Thermodynamics Chemical reactions proceed according to the rules









![Biological reactions DG = DGo + RTln ([products]/[reactants]) Where R = gas constant, T Biological reactions DG = DGo + RTln ([products]/[reactants]) Where R = gas constant, T](https://slidetodoc.com/presentation_image_h2/598720bc4fe747413c19079f48185105/image-12.jpg)

![Transport across membranes DG = RTln ([inside]/[outside]) + z. FEm where z = valence Transport across membranes DG = RTln ([inside]/[outside]) + z. FEm where z = valence](https://slidetodoc.com/presentation_image_h2/598720bc4fe747413c19079f48185105/image-14.jpg)
- Slides: 14

Thermodynamics Chemical reactions proceed according to the rules of thermodynamics • The law of conservation of energy – energy can be converted from one form to another but the total amount of energy is constant • Entropy – the universe is becoming more chaotic ACK!

Thermodynamics Some constants Gas constant: R = 8. 315 Joules/K* mol or 1. 9872 cal/K. mol Faradays constant: F = 96485 Joules/Volt. mol or 23062 cal/Volt* mol

Energy: definitions Energy – ability to do work Energetics – energy transfer Types of energy • Potential – trapped energy • Kinetic – energy of movement

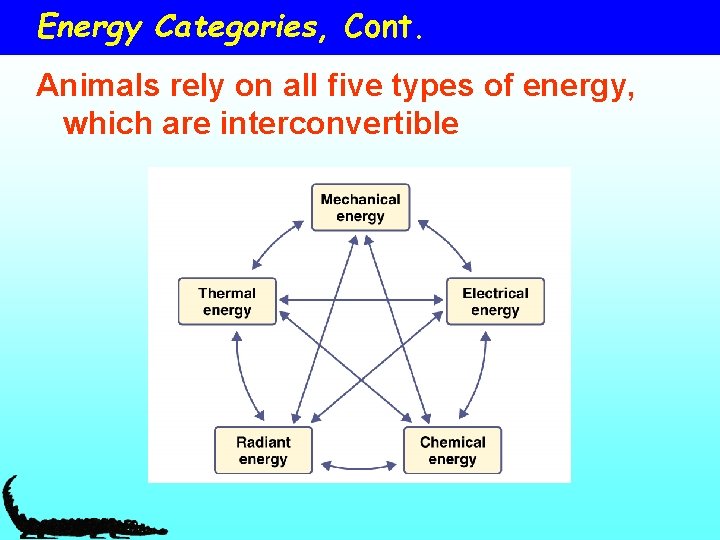
Energy Categories: more definitions • Radiant energy – energy released from one object to another • Mechanical energy – energy to move objects from place to place • Electrical energy – energy that results from the movement of charged particles down a charge gradient • Thermal energy – reflected in the movement of particles and serves to increase temperature • Chemical energy – energy that is held within chemical bonds

Energy Categories, Cont. Animals rely on all five types of energy, which are interconvertible

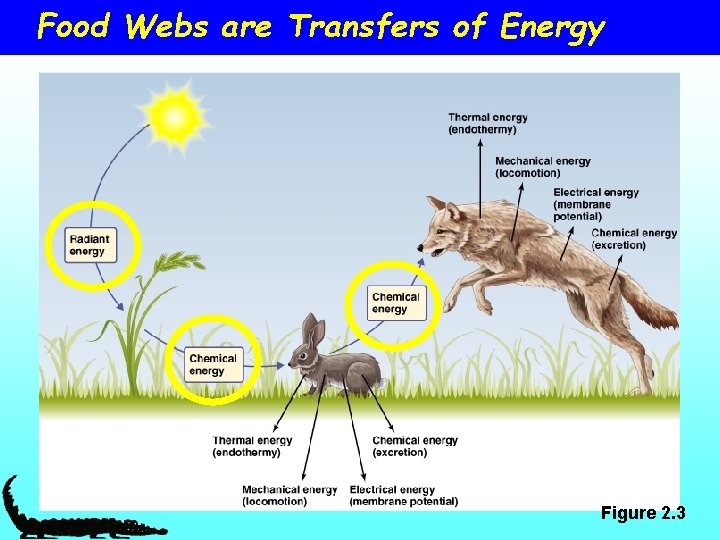
Food Webs are Transfers of Energy Figure 2. 3

Thermodynamics in a biological setting Free Energy (G) 1. Change in free Energy (ΔG) ΔG = Products – Reactants ΔG negative – reaction will proceed forward → ΔG positive – reaction will proceed backward ← ΔG zero – reaction at equilibrium ↔ 2. Standard free Energy – ΔGo: 298 K (25 o. C), 1 atm pressure, p. H 7. 0 and 1 M [initial] for all reactants and products

Thermal Energy Thermal energy movement of molecules Most chemical reactions involve changes in thermal energy • Exothermic reactions – release heat • Endothermic reactions – absorb heat

Chemical Reactions and Thermal Energy Enthalpy – average thermal energy of a collection of molecules i. e. bond energy Change in enthalpy (DH) = Hproducts – Hsubstrates • Exothermic: DH is negative i. e. C 6 H 12 O 6 + 6 O 2 → 6 CO 2 + 6 H 2 O + energy • Endothermic: DH is positive i. e. ADP + Pi → ATP

Chemical Reactions and Thermal Energy Enthalpy and Entropy together Entropy (S) – measure of randomness or disorder Exothermic: DH is negative, increase in DS → reaction will occur spontaneously – negative DG Endothermic: DH is positive, DS is positive → reaction will occur spontaneously. It has to overcome the positive DH

Free Energy: calculations Free energy changes of reactions are additive (coupled reactions): Consider the phosphorylation of glucose to glucose 6 -phosphate: DGo: glucose + Pi ↔ glucose-6 -phosphate + H 2 O = 3. 3 kcal/mol DGo: ATP + H 2 O ↔ ADP + Pi = -7. 3 kcal/mol Summing these reactions together: ATP + glucose ↔ ADP + glucose 6 -phosphate DG° = +3. 3 + (-7. 3) = - 4 kcal/mol (favourable)
![Biological reactions DG DGo RTln productsreactants Where R gas constant T Biological reactions DG = DGo + RTln ([products]/[reactants]) Where R = gas constant, T](https://slidetodoc.com/presentation_image_h2/598720bc4fe747413c19079f48185105/image-12.jpg)
Biological reactions DG = DGo + RTln ([products]/[reactants]) Where R = gas constant, T = temperature in Kelvin Example: glucose + ATP ↔ glucose-6 -phosphte + ADP DGo: glucose + Pi ↔ glucose-6 -phosphate + H 2 O = 3. 3 kcal/mol DGo: ATP + H 2 O ↔ ADP + Pi = -7. 3 kcal/mol Glucose: [5 m. M]; ATP: [2 m. M]; ADP: [0. 15 m. M]; glucose-6 phosphate: [0. 05 m. M] So, DG = - 4. 0 kcal/mol + 1. 9872 cal/K mol)(298 K)ln((0. 05*0. 15)/(5*2)) = -8. 26 kcal/mol

ΔG for reactions that don’t make or break bonds DGo is zero - Examples: glucose transport, ion transport across membranes DG = RTln ([inside]/[outside]) Or for charged ions: DG = RTln ([inside]/[outside]) + z. FEm where z = valence of the ion; F = Faraday constant and Em = membrane potential
![Transport across membranes DG RTln insideoutside z FEm where z valence Transport across membranes DG = RTln ([inside]/[outside]) + z. FEm where z = valence](https://slidetodoc.com/presentation_image_h2/598720bc4fe747413c19079f48185105/image-14.jpg)
Transport across membranes DG = RTln ([inside]/[outside]) + z. FEm where z = valence of the ion; F = Faraday constant and Em = membrane potential Example: Diffusion of Cl- from out to in Cl- outside cell: 120 m. M; Cl- inside cell: 10 m. M; Em = -80 m. V DG = (1. 987 cal/K mol)(298 K)(ln(10/120) + ( -1)(23062 cal/V mol)(-0. 08 V) = 376 cal/mol