Proteins Proteins are polymers made up of amino






- Slides: 12


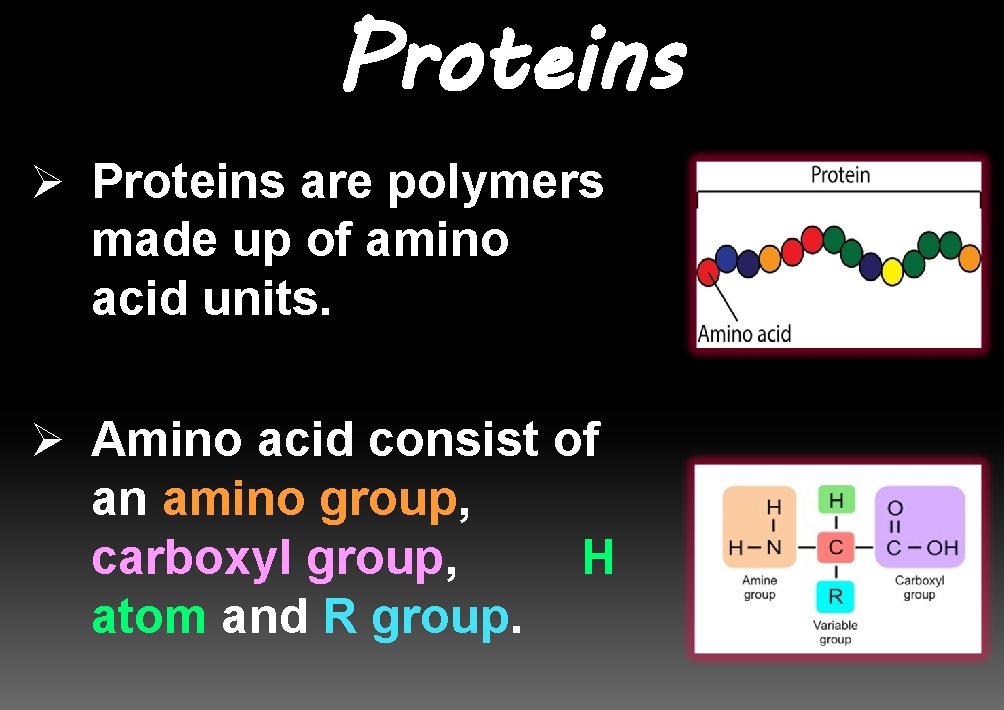
Proteins Ø Proteins are polymers made up of amino acid units. Ø Amino acid consist of an amino group, carboxyl group, atom and R group. H

Function of proteins q Act as structural such as : Skin , hair , nails , muscle and connective tissue q Act as catalysts such as enzyme q Act as hormones such as insulin q Act as transport such as hemoglobin

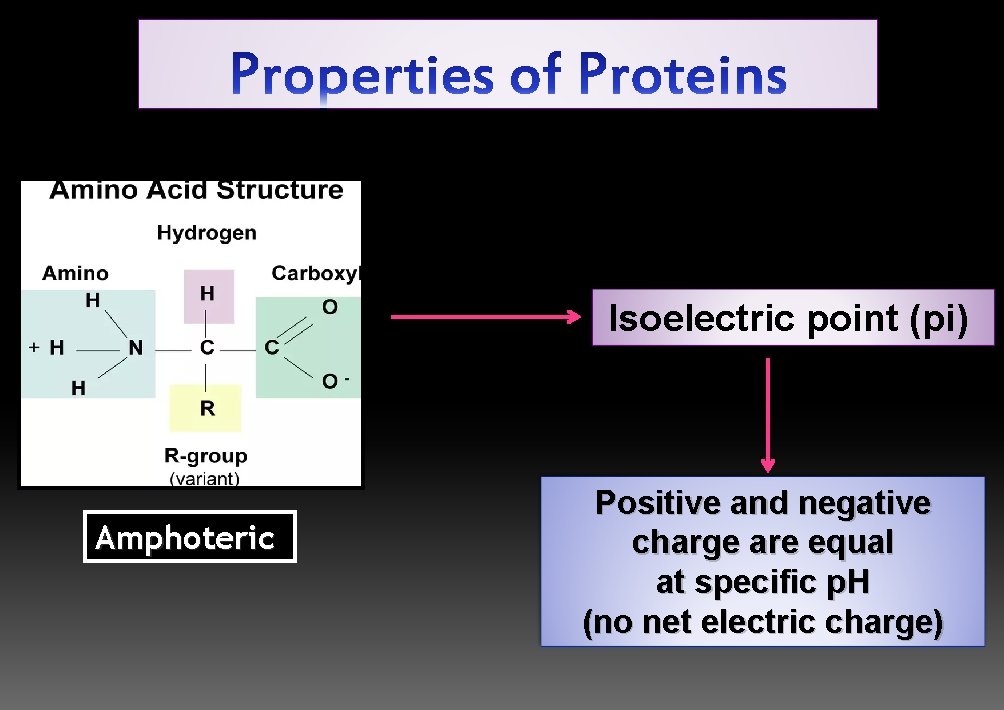
Isoelectric point (pi) Amphoteric Positive and negative charge are equal at specific p. H (no net electric charge)

Precipitation of proteins v Effect of strong acid and strong base v Effect of neutral salts v Effect of alkaloid reagents v Effect of polar solvent v Effect of heat

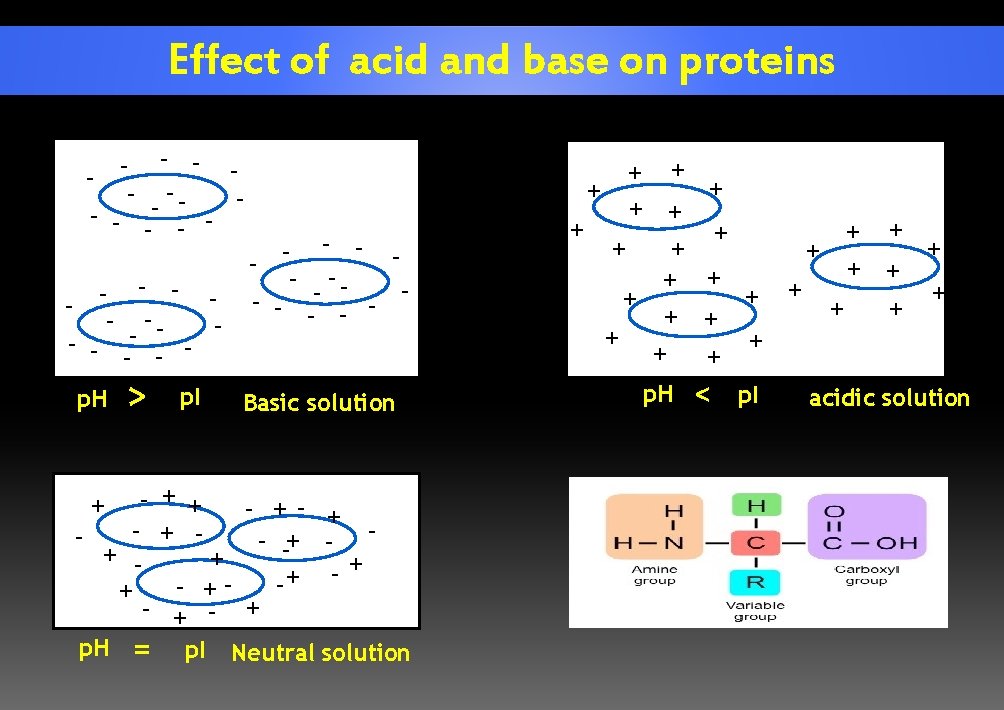
Effect of acid and base on proteins - - - -- - - p. H > p. I + + - + + + - = p. I + p. H - -- - - Basic solution + - ++ - - +- + - -+ - + + + Neutral solution + + + + p. H + + + < + + p. I + + + + + acidic solution

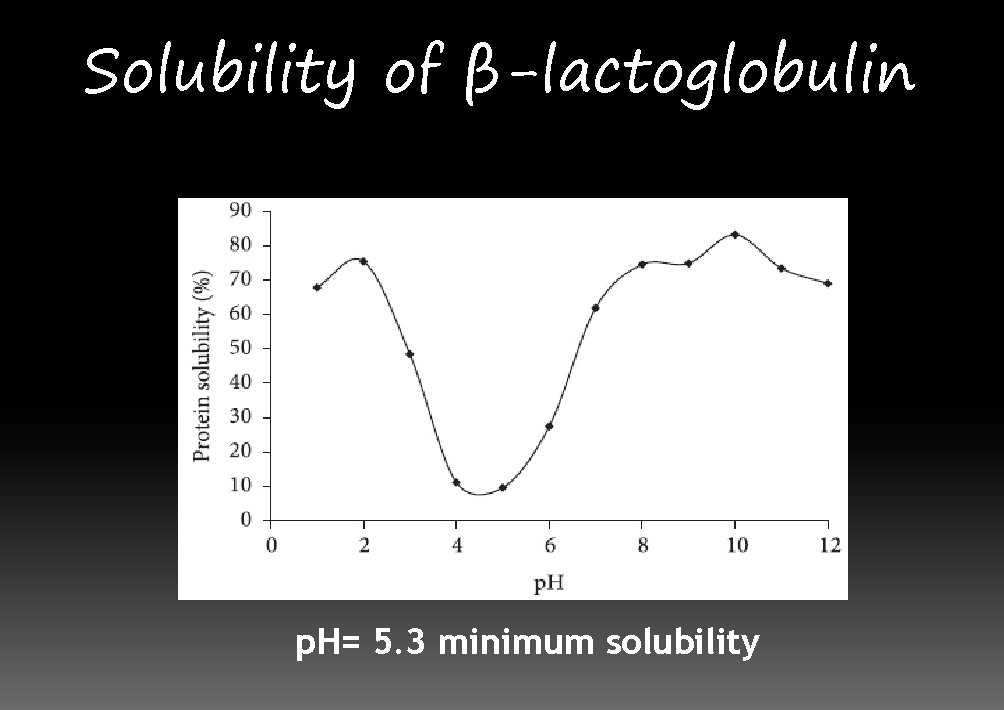
Solubility of β-lactoglobulin p. H= 5. 3 minimum solubility

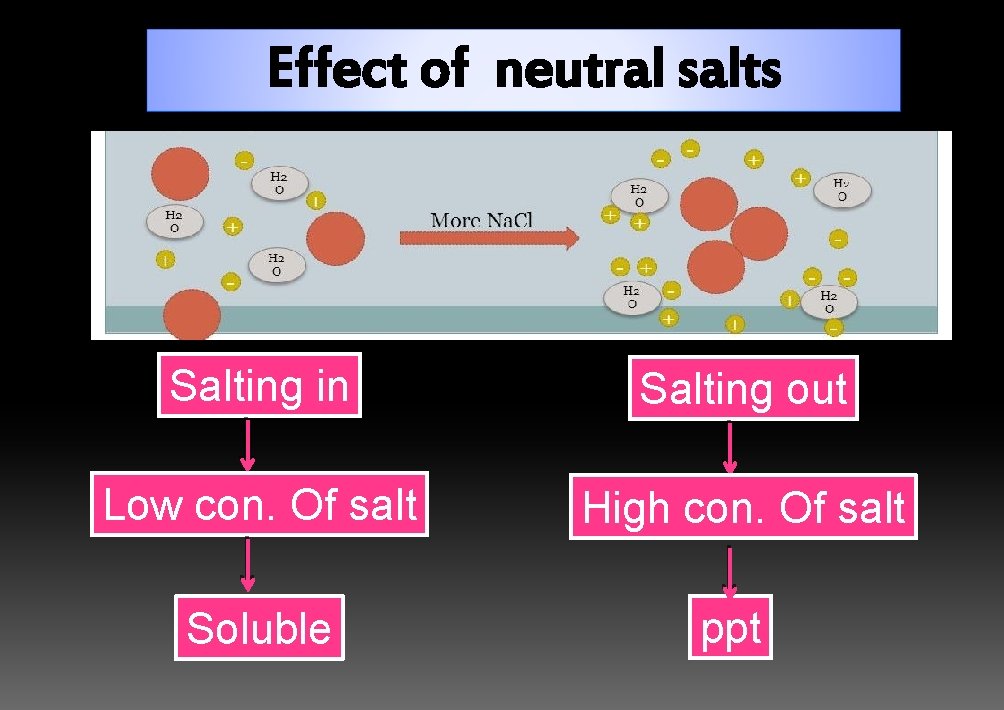
Effect of neutral salts Salting in Salting out Low con. Of salt High con. Of salt Soluble ppt

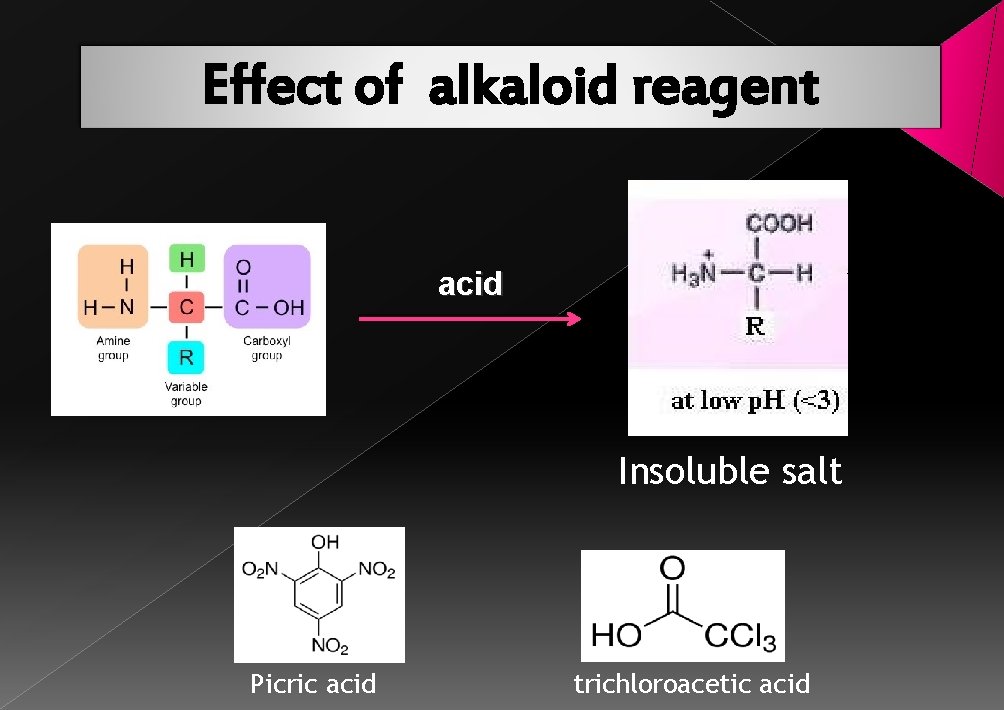
Effect of alkaloid reagent acid Insoluble salt Picric acid trichloroacetic acid

Effect of polar solvent o Polar solvent such as ethanol, methanol and acetone o Organic solvent with water made miscible solution. o Miscible solution decrease the solubility of protein.

Effect of heat Protein types are : - Fibrous protein (insoluble in water) - Globular protein (soluble in water) Protein coagulation results in the disorganization of a protein’s structures without the hydrolysis of peptide bonds lead to precipitation of protein.

Precipitation effects 1 - acid and base effect : 2 ml H 2 SO 4 (con. ) 1 ml albumen 2 ml Na. OH (saturated) 1 ml albumen 3 - alkaloid reagent effect : 2 - neutral salt effect : 10 drop cupric sulphate 3 ml albumen 3 drop lead actate 3 ml albumen 4 - polar solvent effect: 5 ml water 10 drop tannic acid 1 ml albumen 2 ml ethanol 1 ml albumen