OxidationReduction Reactions Oxidationreduction reaction a chemical reaction involving
















- Slides: 20

Oxidation-Reduction Reactions • Oxidation-reduction reaction – a chemical reaction involving the transfer of electrons – Oxidation – loss of electrons – Reduction – gain of electrons

Oxidation occurs when: An atom or molecule gains oxygen atoms 2 Mg + O 2 2 Mg. O An atom or molecule loses electrons Mg Mg+2 + 2 e. An atom or molecule loses hydrogen CH 3 OH CH 2 O + H 2

Reduction occurs when: An atom or molecule loses oxygen atoms 2 KCl. O 3 2 KCl + 3 O 2 An atom or molecule gains electrons Cu+2 + 2 e- Cu(s) An atom or molecule gains hydrogen CO + 2 H 2 CH 3 OH

Oxidation-Reduction Reactions – Which element is oxidized? – Which element is reduced?

• Assignment: • Page 651/ section 18 -2: 1 -7

Balancing Oxidation-Reduction Reactions by the Half-Reaction Method • Half reaction – equation which has electrons as products or reactants

Balancing Oxidation-Reduction Reactions by the Half-Reaction Method

Electrochemistry: An Introduction • Electrochemistry – the study of the interchange of chemical and electrical energy • Two types of processes – Production of an electric current from a chemical reaction – The use of electric current to produce chemical change

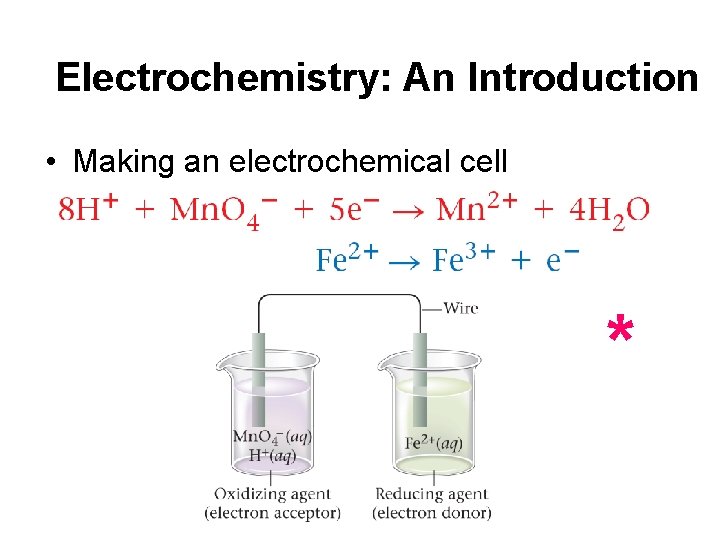
Electrochemistry: An Introduction • Making an electrochemical cell *

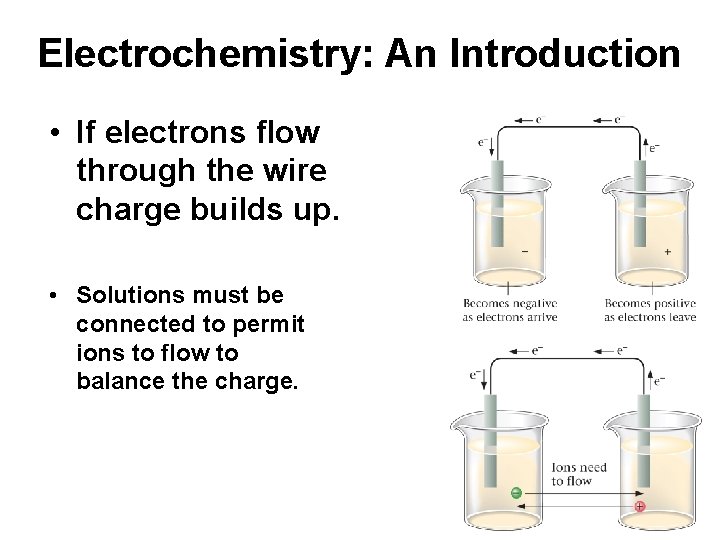
Electrochemistry: An Introduction • If electrons flow through the wire charge builds up. • Solutions must be connected to permit ions to flow to balance the charge.

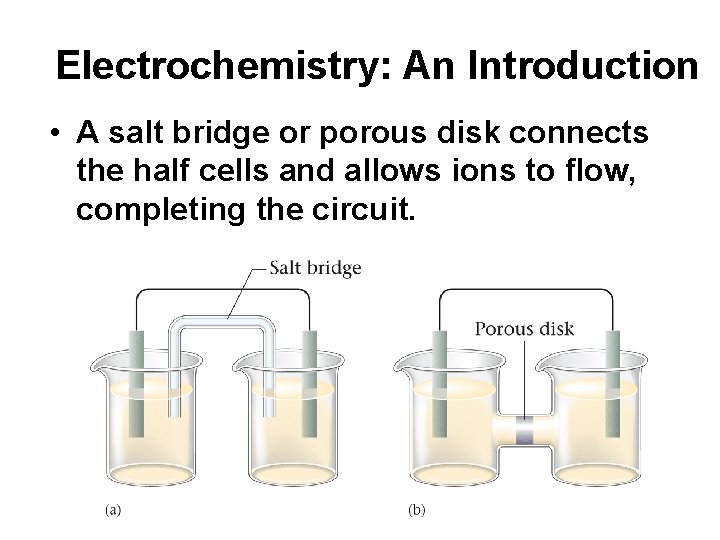
Electrochemistry: An Introduction • A salt bridge or porous disk connects the half cells and allows ions to flow, completing the circuit.

Electrochemistry: An Introduction • Electrochemical battery (galvanic cell) – device powered by an oxidationreduction reaction where chemical energy is converted to electrical energy • Anode – electrode where oxidation occurs • Cathode – electrode where reduction occurs

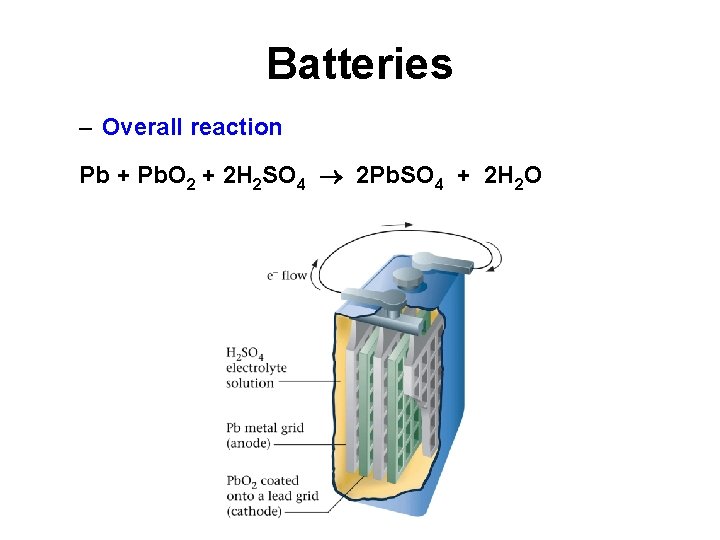
Batteries • Lead Storage Battery – Anode reaction - oxidation Pb + H 2 SO 4 Pb. SO 4 + 2 H+ + 2 e – Cathode reaction - reduction Pb. O 2 + H 2 SO 4 + 2 e + 2 H+ Pb. SO 4 + 2 H 2 O

Batteries – Overall reaction Pb + Pb. O 2 + 2 H 2 SO 4 2 Pb. SO 4 + 2 H 2 O

Batteries • Electric Potential – the “pressure” on electrons to flow from anode to cathode in a battery

Batteries • Dry Cell Batteries – do not contain a liquid electrolyte – Acid version • Anode reaction - oxidation Zn 2+ + 2 e • Cathode reaction – reduction 2 NH 4+ + 2 Mn. O 2 + 2 e Mn 2 O 3 + 2 NH 3 + 2 H 2 O

Batteries • Dry Cell Batteries – do not contain a liquid electrolyte – Alkaline version • Anode reaction - oxidation Zn + 2 OH Zn. O + H 2 O + 2 e • Cathode reaction – reduction 2 Mn. O 2 + H 2 O + 2 e Mn 2 O 3 + 2 OH

Batteries • Dry Cell Batteries – do not contain a liquid electrolyte – Other types • Silver cell – Zn anode, Ag 2 O cathode • Mercury cell – Zn anode, Hg. O cathode • Nickel-cadmium – rechargeable

Corrosion • Corrosion is the oxidation of metals to form mainly oxides and sulfides. – Some metals, such as aluminum, protect themselves with their oxide coating. – Corrosion of iron can be prevented by coatings, by alloying and cathodic protection. * Cathodic protection of an underground pipe

Electrolysis • Electrolysis – a process involving forcing a current through a cell to produce a chemical change that would not otherwise occur *