Catalyst Deactivation Hsin Chu Professor Dept of Environmental















- Slides: 21

Catalyst Deactivation 朱信 Hsin Chu Professor Dept. of Environmental Eng. National Cheng Kung University 1

1. Introduction Deactivation a. high temperature exposure: automobile catalytic converter, close to 1000℃ b. poisoning: exhaust or process contaminants adsorbing onto or blocking active sites c. attrition and erosion of the washcoat from the support l Model Reaction A convenient tool for studying deactivation and regeneration l 2

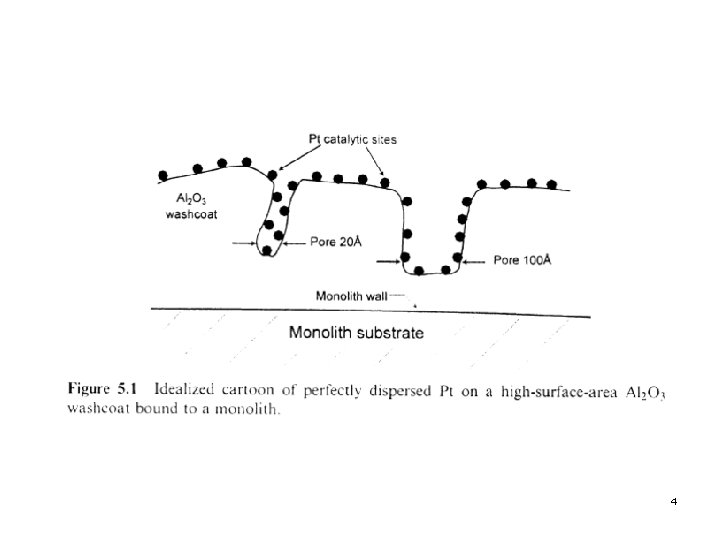
2. Thermally Induced Deactivation l. A perfectly dispersed (100% dispersion) catalyst is one in which every atom (or molecule) of active component is available to the reactants. This is shown is Fig. 5. 1 (next slide). 3

4

Some catalysts are made in this highly active state but are highly unstable, and thermal effects cause crystal growth, resulting in a loss of catalytic surface area. l Additionally, the carrier with a large internal surface network of pores tends to undergo sintering with a consequent loss in internal surface area. l Besides, reactions of the catalytically active species with the carrier, resulting in the formation of a less catalytically active species. l 5

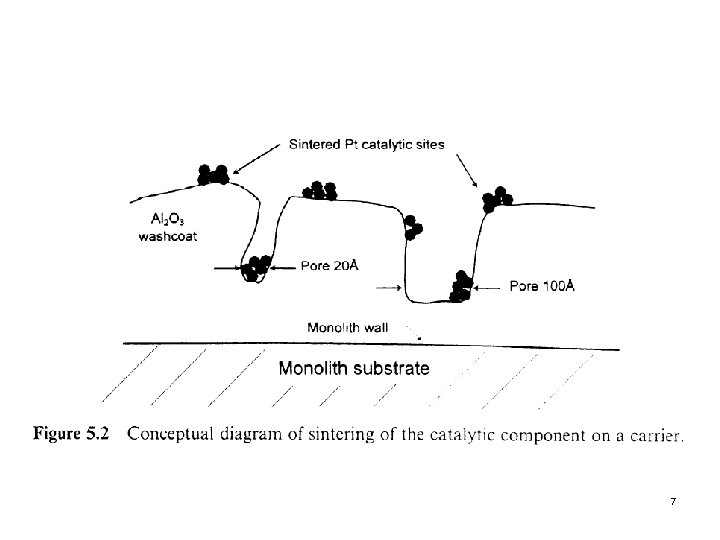
2. 1 Sintering of the Catalytic Component Next slide (Fig. 5. 2) Sintering by growth of catalyst crystals This condition can be measured by selective chemisorptions techniques in which a thermally aged catalyst adsorbs much less adsorbate than when it was fresh. l Stabilizer Certain rare-earth oxides such as Ce. O 2 and La 2 O 3 have been effective in reducing sintering rates of Pt in the automobile exhaust catalytic converter. It may fix the catalytic components to the surface minimizing mobility and crystal growth. l 6

7

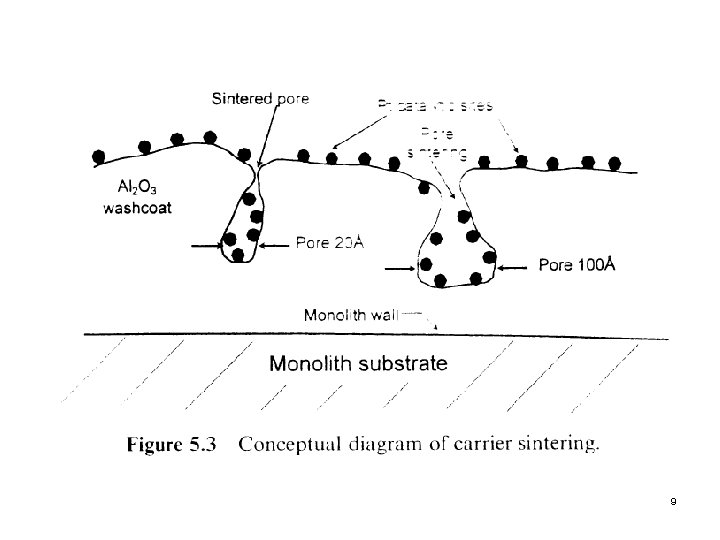
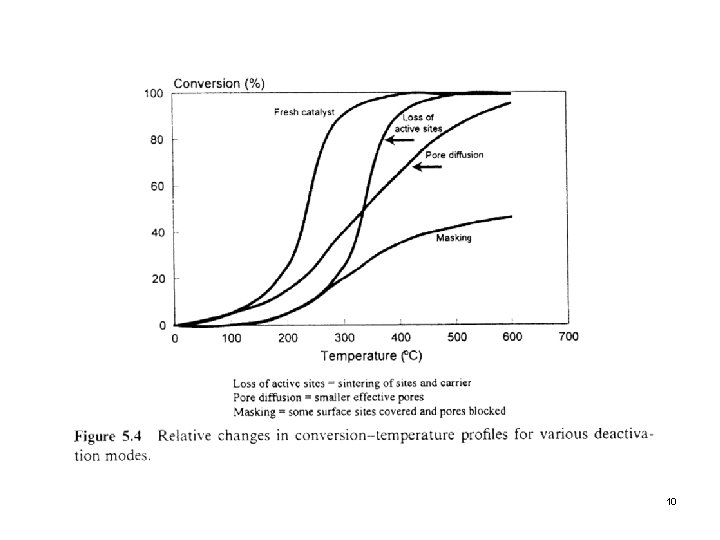
2. 2 Carrier Sintering Within a given crystal structure, such as γ-Al 2 O 3, the loss of surface area is associated with loss of H 2 O and a gradual loss of the internal pore structure network, as shown in the next slide (Fig. 5. 3) l The presence of these phenomena is determined by a progressive decrease in the activation energy of the reaction. l Second slide (Fig. 5. 4) Conversion profiles for various deactivation modes l 8

9

10

Second mechanism for carrier change in crystal structure γ-Al 2 O 3 → α-Al 2 O 3 150 m 2/g < 5 m 2/g Anatase Ti. O 2 Rutile Ti. O 2 60 m 2/g < 10 m 2/g l Stabilizer Ba. O, La 2 O 3, Si. O 2, or Zr. O 2 can retard the rate of sintering in certain carriers. They are believed to form solid solutions with the carrier surface, decreasing their surface reactivity, which leads to sintering. l 11

2. 3 Catalytic Species-Carrier Interactions l Rh 2 O 3 reacts with a high-surface-area γ-Al 2 O 3, forming an inactive compound during hightemperature lean conditions in the automobile exhaust. (for NOx removal) l Therefore, it is better to use carriers such as Si. O 2, Zr. O 2, Ti. O 2, and their combinations that are less reactive with Rh 2 O 3 than Al 2 O 3. However, these alternative carriers are not as stable against sintering. 12

3. Poisoning l Selective poisoning A chemical directly reacts with the active site or the carrier, rendering it less or completely inactive. l Nonselective poisoning Deposition of fouling agents onto or into the catalyst carrier, masking sites and pores, resulting in a loss in performance. 13

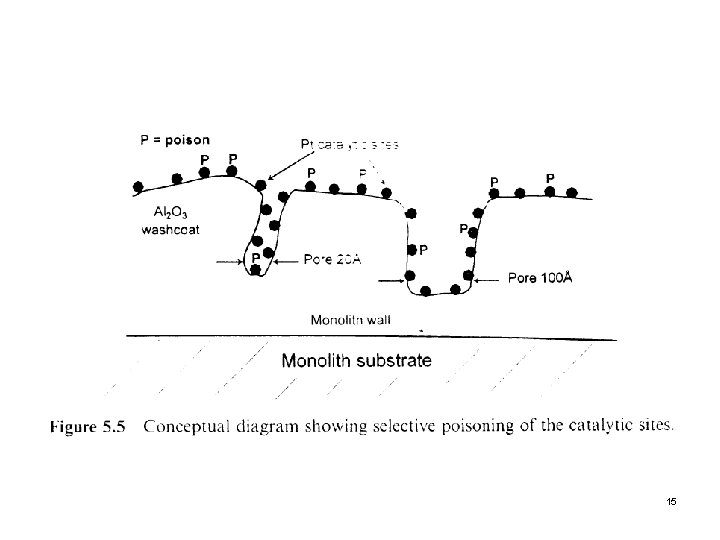
3. 1 Selective Poisoning Next slide (Fig. 5. 5) A poison directly reacts with an active site l Permanent deactivation Pb, Hg, and Cd react directly with Pt, forming a catalytically inactive alloy. l Reversible deactivation SO 2 merely adsorbs onto a metal site (i. e. , Pd). Heat treatment, washing, or simply removing the poison from the process stream, often desorbs the poison from the catalytic site and restoring its catalytic activity. l 14

15

When active sites are directly poisoned, there is a shift to high temperature but with no change in the slope of conversion profile since the remaining sites can function as before with no change in activation energy. l When the carrier reacts with a constituent in the gas stream to form a new compounds, as in the case of Al 2(SO 4)3, pores are generally partially blocked, resulting in increased diffusion resistance. This will cause a decrease in the activation energy. l 16

SO 2 chemisorbs onto Pd/γ-Al 2 O 3, causing deactivation for methane oxidation. Some of the adsorbed SO 2 is converted to SO 3, which spills over, forming Al 2(SO 4)3. Using nonsulfating carriers such as Zr. O 2 or Si. O 2 leads to a faster rate of deactivation since no reservoir is available for spillover. l With the Pt/γ-Al 2 O 3 catalyst the SO 2 is readily converted to SO 3, which rapidly desorbs and reacts with the Al 2 O 3, forming Al 2(SO 4)3, which slowly causes pore plugging. By using nonsulfating carriers, the Pt catalyst can be made resistant to deactivation. l 17

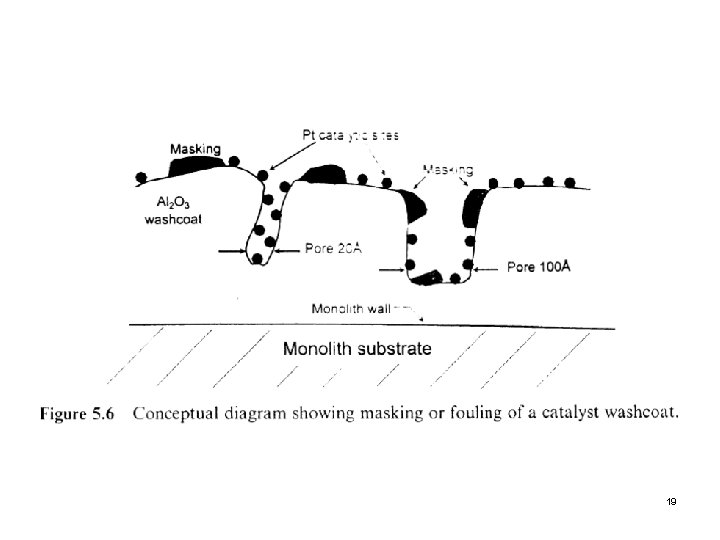
3. 2 Nonselective Poisoning Aerosol or high-molecular-weight material from upstream equipment physically deposit onto the surface of the washcoat to cause deactivation is referred to as “fouling” or “masking”. l Reactor-scale metals (Fe, Ni, Cr, etc. ) resulting from corrosion, silica/alumina-containing dusts, phosphorous from lubricating oils, and similar compounds are good examples. l Next slide (Fig. 5. 6) Masking or fouling of a catalyst washcoat l Second slide (Fig. 5. 7) SEM of fresh and aged Pt/Al 2 O 3 surfaces l 18

19

20

4. Washcoat Loss l Attrition or Erosion Irreversible deactivation a. high linear velocities of gas flow b. thermal expansion differences between the washcoat and the monolith, especially metal substrates 21