REDOX OxidationReduction Reactions OxidationReduction Reactions Electrons transferred from









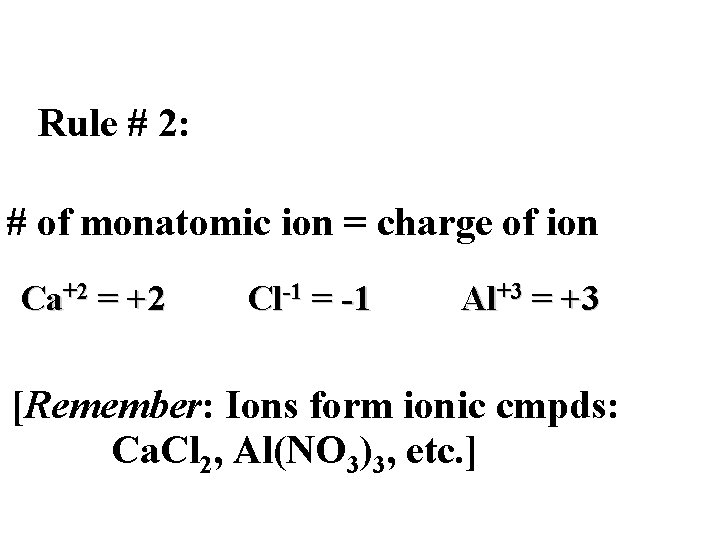


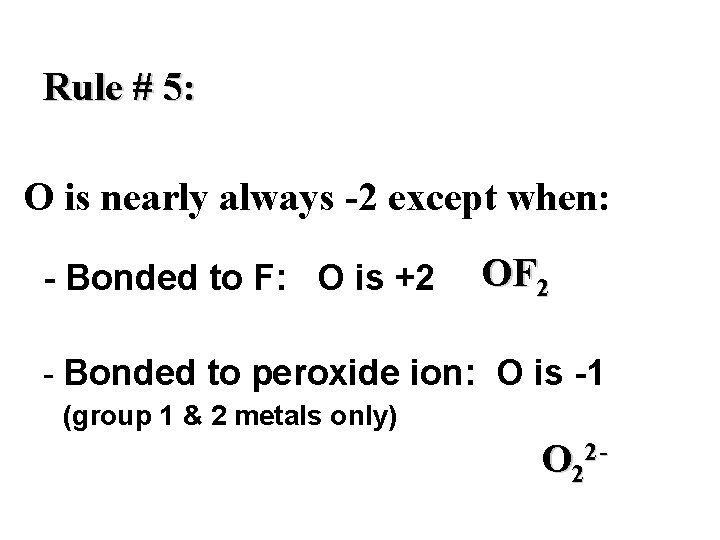









- Slides: 23

REDOX Oxidation-Reduction Reactions

Oxidation-Reduction Reactions • Electrons transferred from 1 atom to another • Redox reactions: – All single-replacement rxns – All combustion rxns

LEO GOES GER!!!

Oxidation = loss of electrons LOSS of ELECTRONS = OXIDATION LEO

Reduction = gain of electrons GAIN of ELECTRONS = REDUCTION GER

REDOX Oxidation & Reduction reactions are complementary reactions • occur together & simultaneously or not at all

Oxidation Numbers In Ionic Compounds: – # of electrons lost/gained determined by ions that form compound

Assigning Oxidation Numbers

Rule #1: # of free, uncombined element = 0 Na He O 2 N 2 S 8 Cl 2 P
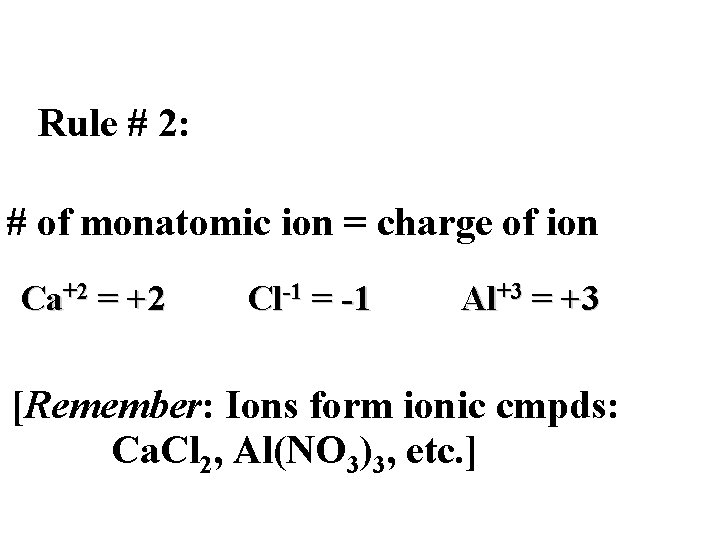
Rule # 2: # of monatomic ion = charge of ion Ca+2 = +2 Cl-1 = -1 Al+3 = +3 [Remember: Ions form ionic cmpds: Ca. Cl 2, Al(NO 3)3, etc. ]

Rule # 3: F is always -1 CF 4

Rule # 4: H is nearly always +1 • except if bonded to metal then -1 H 2 O, HNO 3, H 2 SO 4, Li. H, Ca. H 2, Na. H
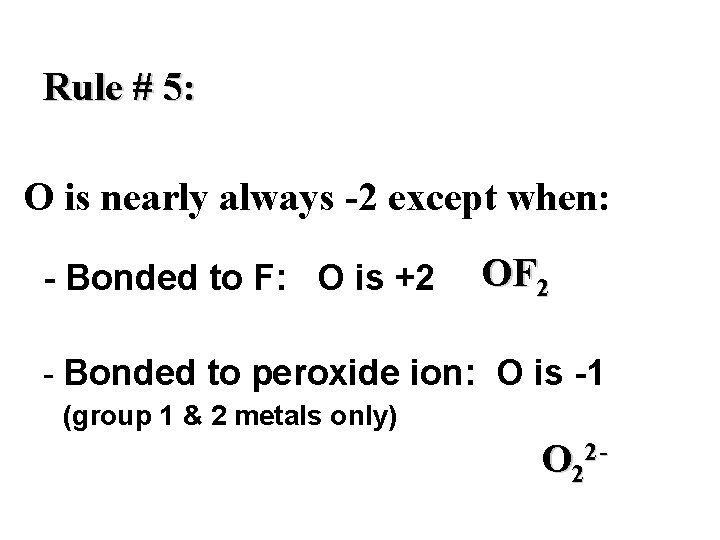
Rule # 5: O is nearly always -2 except when: - Bonded to F: O is +2 OF 2 - Bonded to peroxide ion: O is -1 (group 1 & 2 metals only) O 22 -

Rule # 6 for all neutral compounds: Sum oxidation #’s = 0 S H 2 O CO 2 NO SO 3

Rule # 7 Sum oxidation #’s in polyatomic ion = charge of ion Sum in SO 4 -2 = -2 Sum in NO 3 -1 = -1

Rule # 8 Covalent cmpds: oxidation # more electronegative atom = negative charge (as if was ion) *NH 3: N = -3, H = +1 Si. Cl 4: Si = +4, Cl = -1

Assign Oxidation #’s • KCl K = +1, Cl = -1 • Ca. Br 2 Ca = +2, Br = -1 • CO C = +2, O = -2 • CO 2 C = +4, O = -2 • Al(NO 3)3 Al = +3, O = -2, N = +5 • Na 3 PO 4 Na = +1, O = -2, P = +5 • H 2 S H = +1, S = -2 • NH 4+1 N = -3, H = +1 • SO 3 -2 S = +4, O = -2

4 3 2 1 0 -1 -2 -3 -4 2) And if you’re lucky you strike oil & it shoots up 1) You dig down with an oil rig

Electrons are Negative! • Why use the word “reduced” when electrons are gained? Look how oxidation number changes Ex: Cl gains an electron → Cl-1 • oxidation # ↓ from 0 to -1; the # was reduced

Writing Equations • Even though oxidation & reduction reactions occur together we write separate equations for each process – known as Half-Reactions

Reduction Half-Reactions • I 2 + 2 e- 2 I-1 • O 2 + 4 e- 2 O-2 Electrons are gained so put on reactant side! • Half-reactions must demonstrate: – conservation of mass & conservation of charge – # atoms on left must = # atoms on right – total charge on left must = total charge on right

Oxidation Half-reactions • K K 1+ + 1 e • Fe 2+ Fe 3+ + 1 e • Cu 2+ + 2 e- Electrons are lost so they are put on product side! • Total Charge on left = Total Charge on right • # atoms on left = # atoms on right

Vocabulary Interlude Oxidizing Agent: Substance being reduced – Accepts electrons – aids oxidation for another species Reducing Agent: Substance being oxidized – Loses electrons – aids reduction for another species