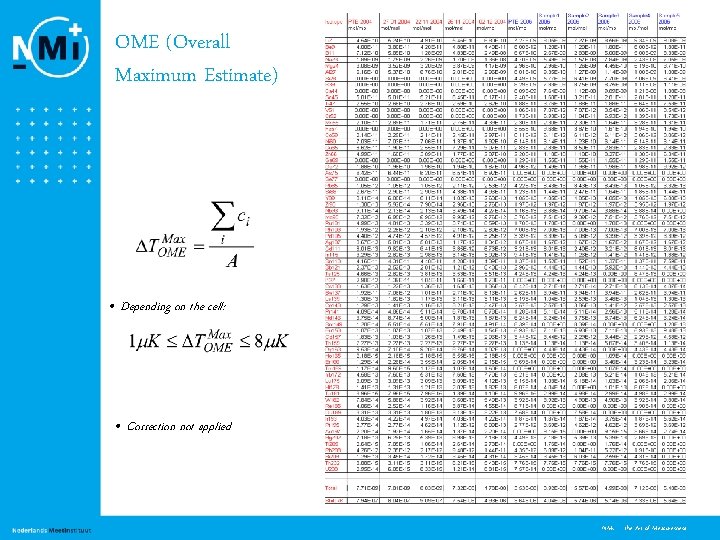
OME (Overall Maximum Estimate) • Depending on the cell: • Correction not applied 14 NMi - the Art of Measurement