Preexposure Prophylaxis Implementation issues Grant Colfax MD Director
















- Slides: 17

Pre-exposure Prophylaxis: Implementation issues Grant Colfax, MD Director of HIV Prevention and Research San Francisco Department of Public Health

Acknowledgements • • • Albert Liu Susan Buchbinder Robert Grant Jared Baeten Connie Celum

Frequently asked questions regarding Pre. P Implementation • If proved efficacious, will Pre. P be effective? • Is Pre. P scaleable such that it will have a public health benefit? • How will Pre. P integrate into other prevention efforts? • How best address concerns HIV prevention will become “medicalized? ”

More implementation concerns • Politics! Often block or delay efficacious interventions – Needle exchange, birth control, HPV vaccination • Pre. P has potential to increase disparities – Access to Pre. P likely to reflect access to care overall • Who gets Pre. P and for how long? • Pre. P users may face stigma/discrimination

Pr. EP implementation: Important considerations beyond the trials • What level of Pr. EP efficacy and strength of evidence would warrant widespread delivery? • Which settings would be most appropriate for Pr. EP? – Clinics (primary/HIV care), community-based organizations, pharmacies? – How best to integrate HIV testing, risk reduction and adherence counseling, safety monitoring? – Resources for provider training and support • How best to target high risk populations? – High-risk men and women, sex workers, discordant couples, IDUs • How best to communicate about Pr. EP? – Managing expectations of Pr. EP and current trials • How much will Pr. EP cost, and who will pay for Pr. EP? Slide from Al Liu AVAC Pr. EP Primer. August 2009

Important Pr. EP considerations and remaining questions after trials completed • Adherence / pill taking • Safety • Risk compensation / behavior change - -in setting of known efficacy • Resistance • Implementation From Al Liu

Adherence / Pill taking • Current trials all using daily dosing of TDF or FTC/TDF • Provides best case scenario for proof-of-concept • Promoting pill taking and accurate adherence measurement are critical for current trials • Different measurement strategies include pill counts, self-report, MEMS, diaries, biomarkers (drug levels in plasma, PBMCs, and hair) • Novel strategies for promoting adherence (next step counseling approach, cell phones, home visits) • Adherence will be important during implementation – Can daily use be sustained over long periods of time? – Is daily use needed for protection? From Al Liu

Safety • TDF and FTC/TDF have good safety profiles in HIV-positive men and women • Current Pr. EP trials will provide substantial safety information in healthy HIV-negative populations • Additional data needed on long-term safety – Current trials exclude pregnant and breast-feeding women, adolescents – Renal, BMD effects w/ TDF-based regimens • Implementation issues – What frequency of safety monitoring is needed? From Al Liu

Risk compensation • Clinical trials will provide data on the effect of Pr. EP on risk behavior in the context of clinical trials – Intensive risk reduction counseling and testing – Reminders of placebo controlled trial, efficacy not yet known • Implementation issues – How will risk change with open-label drug and when efficacy is known? – What are best counseling messages to minimize risk compensation and maximize adherence? From Al Liu

Drug resistance? • Efficacy of Pr. EP will be important – The best way to prevent resistance is to prevent HIV • Individuals who seroconvert during Pr. EP study participation will immediately stop medication – Monthly HIV testing to reduce likelihood of developing resistance • Resistance to TDF (K 65 R) and to FTC (M 184 V/I) comes at a cost to the virus • Implementation issues: – What frequency of HIV testing should be used? – Ongoing data on resistance should be collected From Al Liu

Slide courtesy of Al Liu

Who will pay for Pre. P? • Estimated 50% of HIV infected persons in US not on therapy • 21% of persons with HIV in SF unmet need for medical care • ADAPT programs in many states are limiting coverage • Prevention funding streams don’t pay for medications • Off label issues

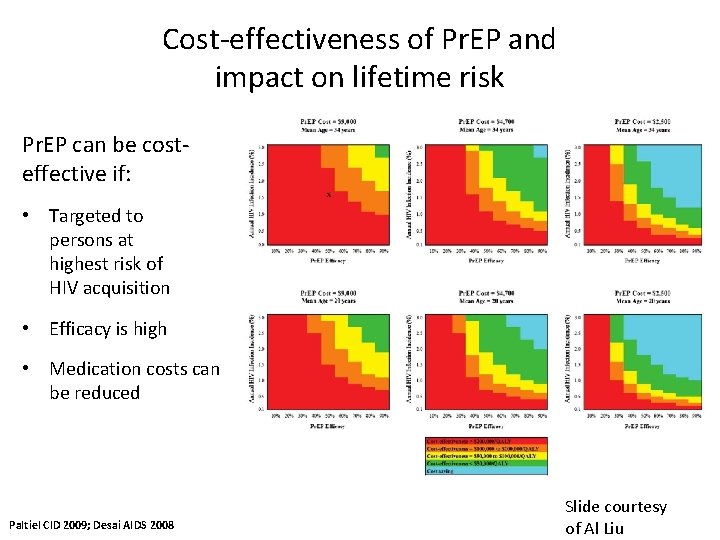
Cost-effectiveness of Pr. EP and impact on lifetime risk Pr. EP can be costeffective if: • Targeted to persons at highest risk of HIV acquisition • Efficacy is high • Medication costs can be reduced Paltiel CID 2009; Desai AIDS 2008 Slide courtesy of Al Liu

Substance users and Pre. P • Access • Adherence – We know HIV-infected drug users can adhere to ART – We know clinicians cannot predict who will or will not adhere – Paucity of effective adherence interventions • Effect of other comorbid conditions (eg, mental illness) • Integration with tx services

Will episodic substance users adhere to Pre. P? • Episodic use associated with high-risk sexual behavior • Binges may be time where exposure to Pre. P may be most important – May be times where adherence is low – Potential for resistance may be high

Planning for implementation: What’s happening nationally? • Community HIV/AIDS Mobilization Project (CHAMP) has formed Pr. EP Prevention Research Advocacy Working Group – Collaborative community representatives drawn from research, policy and advocacy – Working groups on implementation, research, and communications/outreach • CDC Pr. EP planning activities – – – Pr. EP Implementation Planning Working Group Discussions with broad range of stakeholders Outreach and community preparedness Plan for development of Public Health Service guidelines for Pr. EP Develop risk assessment tools Plan for implementation pilots and research • We need to fund implementation and evaluation research NOW! Adapted from Al Liu

What’s going on in your network re: Pre. P? • • • Community education/planning Implementation Cost Scaleability? Specific concerns re: certain populations? What should HPPC be doing now re: preparing for Pre. P (Prep Pre. P)?