Pr EP Preexposure Prophylaxis for HIV Brought to

Pr. EP Pre-exposure Prophylaxis for HIV Brought to you by Sara Hayes shayes@mainefamilyplanning. org

Disclosures Financial: none Other: I’m glad I don’t have to do this kind of talk for a living

Who all……?

DEFINITIONS • HIV Pr. EP: – Pre- Exposure Prophylaxis – Use of a combination of antiretroviral medications in an HIV-negative person before HIV exposure to prevent HIV acquisition – Truvada, Emtricitabine /Tenofovir , TDF/FTC, is the only FDA-approved medication for HIV Pr. EP

• Refers to Post Exposure Prophylaxis started within 72 hrs of an HIV exposure • combination of 2 antiretroviral meds (ART) Truvada (emtricitabine + tenofovir) + dolutegravir 50 mg •

• asap after rapid HIV negative status obtained or non-rapid HIV test sent. • Both are taken once daily for 28 days • May switch to Pr. EP after the 28 days

Just Say “NO” ain’t work’n • Approximately 40, 000 people in the United States are infected with HIV each year*. • From 2008 through 2014, estimated annual HIV incidence declined 18% overall but progress was uneven. • *Centers for Disease Control and Prevention. Fact sheet: HIV incidence: Estimated annual infections in the U. S. , 2008 -2014 -Overall and by transmission route. https: //www. cdc. gov/nchhstp/newsroom/docs/factsheets/hiv-incidencefactsheet_508. pdf Accessed 27 Feb 2017.

Just Say “NO” ain’t work’n • declines among heterosexuals, PWID (IDU), and white MSM, • no decline among black MSM • an increase was documented among Latino MSM.

In 2015, transmission mode • 67% of the 39, 513: male-male sexual activity without injection drug use, • 3% : male-male sexual activity with injection drug use, • 24%: male-female sexual contact without injection drug use, and • 6%: injection drug use.

In 2015, • Among the 24% of persons with newly diagnosed HIV infection attributed to heterosexual activity, 64% were African. American women and men*. • *Centers for Disease Control and Prevention. HIV Surveillance Report, 2015; vol 27. https: //www. cdc. gov/hiv/pdf/library/reports/surveillance/cdc-hivsurveillance-report 2015 -vol-27. pdf Accessed 27 Feb 2017.

Pr. EP Effectiveness • Pr. EP efficacy is highly dependent upon adherence to the medication. • If taken once daily as prescribed, TDF-FTC reduces the risk of HIV infection by at least 90%. • Protection from HIV diminishes progressively with lower levels of adherence. • Grant RM et al. Preexposure chemoprophylaxis for HIV prevention in men who have sex with men. N Engl J Med. 2010; 363: 2587.

PREP PRINCIPLES • Pr. EP should not be offered as a sole intervention for HIV prevention. • Pr. EP should only be prescribed as part of a comprehensive prevention plan.

KEY PREP PRINCIPLES • Every effort is made to make sure that a patient on or seeking Pr. EP is not HIV positive or in the window period for becoming positive because drug resistant HIV has been identified in people with undetected HIV who subsequently received Truvada as Pr. EP.

KEY PREP PRINCIPLES • Pr. EP is indicated for individuals who have a documented negative HIV test result and are at ongoing high risk for HIV infection. • A negative HIV test result needs to be confirmed as close to initiation of Pr. EP as possible, ideally on the same day the prescription is given. • Clinicians should wait to prescribe Pr. EP until confirmation of a negative test result is available.

KEY PREP PRINCIPLES • Efficacy of Pr. EP is dependent on adherence. Pr. EP should only be prescribed to those who are able to adhere to the regimen and express a willingness to do so

KEY PREP PRINCIPLES • Although consistent condom use is a critical part of a prevention plan, lack of use of barrier protection is not a contraindication to Pr. EP. • Pr. EP may help protect the HIV seronegative partner in a serodiscordant relationship during attempts to conceive.


Indications for Pr. EP Men who have sex with men (MSM) who: • Engage in condomless anal sex outside of a mutually monogamous relationship with an HIV-uninfected sexual partner, or • Have been diagnosed with a sexuallytransmitted infection in the last 6 months, or • Have an HIV-infected sexual partner;

Indications for Pr. EP Heterosexually-active people who: • Engage in condomless sex with a partner or partners of unknown HIV status who are at high risk of infection (for example, people who inject drugs or bisexual men) or • Have an HIV-infected sexual partner

Indications for Pr. EP Injection drug users • especially those who share injection equipment.

CONTRAINDICATIONS • Estimated creatinine clearance (e. Cr. Cl) < 60 m. L/min • HIV infection, documented or suspected

3 Steps to Pr. EP

1. Determine eligibility and perform baseline testing • High risk based on sexual and/or drug use hx • Negative HIV antibody-antigen test, and no symptoms of acute HIV in the prior four weeks • Serum creatinine to ensure the estimated creatinine clearance is > 60 m. L/minute • Hepatitis B surface antigen to assess for chronic hepatitis B infection • Preg test for those with childbearing potential

# 2 Prescribe • Tenofovir disoproxil fumarate-emtricitabine 300/200 mg 1 tablet by mouth daily • No more than a 90 -day supply of medication at any given time

#3: Monitor • HIV antibody/antigen test every three months • Preg Test q three months if risk for pregnancy • Serum creatinine at three months, then every six months if stable • STI screen at least every six months • Risk reduction counseling at every visit • Re-assess the need for Pr. EP at least once per year

Initial evaluation • Sexual history • Substance use history, with specific queries of IDU use • HIV exposure or high-risk event within the past 72 hours – Consider immediate PEP followed by a seamless transition to Pr. EP if indicated

MEDICAL HISTORY Inquire specifically about any history of: • kidney disease • liver disease • chronic hepatitis B infection. • bone disease – osteoporosis – pathologic or fragility bone fractures

Medication History • Document an accurate medication list with special attention to NSAIDs. – Acyclovir, valacyclovir, cidofovir, ganciclovir, valganciclovir, aminoglycosides, high-dose or multiple NSAIDs, or other drugs that reduce renal function or compete for active renal tubular secretion: Serum concentrations of these drugs and/or TDF may be increased. Monitor for doserelated renal toxicities.

ACUTE RETROVIRAL SYNDROME ASSESSMENT • Inquire about the presence of fever, fatigue, myalgia, rash, headache, sore throat, cervical adenopathy, arthralgia, night sweats, or diarrhea within the past month. • If acute or recent HIV infection is suspected, order an HIV RNA test.

STI SYMPTOM ASSESSMENT • Inquire about the presence of dysuria, discharge, anorectal itching or pain, rash, or ulcers. • Test and treat appropriately.

LABS TO ORDER Select appropriate assessment and ICD-10 code: Z 72. 51 “High-risk sexual behavior” and/or Z 20. 6 “HIV exposure” and/or Z 41. 8 “Preventive medication therapy needed” Z 20. 89 “Exposure to a communicable disease”

REQUIRED LABS • HIV antibody – HIV RNA if there is clinical suspicion for acute retroviral syndrome OR IF THEY HAVE HAD SEX WITH A KNOWN HIV+ PERSON WITHIN THE PAST MONTH. HIV RNA is a test of viral load • Serum creatinine (ensure weight is documented for calculation of e. Cr. Cl)

LABS TO ORDER • • • RECOMMENDED LABS Syphilis/rpr GC/CT urine or vaginal GC/CT rectal and pharyngeal for MSM Hbs. Ag, HBs. AB, &HBc. AB HCV antibody (risk-based screening)

COUNSELING “The pill Truvada has two drugs in it that are commonly used to treat HIV in persons who are HIV-positive. When taken daily by people who are HIVnegative, they can block HIV from infecting the body. The pill needs to be taken every day in order for the body to build up sufficient drug levels to block HIV. “

COUNSELING “It cannot be expected to work if it is only taken just before or just after sex. Pr. EP reduces but does not eliminate HIV transmission risk. You still need to use condoms even if you are taking Pr. EP because Pr. EP does not protect against other sexually transmitted diseases. ”

COUNSELING • Efficacy – “If taken daily, Truvada greatly reduces but does not eliminate the risk of getting HIV” • Missed doses – “Take it as soon as you remember that day. Take only 1 dose per day. Do not take 2 doses to make up for a missed dose. ”

COUNSELING • Resistance – “People who become infected with HIV while taking Truvada for Pr. EP are more likely to become resistant (meaning it won’t work) to one or both of the meds in Truvada which is also used to treat HIV/AIDS. ” • Notify provider and have HIV testing prior to restarting Pr. EP if stopped for seven or more days

COUNSELING • Potential side effects – Truvada start-up syndrome with possible symptoms including: • • • Nausea Abdominal upset Loose stools Flatulence Headache – Most symptoms, if they occur, are mild and resolve within a few days to a few weeks

COUNSELING – Renal toxicity • “Because Truvada can cause kidney problems, we will be doing some blood work to check on your kidneys before you start Pr. EP and while you are on it. ” – Bone mineral density loss • “For some people, Truvada can make your bones less dense. This is usually very mild, happening in the first few months and then either stabilizes or goes back to normal.

COUNSELING • Other prevention strategies – Condom use • “While Pr. EP can help protect you against HIV it is not fool proof and it does not protect you against other STIs” – Contraception • Assess for need and provide as indicated • Time to achieving protection – Rectal: seven days – Vaginal: 20 days

COUNSELING • Symptom reporting – Notify staff immediately of symptoms of acute retroviral syndrome or STIs • Refer to educational handout • Long term safety – “Pr. EP has been studied in many clinical trials over the years has shown that Pr. EP has been used safely since 2004. ”

COUNSELING • Sharing medication – Pr. EP is exclusively for personal use and should not be shared with others • Follow-up expectations – Provider visit and labs at least every three months – Expect notification upon provider’s receipt of initial lab results • Prescription will be sent if okay to start Pr. EP

COUNSELING • Reasons for discontinuing Pr. EP – + HIV test – Development of kidney disease – Use of medication for untended purposes or sharing – Non adherence to medication or appointments – Change in risk behaviors… ie Pr. EP no longer needed.
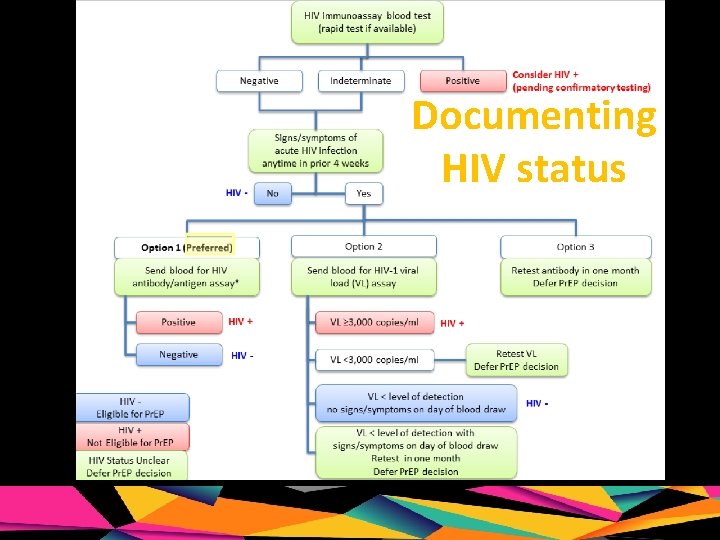
Documenting HIV status

STARTING Pr. EP UPON RECEIPT OF SCREENING LAB RESULTS To start Pr. EP: • HIV antibody and RNA, if ordered, should be nonreactive/not detected within the past 10 days • Repeat HIV antibody test if more than 10 days have elapsed an RNA test was not ordered
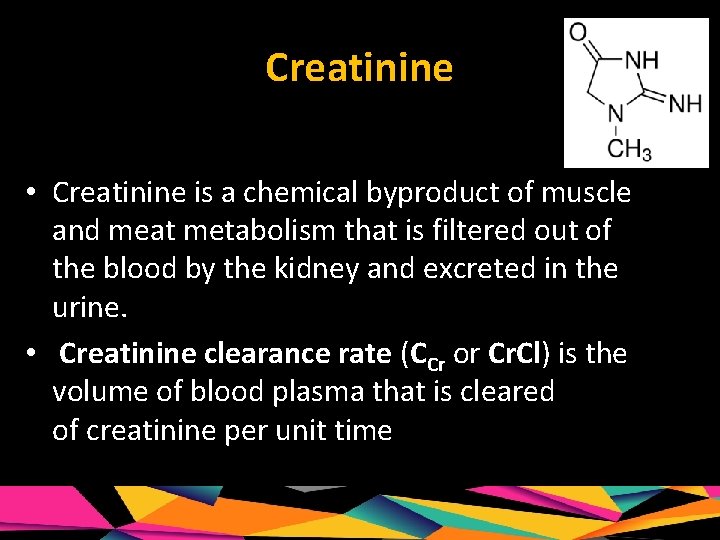
Creatinine • Creatinine is a chemical byproduct of muscle and meat metabolism that is filtered out of the blood by the kidney and excreted in the urine. • Creatinine clearance rate (CCr or Cr. Cl) is the volume of blood plasma that is cleared of creatinine per unit time

Creatinine • Calculating Creatinine Clearance • http: //www. mdcalc. com/creatinine-clearancecockcroft-gault-equation/ • Need to know: sex, age, weight, creatinine level

Creatinine • Cr. Cl must be > 60 m. L/min • If < 60 m. L/min, provider to follow up appropriately. Consider repeat testing • Follow up with other lab results appropriately. Do not wait for STI, HCV antibody, or other non-essential lab results to start Pr. EP.

Ok to start…. • Contact patient to inform whether cleared to start Pr. EP • Order creatinine for next visit (schedule for about 11 wks) • Consider ordering HIV as well if pt wants to get done at lab ahead of time instead of at clinic
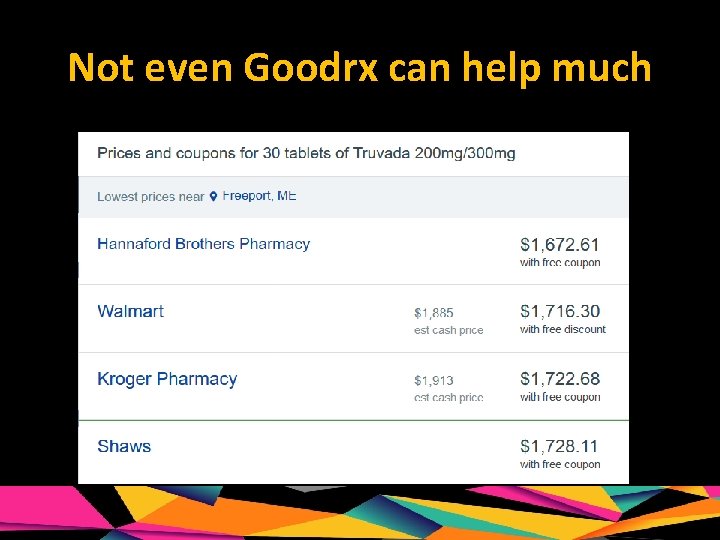
Not even Goodrx can help much

Patient assistance program 1. If Insured: -Gilead HIV Co-pay Assist. Coupon Card www. gileadcopay. com, 1 -877 -505 -6986. 2. If not insured: -Gilead US Advancing Access Program 1 -800 -226 -2056, can download enrollment form -Partnership for Prescription Assistance Program 1 -888 -477 -2669, www. pparx. org

But what I do…. • If patient has no insurance or has private insurance I send the order to Apothecary by Design in Portland. – They do the pt assistance/copay assistance part – If PA needed, I do that – Will send the meds to the patient at home – Does follow-up calls with them • Pt has Mainecare… they pay for it.

3 month visits: ASSESSMENT • Side effects, – e. g. , headache, nausea, loose stools, flatulence – Consider symptom management with OTC medications • • • Acute HIV and STI symptoms Adherence HIV risk assessment and sexual history Desire to continue Pr. EP Changes to medical history

3 month visits: ASSESSMENT • Contraception for women • Review lab results with patient • Symptom-directed physical exam – Ensure weight is documented for calculation of e. Cr. Cl

Labs • HIV test (may have been ordered ahead to be done with creatinine) – RNA test if any sx of viral infection • STI testing as appropriate • PT if needed • Order next creatinine for next visit

HIV TESTING AFTER Pr. EP INITIATION • HIV antibody testing should be conducted at least every three months for patients taking Pr. EP. • An HIV RNA test should be ordered whenever there is clinical suspicion for acute retroviral syndrome.

HIV TESTING AFTER Pr. EP INITIATION • HIV testing should be conducted for patients who have stopped Pr. EP (whether held by clinician or by patient) for seven or more days prior to resuming Pr. EP. • HIV antibody and RNA, if ordered, must be nonreactive/not detected within the past 10 days prior to restarting Pr. EP. • Repeat HIV antibody test if more than 10 days have elapsed an RNA test was not ordered

CREATININE MONITORING • Creatinine should be checked at least every three to six months during Pr. EP use. • Consider more frequent monitoring if clinically indicated, e. g. , in the setting of diabetes or uncontrolled hypertension. – If the e. Cr. Cl is < 60 m. L/min, Truvada should be held immediately via a telephone encounter and the creatinine should be repeated in two to four weeks. Include HIV antibody testing. – If the rechecked e. Cr. Cl is ≥ 60 m. L/min and HIV antibody is nonreactive, Pr. EP may be restarted and creatinine should be rechecked again in one month

CREATININE MONITORING • If the creatinine is > 1. 5 x baseline (but e. Cr. Cl is ≥ 60 m. L/min), initiate a telephone encounter and assess for any other potential causes of the creatinine elevation, e. g. , dehydration, protein supplement use, new medications, and NSAIDs. • Advise patient to continue employing other prevention strategies as Pr. EP may need to be held. • Recheck creatinine in two weeks. • If the rechecked e. Cr. Cl is < 60 m. L/min, Truvada should be held immediately via a telephone encounter. • Patients who want to be on Pr. EP but have sustained creatinine elevations > 1. 5 x baseline and/or e. Cr. Cl < 60 m. L/min should be referred to a nephrologist.

Refill policy – Pharmacies may send automatic refill requests well in advance of patients’ running out of pills. These requests should be denied as lab testing likely will not yet have occurred. – No more than 90 tabs at any given time should be provided. – If a patient does not attend a provider visit, but has completed his/her labs as required and results are normal, one refill may be provided. The patient must reschedule and attend a provider visit for further refills.

DISCONTINUING Pr. EP • By clinician – Sustained e. Cr. Cl < 60 m. L/min – HIV seroconversion • By patient – Provide counseling on HIV risk reduction and education on safely restarting Pr. EP. – Advise continued use of Truvada (as PEP) if there was a highrisk event within the last seven days and the patient had been adherent to Pr. EP. – Continue for 28 days post-high-risk event – Conduct HIV antibody testing four weeks after Truvada discontinuation for any reason other than seroconversion. – Prevention strategies.

IMPORTANT CONSIDERATIONS • Does the patient have chronic active hepatitis B virus (HBV) infection? – TDF/FTC is active against HBV infection. – may be used simultaneously as treatment for HBV infection and as Pr. EP. – Discontinuation of TDF/FTC in a pt with Hep B can cause rebound viremia.

IMPORTANT CONSIDERATIONS • Is the patient pregnant or attempting to conceive? – Is the partner virologically suppressed with ART? – Sex limited to time of ovulation? If yes and yes, Pr. EP might not be that useful?

IMPORTANT CONSIDERATIONS – Clinicians should counsel patients who are pregnant or who are trying to become pregnant that no major safety concerns with TDF-FTC for Pr. EP in pregnancy have been identified but that experiences with the medication in this context are limited. – Breast feeding on Pr. EP is ok.

IMPORTANT CONSIDERATIONS • Benefits to continuing during pregnancy – Decreased risk of acquisition of acute HIV infection during pregnancy, which is a significant risk factor for mother-to- child HIV transmission • Potential toxicity – Available data suggest that TDF/FTC does not increase risk of birth defects; however, there are not enough data to exclude the possibility of harm

IMPORTANT CONSIDERATIONS • Is the patient an adolescent? • CDC and the International Antiviral Society-USA have now extended the use of TDF/FTC to include Pr. EP for adolescents at high sexual or other behavioral risk for HIV infection.

IMPORTANT CONSIDERATIONS • ls the patient at risk for chronic kidney disease (>65 years of age, black race, hypertension, or diabetes)? – Discuss possibility of kidney disease with individuals who have pre-existing risk factors.

IMPORTANT CONSIDERATIONS Does the patient have osteopenia/osteomalacia/osteoporosis? • There may be a risk of bone loss associated with tenofovir. – Discuss risk of bone loss with individuals with preexisting risk factors or demonstrated osteoporosis/osteomalacia/osteopenia.

People with HIV-infected sexual partners • The benefit of Pr. EP may be minimal for people who are in monogamous sexual relationships with HIV-infected but virologically suppressed partners.

People with HIV-infected sexual partners • a large observational study of both heterosexual and MSM serodifferent couples, indicated that people living with HIV who are virologically suppressed are not sexually infectious. • Rodger AJ, et al. Sexual activity without condoms and risk of HIV transmission in serodifferent couples when the HIV-positive partner is using suppressive antiretroviral therapy. JAMA. 2016; 316(2): 171

People with HIV-infected sexual partners • Pr. EP may still be indicated for the HIVuninfected partner in such couples if that person engages in sexual activity with people outside of the partnership.

Same-day access to Pr. EP • Advantages • Less loss to followup? • Risk of HIV acquisition while waiting for Pr. EP • Disadvantages • Logistical challenges • Lengthy initial visit

Same-day access to Pr. EP 1. counsel the patient about Pr. EP and provides written educational materials 2. draw serum creatinine, HIV antibody/antigen, hepatitis B surface antigen (+ routine STI screening/treatment, as needed) 3. Prescription for 30 days of TDF-FTC

• https: //www. lgbthealtheducation. org/prepaction-kit/

Pr. EP Action Kit • Tips on taking a comprehensive sexual history • Frequently asked questions about Pr. EP • A pocket card for Pr. EP prescribing and monitoring • Information on Pr. EP use in special populations, such as pregnant people and adolescents

Pr. EP Action Kit • Resources to make clinical environments more welcoming to LGBT people, since the HIV epidemic is more prevalent in some LGBT populations • 7 Steps: To Affirmative Health Care for LGBT People • Tips on taking a history of sexual health • Pre-exposure Prophylaxis for HIV (Pr. EP) and Special Populations

Helplines • Pr. EPline, 855 -448 -7737 The CCC Pre-Exposure Prophylaxis Service 11 a. m. – 6 p. m. EST • PEPline (888) 448 -4911 non-occupational PEP 9 a. m. – 8 p. m. ET Monday – Friday, and 11 a. m. – 8 p. m. ET on weekends & holidays.

Stuff I probably won’t get to but you may want to know about or not.

HIV Testing for Dummies Most of us Different HIV tests work by looking for three different things. • Proteins on the surface of the virus (antigens/Ag) like protein 24 (called p 24). • An immune response to the virus (antibodies/Ab). • Genetic material from the virus (HIV RNA or DNA).

4 th generation (Ag/Ab) • are recommended four weeks after exposure. They give an earlier result than antibody-only tests that are recommended after six weeks. • detect over 95% of infections at four weeks after exposure. – As with antibody only tests, a small percentage of people (less than 5%) may have a delayed response to HIV. So a negative test at four weeks needs to be confirmed after three months.

Viral load (RNA PCR) test • looks directly for HIV in blood. • It has the shortest potential window period and can be used from 3 days to 4 weeks after an exposure. • After infection, viral load is usually very high within the first 4 weeks and so this test can be used to confirm a suspected infection if someone has symptoms.

Viral load (RNA PCR) test • If symptoms are related to HIV, then the viral load test will be positive. HIV symptoms are related to viral load.

Viral load (RNA PCR) test In adults, viral load tests are only usually offered when there is both: • A recent high risk exposure (ie condom break with a known HIV positive partner who is not on treatment); and • Symptoms of HIV infection (fever, extreme tiredness, heavy ‘flu-like illness etc).

Antibody testing • Most people generate an antibody response within 4 weeks, but occasionally it can take longer. • Although Ag/Ab tests are preferred due to shorter window period, other rapid tests are acceptable (but not the oral ab tests)

P 24 Antigen tests P 24 • These are viral proteins that make up most of the virus. Produced 2 -3 wks after infection and before AB. • Only detected for 1 -2 months but by the time the p 24 levels have dropped antibodies will be present.

Average Time After Exposure to Detect HIV Ag/Ab

What’s new? • Dapivarine vag ring (being studied in sub Saharan Africa) – minimal absorption elsewhere in the body. – Women insert the product themselves and replace it every month. • Dapivarine vag ring + contraceptive

IPERGAY • (INTERVENTION PRÉVENTIVE DE L’EXPOSITION AUX RISQUES AVEC ET POUR LES GAYS) • ON Demand Pr. EP • Randomized blinded study 400 MSM France and Canada • Molina JM, Capitant C, Spire B, et al. On-demand preexposure prophylaxis in men at high risk for HIV-1 infection. N Engl J Med. 2015; 373(23): 2237 -2246.

IPERGAY • 2 pills (TDF/FTC or placebo) between 2 and 24 hours before sex, • 1 pill 24 hours after the first dose, • 1 pill 48 hours after the first dose, • continuing daily pills if sexual activity continues until 48 hours after the last sex.

IPERGAY • If more than a 1 week break occurred since the last pill, retreatment initiation was with 2 pills before sex or if less than a 1 week break occurred since the last pill, retreatment initiation was with 1 pill before sex. • Each pre-sex dose was then followed by the 2 post-sex doses.

IPERGAY • In the blinded phase of the trial, efficacy was 86% (95% CI: 40 -98). • By self-report, patients took a median of 15 pills per month. • By measured plasma drug levels in a subset of those randomized to TDF/FTC, 86% had TDF levels consistent with having taken the drug during the previous week.

IPERGAY redux • All of the open-label study participants were provided peri-coital Pr. EP as in the original trial. • After a mean follow-up time of 18. 4 months (IQR: 17. 7 -19. 1), the HIV incidence observed was 0. 19 per 100 py which, compared to the incidence in the placebo group of the original trial (6. 60 per 100 py), represented a 97% (95% CI: 81 -100) relative reduction in HIV incidence.

IPERGAY redux • The one participant who acquired HIV had not taken any Pr. EP in the 30 days before his reactive HIV test and was in an ongoing relationship with an HIV positive partner. • Molina JM, Charreau I, Spire B, et al. Efficacy, safety, and effect on sexual behaviour of on-demand pre-exposure prophylaxis for HIV in men who have sex with men: an observational cohort study. Lancet HIV. 2017: online first: available at : • http: //dx. doi. org/10. 1016/S 2352 -3018(17)30089 -9

Sexual Risk Assessment for MSM • In the past 6 months: • Have you had sex with men, women, or both? • (if men or both sexes) How many men have you had sex with? • How many times did you have receptive anal sex (you were the bottom) with a – man who was not wearing a condom?

Sexual Risk Assessment for MSM • How many of your male sex partners were HIV -positive? • (if any positive) With these HIV-positive male partners, how many times did you • have insertive anal sex (you were the top) without you wearing a condom? • Have you used methamphetamines (such as crystal or speed)?

Sexual Risk Assessment for Heterosexuals • In the past 6 months: • Have you had sex with men, women, or both? • (if opposite sex or both sexes) How many men/women have you had sex with? • How many times did you have vaginal or anal sex when neither you nor your – partner wore a condom?

Sexual Risk Assessment for Heterosexuals • How many of your sex partners were HIVpositive? • (if any positive) With these HIV-positive partners, how many times did you • have vaginal or anal sex without a condom?

Sexual Risk Assessment for Heterosexual • How many of your sex partners were HIVpositive? • (if any positive) With these HIV-positive partners, how many times did you – have vaginal or anal sex without a condom? • In

IDU/PWID Risk Assessment • Have you ever injected drugs that were not prescribed to you by a clinician? • (if yes), When did you last inject unprescribed drugs?

IDU/PWID Risk Assessment • In the past 6 months, have you injected by using needles, syringes, or other drug preparation equipment that had already been used by another person? • In the past 6 months, have you been in a methadone or other medication-based drug treatment program?


Thank you for your rapt attention
- Slides: 102