Future HIV Prevention Trials Sexual Transmission Willard Cates



















- Slides: 24

Future HIV Prevention Trials: Sexual Transmission Willard Cates, Jr. , MD, MPH Family Health International 3 rd IAS Conference Rio de Janeiro July 26, 2005

Key Topics • HIV prevention tools • Current trials • Future opportunities • Future challenges

HIV Prevention Research - Why • Continued spread of HIV • Clear moral imperative to conduct prevention science

Prevention vs. Therapeutic Research: Contrasts Prevention Therapeutic • Subjects are relatively healthy, asymptomatic • Subjects have defined diseases or conditions • Low morbidity and mortality • Substantial morbidity/mortality • Long time frame • Short time frame • Sample sizes large • Sample sizes modest • Community base of research • Clinical base of research • Industry support is unusual due to no profitable market • Industry support is common due to profitable market

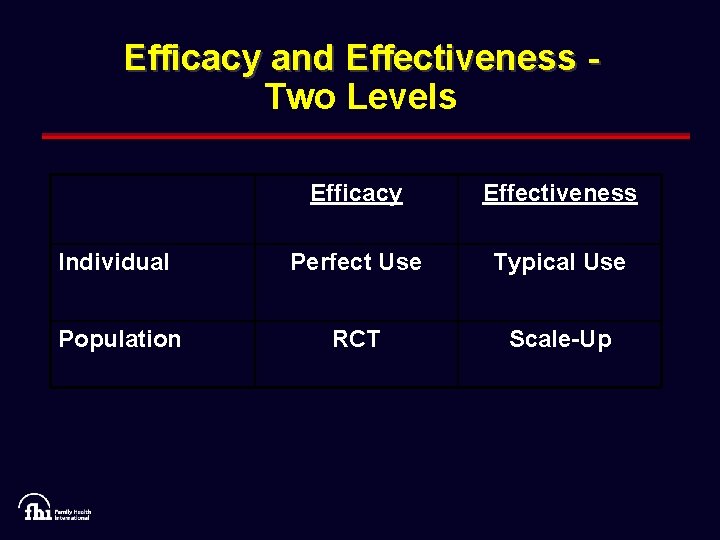
Efficacy and Effectiveness Two Levels Efficacy Effectiveness Individual Perfect Use Typical Use Population RCT Scale-Up

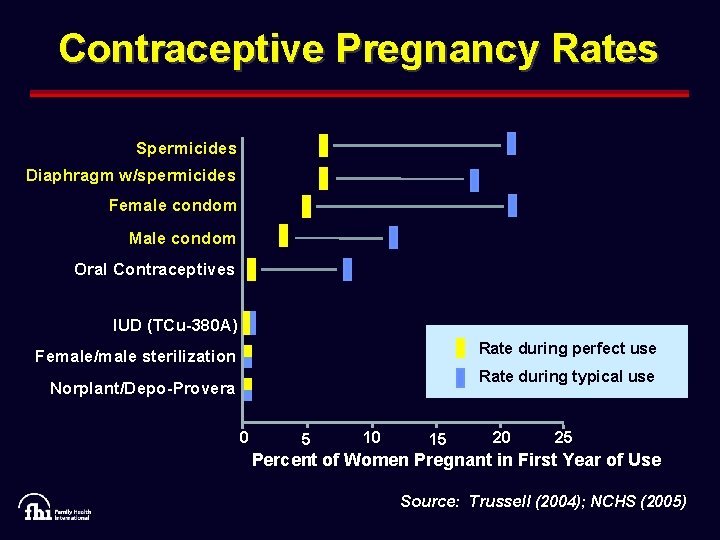
Contraceptive Pregnancy Rates Spermicides Diaphragm w/spermicides Female condom Male condom Oral Contraceptives IUD (TCu-380 A) Rate during perfect use Female/male sterilization Rate during typical use Norplant/Depo-Provera 0 5 10 15 20 25 Percent of Women Pregnant in First Year of Use Source: Trussell (2004); NCHS (2005)

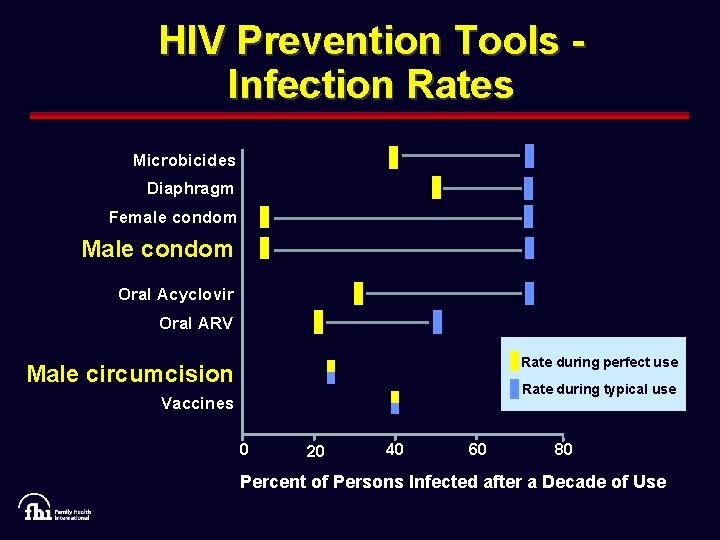
HIV Prevention Tools Infection Rates Microbicides Diaphragm Female condom Male condom Oral Acyclovir Oral ARV Rate during perfect use Male circumcision Rate during typical use Vaccines 0 20 40 60 80 Percent of Persons Infected after a Decade of Use

Topical Microbicides – 2 nd Generation Products – – – Buffergel * Pro 2000 C 31 G * Cellulose sulfate Carraguard * May also protect against pregnancy

Diaphragm • RCT in Zimbabwe, RSA • Diaphragm plus lubricant gel vs. condom • 4, 500 participants planned, 86% enrolled • Results expected 2007

Female Condoms • Biologic plausibility similar to male condoms • If made available, lowers overall level of unprotected acts • Emerging evidence (’ 05 ISSTDR) on STD prevention

Oral Acyclovir Prophylaxis • Acquisition – HSV+, HIV- subjects – HPTN 039 – USA and global – 3, 000 participants – MSM, MSW – Results expected 2007 • Transmission – HSV+, HIV+ subjects – Gates – US and global – 3, 600 HIV discordant couples – Results expected 2008

Oral ARV Pre-exposure Prophylaxis • Tenofovir collaboration – researchers and stakeholders • Focus for prevention trial ethics • Multiple populations globally • Estimated cumulative 4000 participants • Incremental results from summer 2006

ARV Treatment as Prevention • Viral load importance • HPTN 052 – 8 sites, 5 countries • Pilot phase initiated • Full trial – spring 2006, 1, 760 discordant couples planned

Male Circumcision • RSA data - encouraging – 70% protection – Data presented at IAS • Kisumu, Kenya – 2, 700 participants – Results expected 2007, DSMB driven • Rakai, Uganda – 5, 000 participants – Results expected 2007, DSMB driven

Future Directions • 3 rd generation microbicides • Newly designed barriers • Combination ARV pre-exposure • Acute HIV infection

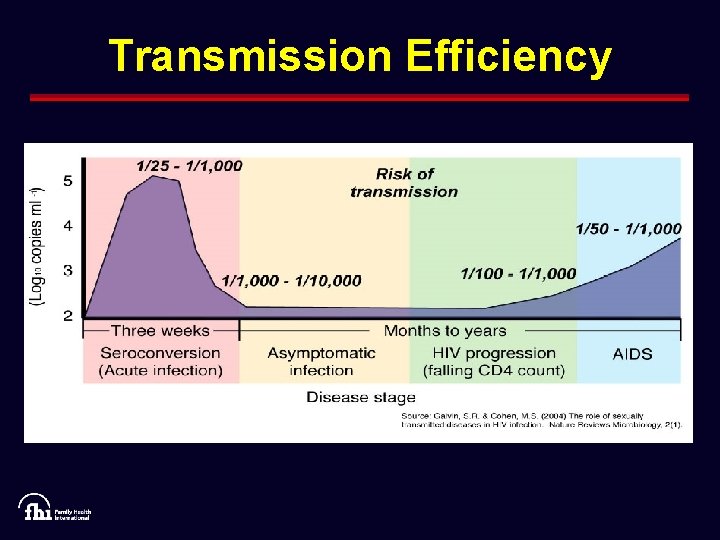
Transmission Efficiency

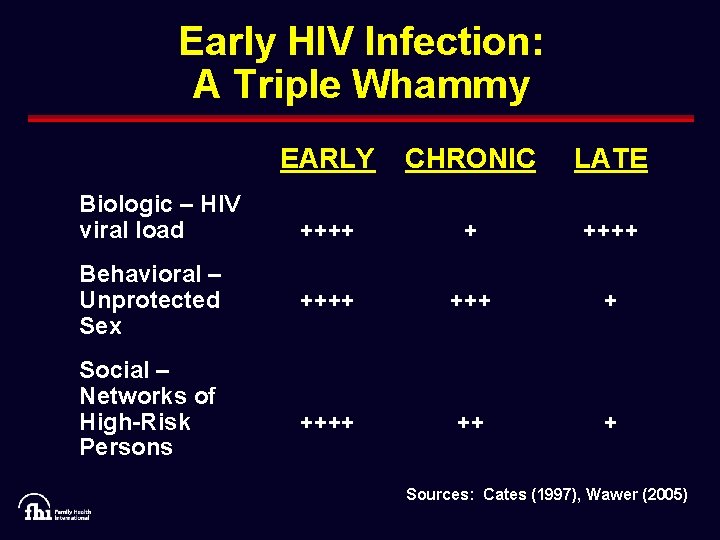
Early HIV Infection: A Triple Whammy Biologic – HIV viral load Behavioral – Unprotected Sex Social – Networks of High-Risk Persons EARLY CHRONIC LATE ++++ + ++++ ++ + Sources: Cates (1997), Wawer (2005)

Future Challenges • Participant care/treatment • Standards of preventive care • Community engagement • Product interruption • Additional study sites

Product Interruption – 3 Ps • Participants – adherence • Politics – Cambodia, Cameroon • Pregnancy – depends on – Coital frequency – Contraceptive use – Testing frequency/methods

Pregnancy Rates – Microb Prep (NIAID) Country Rates* • Durban • Hlabisa • Tanzania • Zambia 21 20 23 22 Population Moderate sex, General population Testing every month * Per 100 person years

Pregnancy Rates – Savvy Ghana (USAID) City Rates* Population • Accra • Kumasi 55 84 Frequent sex, High-risk women, Testing every month * Per 100 person years

HIV Prevention Trials – 2004 -2010 What Numbers Do We Need? Trials Dates Estimated Total Vaccine ’ 05 - ’ 10 124 k Non-Vaccine ’ 04 - ’ 10 96 k Total 220 M Partial Source: Wasserheit (2003)

Conclusion – HIV Prevention Trials, 2005 -2010 • Current Phase IIIs essential • More site capacity needed • Collaboration with other fields useful – vaccine, circumcision, STD rx, etc. • Shared resources helpful - GCP training - Laboratory assessment - Research staff - Ethics standards - Community involvement - Design challenges

Thanks To • Judy Auerbach • Nancy Padian • Michael Cassell • Renee Ridzon • Mike Cohen • Ken Schulz • Helene Gayle • James Trussell • Ron Gray • Sten Vermund • King Holmes • Judy Wasserheit • Cynthia Kay • Gary West