Classifying Chemical Reactions YEAR 10 CHEMISTRY Physical Chemical










- Slides: 11

Classifying Chemical Reactions YEAR 10 CHEMISTRY

Physical & Chemical Changes What is the difference between a physical and a chemical change? A physical change is one in which no new substance is formed, e. g. ice melting. A chemical change or chemical reaction has occurred if one or more new substances are formed. 2

PHYSICAL Changes shape Breaks into smaller pieces Dissolves Mixed with another substance Changes state No new substances formed CHEMICAL A permanent colour change A gas is given off Change in temperature Precipitate forms One metal is deposited on another New substances are formed

Word and formula equations Word equation hydrogen + oxygen water Formula equation 2 H 2 + O 2 2 H 2 O 4

Symbols used in chemical equations (s) solid (l) liquid (g) gas (aq) aqueous - the substance is dissolved in water (solution) a gas is given off ↓ a precipitate forms (a solid in a solution) 5

1. Synthesis Reactions • Synthesis reactions occur when two substances (generally elements) combine and form a compound. • Also called combination or addition reactions. • In general: A + B AB reactant + reactant 1 product • Example: C + O 2 CO 2

1. Synthesis Reactions Hydrogen and oxygen yields water 2 H 2 + O 2 2 H 2 O Magnesium plus nitrogen yields magnesium nitride 3 Mg + N 2 Mg 3 N 2 Iron and sulfur yields iron(II) sulfide Fe + S Fe. S 7

Practice Predict the products. Write and balance the following synthesis reaction equations. • Sodium metal reacts with chlorine gas Na(s) + Cl 2(g) • Solid Magnesium reacts with fluorine gas Mg(s) + F 2(g) • Aluminum metal reacts with fluorine gas Al(s) + F 2(g)

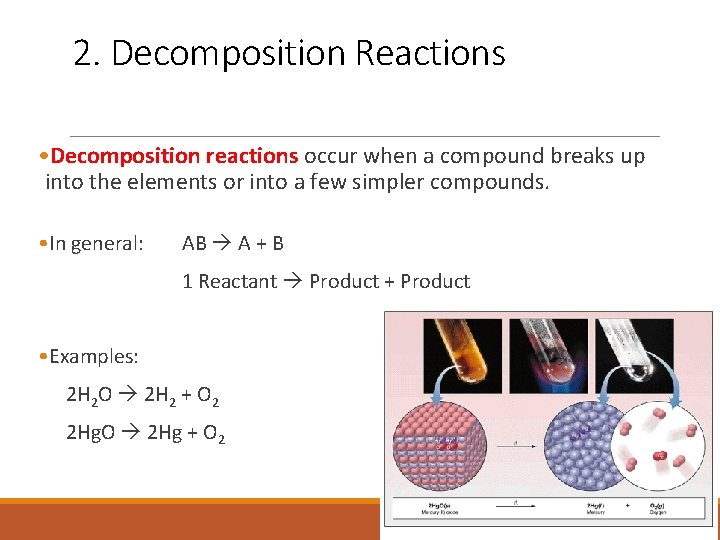
2. Decomposition Reactions • Decomposition reactions occur when a compound breaks up into the elements or into a few simpler compounds. • In general: AB A + B 1 Reactant Product + Product • Examples: 2 H 2 O 2 H 2 + O 2 2 Hg. O 2 Hg + O 2

Practice Predict the products. Then write and balance the following decomposition reaction equations: • Solid Lead (IV) oxide decomposes Pb. O 2(s) • Aluminum nitride decomposes Al. N(s)

Practice Identify the type of reaction for each of the following synthesis or decomposition reactions, and write the balanced equation: • N 2(g) + O 2(g) • Ba. CO 3(s) • Co(s)+ S(s) • NH 3(g) + H 2 CO 3(aq)