REDOX OxidationReduction Reactions OxidationReduction Reactions Electrons are transferred








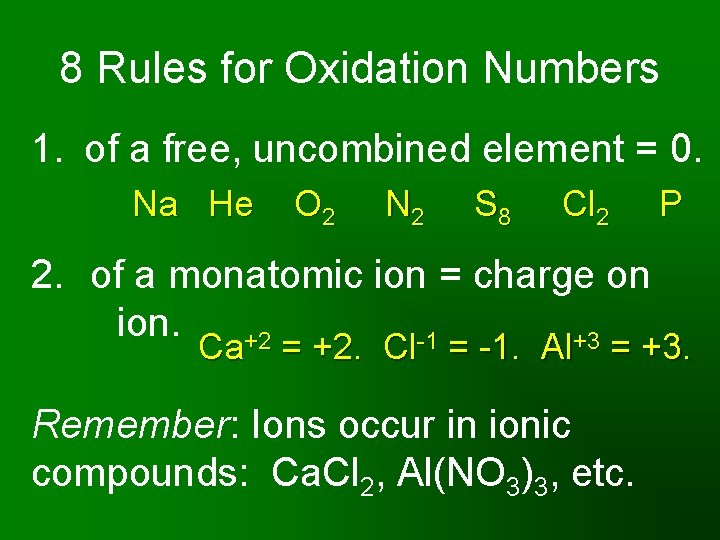



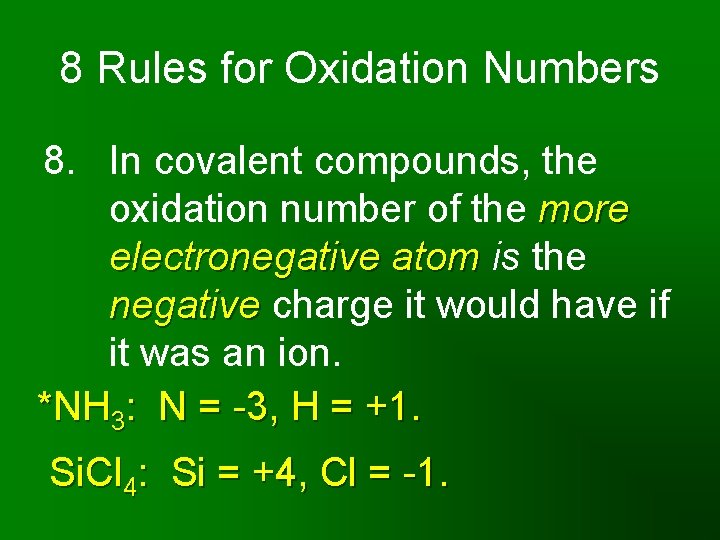















- Slides: 28

REDOX Oxidation-Reduction Reactions

Oxidation-Reduction Reactions • Electrons are transferred from 1 atom to another. • All single-replacement & combustion rxns are redox rxns.

Oxidation = loss of electrons. LOSS of ELECTRONS = OXIDATION LEO

Reduction = gain of electrons. GAIN of ELECTRONS = REDUCTION GER

REDOX • Oxidation & Reduction are complementary. • They occur together & simultaneously or not at all.

LEO GOES GER!!! Oxidation of Cu

Oxidation Numbers • In Ionic Compounds: the number of electrons lost or gained by an atom when it forms ions. Oxidation states of Vanadium

Assigning Oxidation Numbers
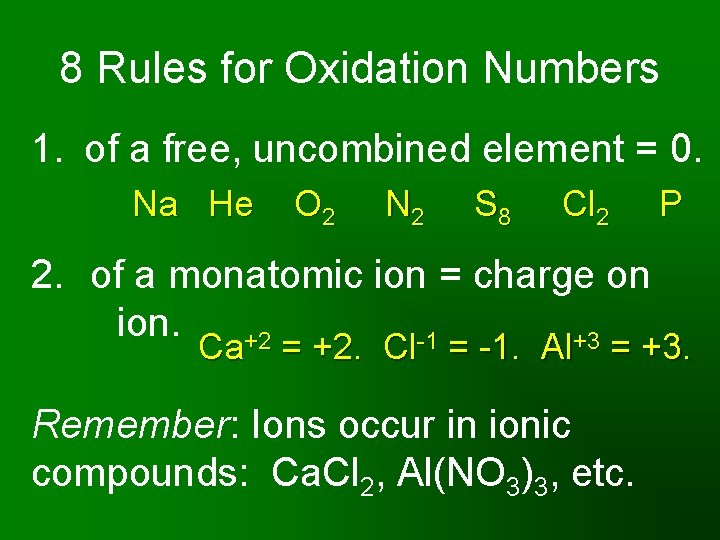
8 Rules for Oxidation Numbers 1. of a free, uncombined element = 0. Na He O 2 N 2 S 8 Cl 2 2. of a monatomic ion = charge on ion. +2 -1 +3 P Ca = +2. Cl = -1. Al = +3. Remember: Ions occur in ionic compounds: Ca. Cl 2, Al(NO 3)3, etc.

8 Rules for Oxidation Numbers 3. Fluorine is always -1. CF 4 4. Hydrogen is nearly always +1, except when it’s bonded to a metal. Then it’s -1. H 2 O, HNO 3, H 2 SO 4 Li. H Ca. H 2 Na. H

8 Rules for Oxidation Numbers 5. Oxygen is nearly always -2 except when its -Bonded to fluorine, where O is +2 OF 2 -In the peroxide ion, where O is -1. O 22 -

8 Rules for Oxidation Numbers 6. The sum of oxidation numbers in a neutral compound is 0. H 2 O CO 2 NO SO 3 7. The sum of oxidation numbers in a polyatomic ion = charge of the ion. Sum in SO 42 - = -2. Sum in NO 3 - = -1.
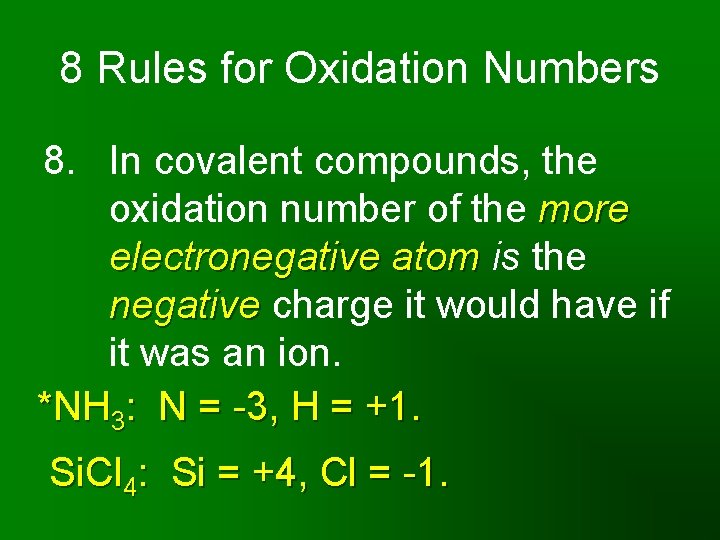
8 Rules for Oxidation Numbers 8. In covalent compounds, the oxidation number of the more electronegative atom is the negative charge it would have if it was an ion. *NH 3: N = -3, H = +1. Si. Cl 4: Si = +4, Cl = -1.

Assign Oxidation Nos • • • K = +1, Cl = -1 KCl Ca. Br 2 Ca = +2, Br = -1 CO C = +2, O = -2 C = +4, O = -2 CO 2 Al(NO 3)3 Al = +3, O = -2, N = +5 Na 3 PO 4 Na = +1, O = -2, P = +5 H 2 S H = +1, S = -2 NH 4+1 N = -3, H = +1 SO 3 -2 S = +4, O = -2

Electrons are Negative! • Why do we use the word “reduced” when electrons are gained? Look at how the oxidation number changes. For example, if Cl gains an electron it becomes Cl-1. The oxidation number decreased from 0 to -1. The oxidation number was reduced.

Writing Equations • Even though oxidation & reduction occur together, we can write separate equations for each process. • Called Half-Reactions. • In order to balance a redox equation, we have to split the full equaton into halfreactions.

Conservation of Mass • # of atoms of each type is the same on both sides of the equation. • Still holds for half-reactions. • Do this step first.

Conservation of Charge • Total charge on LHS must equal total charge on RHS. • In the past, we usually had both sides neutral. (0 = 0. ) • Note: Total charge can be nonzero. Just has to be equal on the 2 sides. • If not balanced, add electrons to whichever side is too positive.

Reduction Half-Reactions Electrons are gained so • I 2 + 2 e- 2 Ithey are like a reactant ! • O 2 + 4 e- 2 O-2 • Half-reactions must demonstrate conservation of mass & conservation of charge. • # of atoms of each element on LHS equals “ “ “ “ RHS. • Total charge on LHS = Total charge on RHS

Oxidation Half-reactions • K K 1+ + 1 e • Fe 2+ Fe 3+ + 1 e • Cu 2+ + 2 e- Electrons are lost so they appear on the product side! • Total Charge on LHS = Total Charge on RHS • # atoms LHS = # atoms RHS

Identifying Half-Reactions • Reduction: electron term is on reactant side. • Oxidation: electron term is on product side.

Vocabulary Interlude • Oxidizing Agent: Is itself reduced. Accepts electrons from something else – aids oxidation for another species. • Reducing Agent: Is itself oxidized. • Loses electrons to something else – aids reduction for another species.

Figuring out what is what! • Given an unbalanced equation. • Goal: Balance it. • Procedure: – Assign oxidation numbers to everything – Split into half-reactions – Balance them separately – “Match” the electrons – Add them together

What’s oxidized & what’s reduced? USE OIL RIG

4 3 2 1 0 -1 -2 -3 -4 2) And if you’re lucky you strike oil & it shoots up 1) You dig down with an oil rig

Oxidizing & Reducing Agents • They are both ALWAYS on the reactant side. • Identify them by seeing how the oxidation 4 numbers change. 3 0 +2 +2 0 Mg + Cu 2+ Mg 2+ + Cu Mg is oxidized, so Mg is the reducing agent! 2 1 0 -1 -2 -3 -4

Oxidizing & Reducing Agents • What’s oxidized & what’s reduced: 0 +2, -1 0 Ca + Fe. Cl 2 Ca. Cl 2 + Fe • Assign oxidation numbers • Figure out what increases & what decreases. Ca is oxidized; Fe 2+ is reduced. Ca = reducing agent; Fe. Cl 2 = oxidizing agnt. 4 3 2 1 0 -1 -2 -3 -4

Oxidizing & Reducing Agents 3, -1 2, -1 4, -1 2 Fe. Br 3 + Sn. Br 2 2 Fe. Br 2 + Sn. Br 4 Fe 3+ is reduced to Fe 2+ Sn 2+ is oxidized to Sn 4+ Fe. Br 3 is the oxidizing agent. Sn. Br 2 is the reducing agent.