PreExposure Prophylaxis Pilot Implementation in the Dominican Republic

Pre-Exposure Prophylaxis Pilot Implementation in the Dominican Republic- Pr. EP-DR Robert Paulino-Ramirez, MD, DTM&H, HIVS Director Instituto de Medicina Tropical & Salud Global Universidad Iberoamericana-UNIBE Dominican Republic
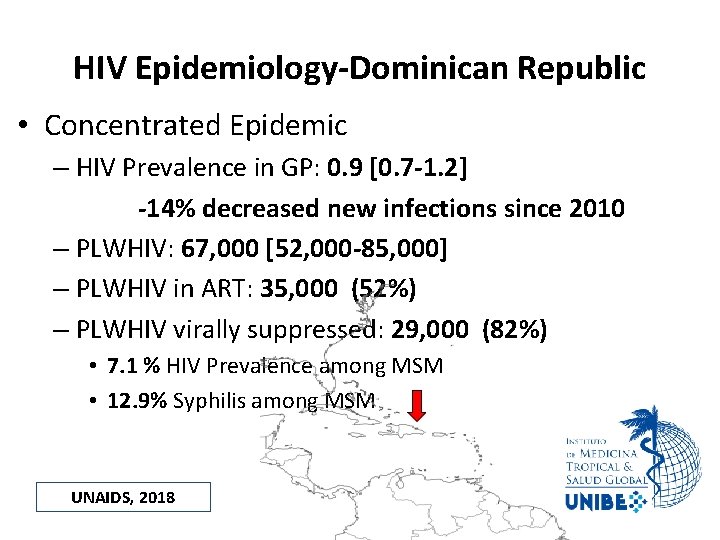
HIV Epidemiology-Dominican Republic • Concentrated Epidemic – HIV Prevalence in GP: 0. 9 [0. 7 -1. 2] -14% decreased new infections since 2010 – PLWHIV: 67, 000 [52, 000 -85, 000] – PLWHIV in ART: 35, 000 (52%) – PLWHIV virally suppressed: 29, 000 (82%) • 7. 1 % HIV Prevalence among MSM • 12. 9% Syphilis among MSM UNAIDS, 2018

Una iniciativa, Un País How We Started? * Approval from the National Bioethical committee was obtained for this pilot implementation.

Training of Health Care Personnel

Elegibility Criteria HIV Negative Status No clinical findings of Acute Retroviral Syndrome Significative Risk for HIV acquisition* No contraindicattions to Pr. EP drugs (TDF/FTC or TDF/3 TC) • Creatinine levels GFRe: 60 m. L/min • Willing to take Pr. EP ______________________ • • * Risk assessment: – Anal or vaginal Sexual Contact without condoms, OR – Recent history (Last 6 months) of any syndromic or diagnosed STI, OR – Recent use of Post-Exposure Prophylaxis-PEP, OR – Serodiscourdant HIV couple.
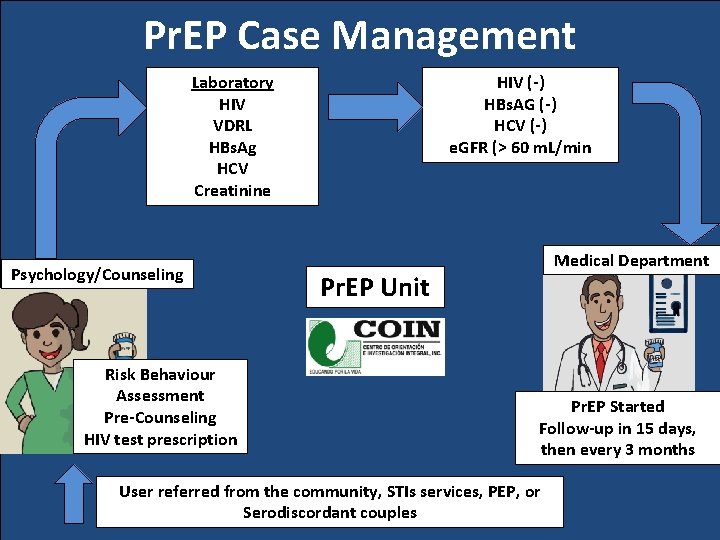
Pr. EP Case Management Laboratory HIV VDRL HBs. Ag HCV Creatinine Psychology/Counseling Risk Behaviour Assessment Pre-Counseling HIV test prescription HIV (-) HBs. AG (-) HCV (-) e. GFR (> 60 m. L/min Medical Department Pr. EP Unit Pr. EP Started Follow-up in 15 days, then every 3 months User referred from the community, STIs services, PEP, or Serodiscordant couples

Monitoring of Data

Results * 942 85% 100% 32% 31% MSM 50% MSM HIV tests done to MSM/TGW Negative results [n=849] Pr. EP offered [n=849] Pr. EP Started [n=271] Acceptability Rates * Data uptaded as July 15 th, 2019

Results (2) 24% Positive Syphilis Test on Baseline 12% Evidence of HBV vaccination 79% 1% 5% 20% e. GFR >100 m. L/min HPV-related dysplasias Assymptomatic Chlamydia infections* Referred Condom Use on Baseline *C. trachomatis/N. gonorrheae was detected by NAAT in Urine (As July 15 th, 2019)

Results Follow-up (12 months) 2% New Positive Syphilis Tests 20% Increase of HBV vaccination 81% 72% e. GFR >100 m. L/min 37% Use of Condoms while in Pr. EP Persistence rate* * Persistence Rate was defined as a continuous use of Pr. EP drugs during the pilot implementation (12 months)

Scaling-Up Pr. EP in the DR • First draft for a National Pr. EP Implementation Strategy released last year • National Pr. EP guidelines are currently on revision to be published soon

Publishing
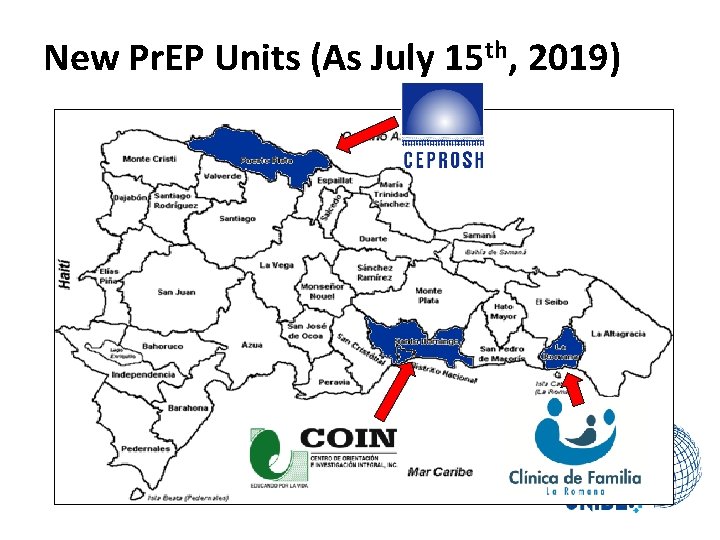
New Pr. EP Units (As July 15 th, 2019)

Conclusions • • • Acceptability was good PEP and voluntary partner referral services opportunity to increase the offer. Many opportunities for a comprehensive approach to STIs not limited to HIV • • Opportunities for HBV detection/treatment vaccination, and Syphilis early Side effects were not significant Importance of mental health interventions and Behavioral modifications It is required more training to health care providers (evidence -based approach of Pr. EP effectiveness)

Acknowledgements - To the DR MOH Team, specially DIGECITSS- Dr. Feliz Báez and Dr. José Ledesma - DR CDC - To PAHO local officer Dr. Rafael Rodríguez - COIN’s Team: Lic. Santo Rosario and Dra. Merelin Muñoz - My Pr. EP Team: - Mayra Rodriguez, MSc - Leandro Tapia, MD - Aneska Mariño, MD - Luis Ortiz, MD - Daniel Reyes, BA - Juana Clase - CBO working with MSM/TGW in the country

Thanks!! Robert Paulino-Ramirez, MD, DTM&H, HIVS Researcher/ Professor Instituto de Medicina Tropical & Salud Global Universidad Iberoamericana-UNIBE Santo Domingo, Dominican Republic 22333 ✉�r. paulino 1 @unibe. edu. do �� www. imtsag. unibe. edu. do
- Slides: 16