METHODS OF POLYMERIZATION CHARACTERIZATION BY TGA DTA MOLECULAR

METHODS OF POLYMERIZATION, CHARACTERIZATION BY TGA, DTA, MOLECULAR WEIGHT AND ITS DETERMINATION, AMORPHOUS AND CRYSTALLINE POLYMERS, BIOPOLYMERS, STRUCTURE -PROPERTY RELATION IN POLYMERS.

INTRODUCTION Many + Parts The word, polymer, implies that polymers are constructed from pieces (monomers) that can be easily connected into long chains (polymer). When you look at the above shapes, your mind should see that they could easily fit together.

A WORD POLYMER IS A COMBINATION OF TWO GREEK WORDS, “POLY” MEANS “MANY” AND “MEROS” MEANING “PARTS OR UNITS”. A POLYMER IS A LARGE MOLECULE OF WHICH IS FORMED BY REPEATED LINKING OF THE SMALL MOLECULES CALLED “MONOMERS”. MORE MONOMER MOLECULES JOINED IN UNITS OF LONG POLYMER. MACROMOLECULES HAVING HIGH MOLECULAR MASS (103 -107 U) POLYMERIZATION: THE PROCESS OF FORMATION OF POLYMER FROM ITS MONOMER DEGREE OF POLYMERIZATION (DP): NUMBER OF REPEATING UNITS OF MONOMER (C 2 H 4)n, where n stands for DP –High DP: hard and heat resistant –Low DP: soft, gummy

A polymer made form just one monomer is polyethylene. It is the most common plastic you see. It is used for bottles, buckets, jugs, containers, toys, even synthetic lumber, and many other things.

EXAMPLE OF SOME POLYMERS

CLASSIFICATION OF POLYMER § CLASSIFICATION BASED ON TYPE OF MONOMER § CLASSIFICATION BASED ON SOURCE § CLASSIFICATION BASED ON STRUCTURE § CLASSIFICATION BASED ON POLYMERISATION § CLASSIFICATION BASED ON MOLECULER FORCE

CLASSIFICATION BASED ON TYPE OF MONOMER (i) HOMOPOLYMERS-Polymers consist of identical monomers, exa. PVC (II) COPOLYMERS: POLYMERS CONSIST OF MONOMERS OF DIFFERENT CHEMICAL STRUCTURES, EXA. BAKELITE, NYLOM-6, 6; POLYESTER Polyester is made from the two monomers, terephthalic acid (note: “ph” is silent) and ethylene glycol (car antifreeze). This makes a popular plastic called PETE, which is short for Polyethylene Terephthalate. The synthesis is also a dehydration reaction because water is given off. PETE
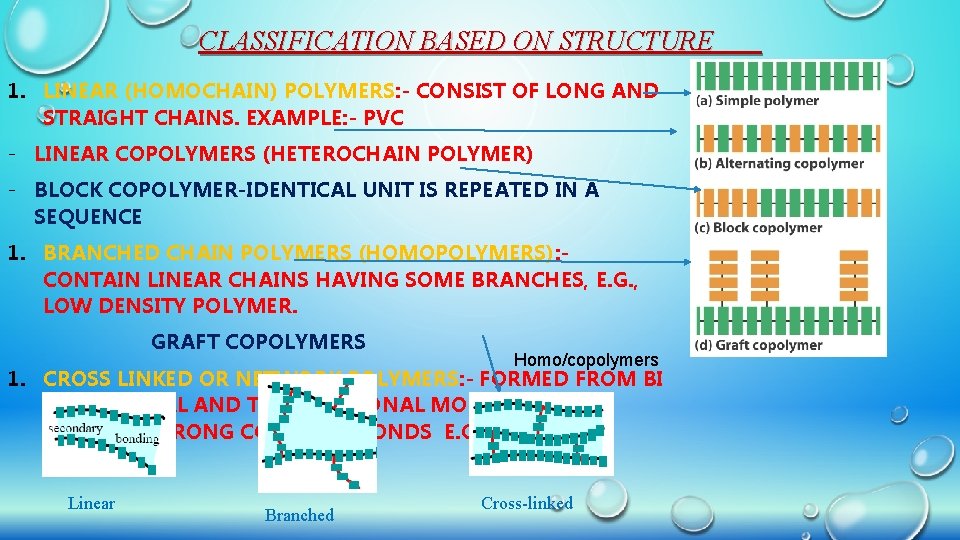
CLASSIFICATION BASED ON STRUCTURE 1. LINEAR (HOMOCHAIN) POLYMERS: - CONSIST OF LONG AND STRAIGHT CHAINS. EXAMPLE: - PVC - LINEAR COPOLYMERS (HETEROCHAIN POLYMER) - BLOCK COPOLYMER-IDENTICAL UNIT IS REPEATED IN A SEQUENCE 1. BRANCHED CHAIN POLYMERS (HOMOPOLYMERS): CONTAIN LINEAR CHAINS HAVING SOME BRANCHES, E. G. , LOW DENSITY POLYMER. GRAFT COPOLYMERS Homo/copolymers 1. CROSS LINKED OR NETWORK POLYMERS: - FORMED FROM BI -FUNCTIONAL AND TRI-FUNCTIONAL MONOMERS AND CONTAIN STRONG COVALENT BONDS E. G. BAKELITE, MELAMINE. Linear Branched Cross-linked
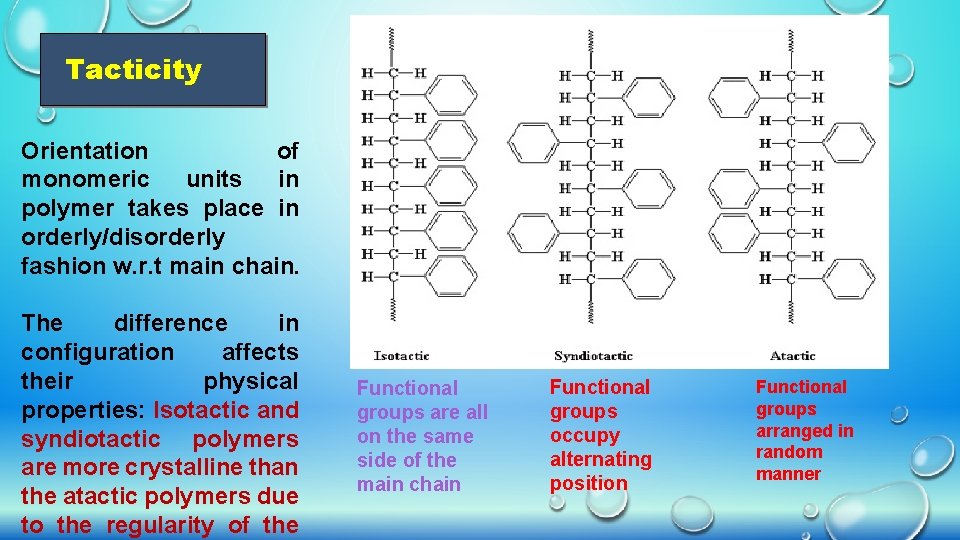
Tacticity Orientation of monomeric units in polymer takes place in orderly/disorderly fashion w. r. t main chain. The difference in configuration affects their physical properties: Isotactic and syndiotactic polymers are more crystalline than the atactic polymers due to the regularity of the Functional groups are all on the same side of the main chain Functional groups occupy alternating position Functional groups arranged in random manner

CLASSIFICATION BASED ON SOURCE 1. NATURAL POLYMERS: - NATURAL POLYMER IS A POLYMER THAT RESULTS FROM ONLY RAW MATERIALS THAT ARE FOUND IN NATURE. EXAMPLE: - PROTEINS, CELLULOSE, STARCH, RUBBER. 2. SEMI-SYNTHESIS POLYMERS: CELLULOSE DERIVATIVES - CELLULOSE ACETATE (RAYON). 3. SYNTHESIS POLYMERS: - SYNTHETIC RUBBERS (BUNA-S, BUNA-R), SYNTHETIC FIBRES (NYLON), PLASTIC (POLYTHENE), POLYESTER. 3 D representation of the structure of a protein

CLASSIFICATION BASED ON POLYMERIZATION 1. ADDITION POLYMERS FORMED BY THE REPEATED ADDITION OF MONOMER MOLECULES POSSESSING DOUBLE OR TRIPLE BONDS 2. CONDENSATION POLYMERS FORMED BY REPEATED CONDENSATION REACTION BETWEEN TWO DIFFERENT BIFUNCTIONAL OR TRI-FUNCTIONAL MONOMERIC UNITS. EG. TERYLENE (DACRON), NYLON 6, 6, NYLON 6.

CLASSIFICATION BASED ON MOLECULAR FORCE 1. Elastomers 2. Fibres These are rubber – like solids with elastic properties. In these elastomeric polymers, the polymer chains are held together by the weakest intermolecular forces. These weak binding forces permit the polymer to be stretched. A few ‘crosslinks’ are introduced in between the chains, which help the polymer to retract to its original position after the force is released as in vulcanised rubber. The examples are buna-S, buna-N, neoprene, etc. Fibres are thread forming solids which possess high tensile strength and high modulus. These characteristics can be attributed to the strong intermolecular forces like hydrogen bonding. These strong forces also lead to close packing of chains and thus impart crystalline nature. The examples are polyamides (nylon 6, 6), polyesters (terylene), etc.

3. Thermoplastic polymers These are the linear or slightly branched long chain molecules capable of repeatedly softening on heating and hardening on cooling. These polymers possess intermolecular forces of attraction intermediate between elastomers and fibres. Some common thermoplastics are polythene, polystyrene, polyvinyls, etc. 4 Thermosetting polymers These polymers are cross linked or heavily branched molecules, which on heating undergo extensive cross linking in moulds and again become infusible. These cannot be reused. Some common examples are bakelite, urea-formaldelyde resins, etc.

TYPES OF POLYMERIZATION 1. ADDITION POLYMERIZATION (CHAIN-GROWTH) 2. CONDENSATION POLYMERIZATION (STEP-GROWTH) 3. COPOLYMERIZATION ADDITION POLYMERIZATION q THE POLYMER IS FORMED FROM THE MONOMER, WITHOUT THE LOSS OF ANY MATERIAL, AND THE PRODUCT IS THE EXACT MULTIPLE OF THE ORIGINAL MONOMERIC MOLECULE. q ADDITION POLYMERIZATION PROCEEDS BY THE INITIAL FORMATION OF SOME REACTIVE SPECIES SUCH AS FREE RADICALS OR IONS (CARBANION OR CARBACATION) AND BY THE ADDITION OF THE REACTIVE SPECIES TO THE OTHER MOLECULE, WITH THE REGENERATION OF THE REACTIVE FEATURE.

• Polymerization occurs in stages that leads to an increase in chain length. • During each stage a reactive intermediate is formed which is subsequently used up in the next • It is four types: free redical, cationic, anionic and coordination polymerization 1. Free radical mechanism: - Alkenes or dienes and their derivatives are polymerized in the presence of a free radical generating initiator (catalyst) like benzoyl peroxide, acetyl peroxide, t-bu peroxide, etc. This process involves three steps: a) Chain initiation step - addition of phenyl free radical formed by the peroxide to the ethene double bond , thereby forming a larger radical. b) Chain propagation step - repetition of this sequence with new and bigger radicals. c) Chain terminating step - the product radical thus formed reacts with another radical to form the polymerized product.
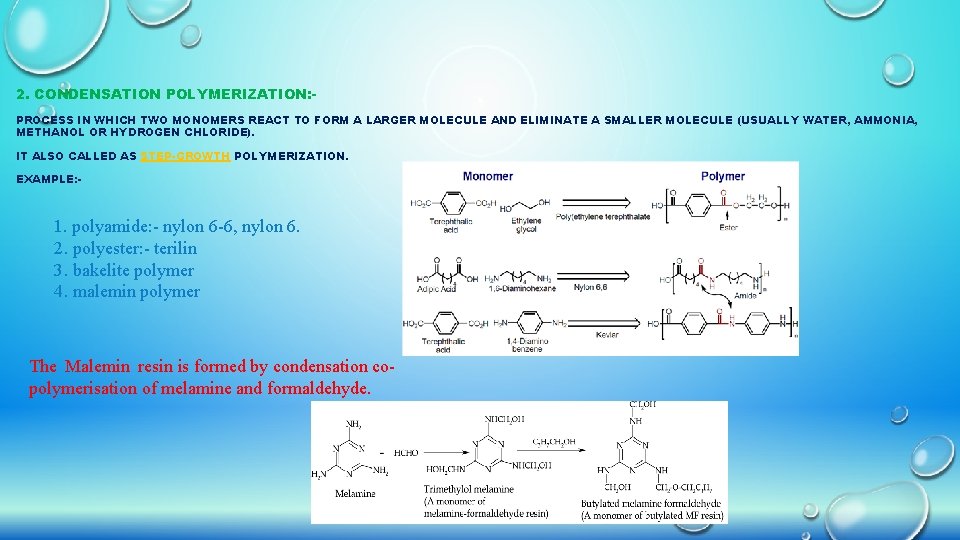
2. CONDENSATION POLYMERIZATION: PROCESS IN WHICH TWO MONOMERS REACT TO FORM A LARGER MOLECULE AND ELIMINATE A SMALLER MOLECULE (USUALLY WATER, AMMONIA, METHANOL OR HYDROGEN CHLORIDE). IT ALSO CALLED AS STEP-GROWTH POLYMERIZATION. EXAMPLE: - 1. polyamide: - nylon 6 -6, nylon 6. 2. polyester: - terilin 3. bakelite polymer 4. malemin polymer The Malemin resin is formed by condensation copolymerisation of melamine and formaldehyde.

COPOLYMERIZATION Copolymerisation is a polymerisation reaction in which a mixture of more than one monomeric species is allowed to polymerise and form a copolymer. The copolymer can be made not only by chain growth polymerisation but by step growth polymerisation also. It contains multiple units of each monomer used in the same polymeric chain. No loss of small molecules. For example, a mixture of 1, 3 – butadiene and styrene can form a copolymer.

CHARACTERISTICS OF POLYMER Ø Ø Ø LOW DENSITY. LOW COEFFICIENT OF FRICTION. GOOD CORROSION RESISTANCE. GOOD MOULD ABILITY. EXCELLENT SURFACE FINISH CAN BE OBTAINED. CAN BE PRODUCED WITH CLOSE DIMENSIONAL TOLERANCES. ECONOMICAL. POOR TENSILE STRENGTH. LOW MECHANICAL PROPERTIES. POOR TEMPERATURE RESISTANCE. CAN BE PRODUCED TRANSPARENT OR IN DIFFERENT COLOURS

THERMOGRAVIMETRIC ANALYSIS (TGA) • PRINCIPLE: IN THERMOGRAVIMETRIC ANALYSIS, THE SAMPLE IS HEATED IN A GIVEN ENVIRONMENT (AIR, N 2, CO 2, HE, AR, ETC. ) AT CONTROLLED RATE. THE CHANGE IN THE WEIGHT OF THE SUBSTANCE IS RECORDED AS A FUNCTION OF TEMPERATURE OR TIME. THE TEMPERATURE IS INCREASED AT A CONSTANT RATE FOR A KNOWN INITIAL WEIGHT OF THE SUBSTANCE AND THE CHANGES IN WEIGHTS ARE RECORDED AS A FUNCTION OF TEMPERATURE AT DIFFERENT TIME INTERVAL. THIS PLOT OF WEIGHT CHANGE AGAINST TEMPERATURE IS CALLED THERMOGRAVIMETRIC CURVE OR THERMOGRAM, THIS IS THE BASIC PRINCIPLE OF TGA

EXAMPLE: TGA CURVE FOR AGNO 3 • THE HORIZONTAL PORTION OF THE CURVE INDICATES THAT, THERE IS NO CHANGE IN WEIGHT (AB & CD) AND THE PORTION BC INDICATES THAT THERE IS WEIGHT CHANGE. • THE WEIGHT OF THE SUBSTANCE (AGNO 3) REMAINS CONSTANT UPTO A TEMPERATURE OF 473°C INDICATING THAT AGNO 3 IS THERMALLY STABLE UPTO A TEMPERATURE OF 473°C. • AT THIS TEMPERATURE IT STARTS LOSING ITS WEIGHT AND THIS INDICATES THAT THE DECOMPOSITION STARTS AT THIS TEMPERATURE. IT DECOMPOSES TO NO 2, O 2 AND AG. • THE LOSS IN WEIGHT CONTINUES UP TO 608°C AND BEYOND THIS TEMPERATURE THE WEIGHT OF THE SAMPLE REMAINS CONSTANT, THIS IS SHOWN BY THE PORTION OF THE CURVE CD. • THE PORTION BETWEEN BC, REPRESENTS THE DECOMPOSITION OF SILVER NITRATE, THE DECOMPOSITION IS COMPLETE AT 608°C LEAVING METALLIC SILVER AS THE STABLE RESIDUE The diagram indicates the TGA curve for Ag. NO 3 → Ag + NO 2 + O 2

INSTRUMENTATION / BLOCK DIAGRAM OF TGA THE APPARATUS REQUIRED FOR TGA ANALYSIS ARE (A) A FURNACE WHICH CAN BE HEATED SO THAT THE TEMPERATURE GIVES LINEARITY WITH TIME. (B) A FURNACE CONTROLLED THERMOBALANCE • A KNOWN WEIGHT OF THE SAMPLE IS TAKEN IN A CRUCIBLE(C), WHICH IS ENCLOSED BY A FURNACE(F). THE FURNACE(F) TEMPERATURE IS RAISED SLOWLY, THE TEMPERATURE OF THE SAMPLE AND THE CORRESPONDING WEIGHT ARE TAKEN. • A PLATINUM/PLATINUM RHODIUM THERMOCOUPLE IS USED TO MEASURE THE SAMPLE TEMPERATURE, AND THE CHANGE IN WEIGHTS ARE FOUND OUT BY FINDING THE BEAM DEFLECTION ON ADDING A KNOWN WEIGHT TO THE PAN. (I. E) THE CHANGE IN THE WEIGHT ARE RECORDED FROM THE BEAM DEFLECTION. (C) RECORDER: A RECORDER RECORDS THE CHANGE IN WEIGHT IN Y AXIS AND W. R. TO TEMPERATURE ON THE X-AXIS. WE GET A THERMOGRAM.

TGA of calcium oxalate monohydrate (cac 2 o 4. h 2 o) • The successive plateau corresponds to the formation of anhydrous salt, calcium carbonate and calcium oxide. (A) Ca. C 2 O 4. H 2 O → Ca. C 2 O 4 + H 2 O (B) Ca. C 2 O 4 (C) Ca. CO 3 → Ca. CO 3 + CO → Ca. O + CO 2 • thermogram indicates that the loss of water begins at 100°c and loss of CO at 400°C and CO 2 at 680°C
Common applications of TGA are: (1) materials characterization through analysis of characteristic decomposition patterns, (2) studies of degradation mechanisms and reaction kinetics, (3) determination of organic content in a sample, (4) determination of inorganic (e. g. ash) content in a sample, which may be useful for corroborating predicted material structures or simply used as a chemical analysis. It is an especially useful technique for the study of polymeric materials, including thermoplastics, thermosets, elastomers, composites,

DIFFERENTIAL THERMAL ANALYSIS (DTA) Differential thermal analysis (or DTA) is a thermoanalytic technique. In DTA, the material under study and an inert reference (alumina) are made to undergo identical thermal cycles, while recording any temperature difference (∆T) between sample and reference. This differential temperature is then plotted against time, or against temperature (DTA curve, or thermogram)…This is a comparison method Changes in the sample, either exothermic or endothermic, can be detected relative to the inert reference. Thus, a DTA curve provides data on the transformations that have occurred, such as glass transitions, crystallization, melting and sublimation.

• IN DTA BOTH TEST SAMPLE & AN INERT REFERENCE MATERIAL (ALUMINA) – CONTROLLED HEATING OR COOLING PROGRAMMING • IF ZERO TEMPERATURE DIFFERENCE B/W SAMPLE & REFERENCE MATERIAL – SAMPLE DOES NOT UNDERGO ANY CHEMICAL OR PHYSICAL CHANGE. • IF ANY REACTION TAKES PLACE TEMPERATURE DIFFERENCE (∆T) WILL OCCUR B/W SAMPLE & REFERENCE MATERIAL. • A DTA CURVE CAN BE USED AS A FINGER PRINT FOR IDENTIFICATION PURPOSES
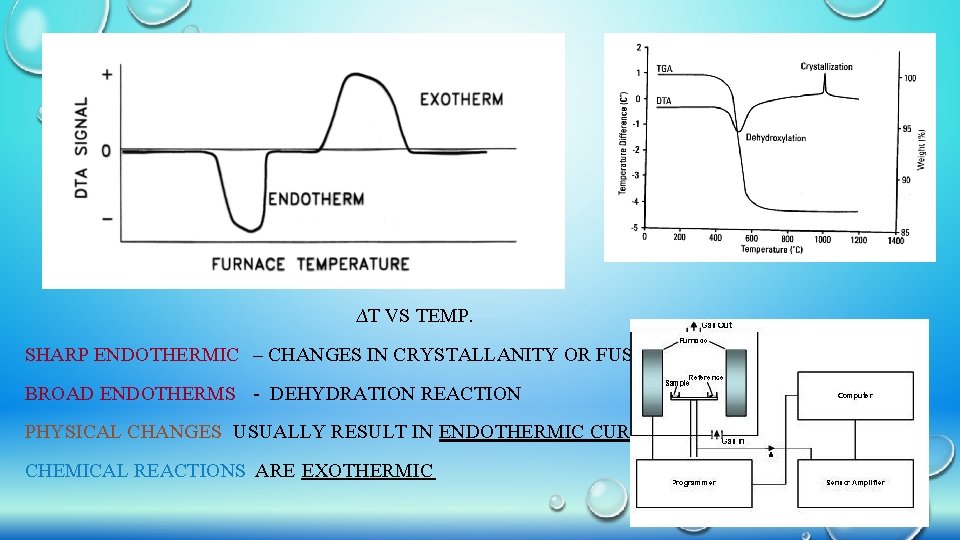
∆T VS TEMP. SHARP ENDOTHERMIC – CHANGES IN CRYSTALLANITY OR FUSION BROAD ENDOTHERMS - DEHYDRATION REACTION PHYSICAL CHANGES USUALLY RESULT IN ENDOTHERMIC CURVES CHEMICAL REACTIONS ARE EXOTHERMIC

APPLICATIONS OF DIFFERENTIAL THERMAL ANALYSIS ØQUALITATIVE AND QUANTITATIVE IDENTIFICATION OF MINERALS: DETECTION OF ANY MINERALS IN A SAMPLE ØPOLYMERIC MATERIALS: DTA USEFUL FOR THE CHARACTERIZATION OF POLYMERIC MATERIALS IN THE LIGHT OF IDENTIFICATION OF THERMO PHYSICAL , THERMO CHEMICAL, THERMO MECHANICAL AND THERMO ELASTIC CHANGES OR TRANSITIONS. ØMEASUREMENT OF CRYSTALLINE: MEASUREMENT OF THE MASS FRACTION OF CRYSTALLINE MATERIAL IN SEMI CRYSTALLINE POLYMERS. ØANALYSIS OF BIOLOGICAL MATERIALS: DTA CURVES ARE USED TO DATE BONE REMAINS OR TO STUDY ARCHAEOLOGICAL MATERIALS.





APPLICATION OF POLYMERS 1. MEDICINE: - MANY BIOMATERIALS, ESPECIALLY HEART VALVE REPLACEMENTS AND BLOOD VESSELS, ARE MADE OF POLYMERS LIKE DACRON, TEFLON AND POLYURETHANE. 2. CONSUMER SCIENCE : - PLASTIC CONTAINERS OF ALL SHAPES AND SIZES ARE LIGHT WEIGHT AND ECONOMICALLY LESS EXPENSIVE THAN THE MORE TRADITIONAL CONTAINERS. CLOTHING, FLOOR COVERINGS, GARBAGE DISPOSAL BAGS, AND PACKAGING ARE OTHER POLYMER APPLICATIONS. 3. INDUSTRY: - AUTOMOBILE PARTS, WINDSHIELDS FOR FIGHTER PLANES, PIPES, TANKS, PACKING MATERIALS, INSULATION, WOOD SUBSTITUTES, ADHESIVES, MATRIX FOR COMPOSITES, AND ELASTOMERS ARE ALL POLYMER APPLICATIONS USED IN THE INDUSTRIAL MARKET. 4. SPORTS: - PLAYGROUND EQUIPMENT, VARIOUS BALLS, GOLF CLUBS, SWIMMING POOLS, AND PROTECTIVE HELMETS ARE OFTEN PRODUCED FROM

K N A H T OU Y
- Slides: 33