NICOTINE Nicotine Chief alkaloid of tobacco plant Nicotina

NICOTINE

Nicotine • Chief alkaloid of tobacco plant (Nicotina tabacum) • Occurs in the plant leaves as salt of malic acid and citric acid to the extent of 4 to 5 percent. • Named after the Frenchman Nicot who introduced tobacco in France in 1560.

Isolation of Nicotine Finely powdwred of tobacco plant extract with dilute acid remove cellulose, chlorophyll etc Water soluble salts of alkaloids made basic with lime or sodium hydroxide steam distillation remove water-soluble nonvolatile materials Steam distille acidified to about p. H 3 with solid oxalic acid concentrated to a syrup cooling Crystalline nicotine oxalate transferred to a separating funnel treated with excess of aqueous KOH Nicotine rises to the surface as brown oil Extract with ether solution evaporate Nicotine

Isolation of Nicotine 1. Waste part of the tobacco plant are finely powdwred and extacted with dilute acid The water soluble salts of alkaloids are thus removed in solution from cellulose, chlorophyll etc. 2. The acid extract is then made basic with lime or sodium hydroxide, and steam distilled. Steam-distillation separates the nicotine from water-soluble nonvolatile materials (sugars, inorganic salts etc) 3. The distillate is acidified to about p. H 3 with solid oxalic acid and concentrated to a syrup. On cooling, the crystalline salt of nicotine and oxalic acid separates. 4. The crystalline nicotine oxalate is then transferred to a separating funnel and treated with excess of aqueous KOH. The nicotine thus set free rises to the surface as a brown oil and separated by extraction with ether. 5. The ethereal solution of the alkaloid is dried and the ether evaporated. For further purification the residue is fractionally distilled under vacuum.

Properties of Nicotine 1. 2. 3. 4. 5. It exists in a liquid form. It is colourless. The boiling point is 246° C It has a tobacco like smell. It is soluble in water and also in organic solvents such as ethanol, ether and benzene. 6. It is a deadly poison to animals. 7. In small quantities, nicotine stimulate the nervous system for a while. A low nicotine content tobacco is used for smoking purposes.
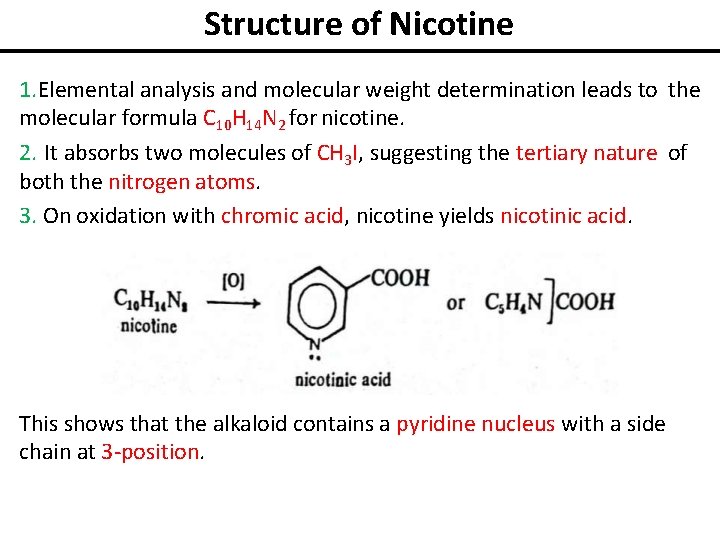
Structure of Nicotine 1. Elemental analysis and molecular weight determination leads to the molecular formula C 10 H 14 N 2 for nicotine. 2. It absorbs two molecules of CH 3 I, suggesting the tertiary nature of both the nitrogen atoms. 3. On oxidation with chromic acid, nicotine yields nicotinic acid. This shows that the alkaloid contains a pyridine nucleus with a side chain at 3 -position.
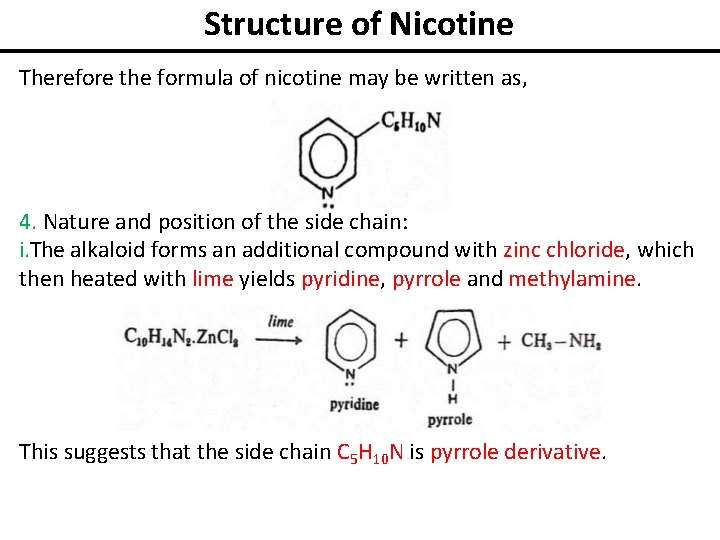
Structure of Nicotine Therefore the formula of nicotine may be written as, 4. Nature and position of the side chain: i. The alkaloid forms an additional compound with zinc chloride, which then heated with lime yields pyridine, pyrrole and methylamine. This suggests that the side chain C 5 H 10 N is pyrrole derivative.
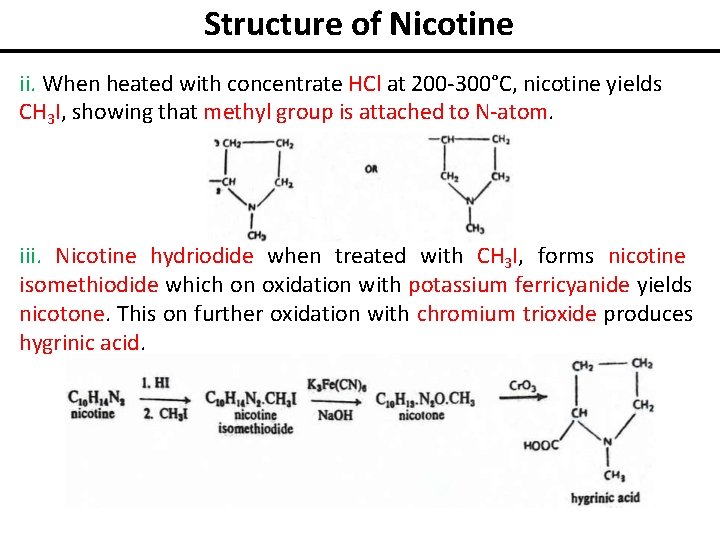
Structure of Nicotine ii. When heated with concentrate HCl at 200 -300°C, nicotine yields CH 3 I, showing that methyl group is attached to N-atom. iii. Nicotine hydriodide when treated with CH 3 I, forms nicotine isomethiodide which on oxidation with potassium ferricyanide yields nicotone. This on further oxidation with chromium trioxide produces hygrinic acid.
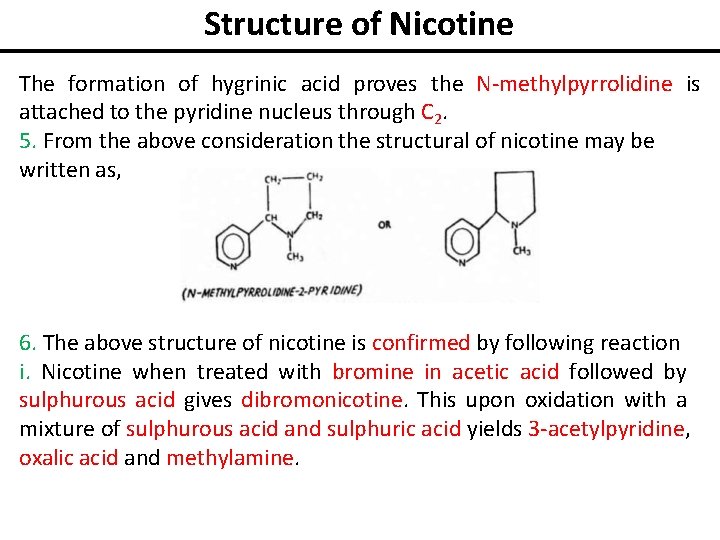
Structure of Nicotine The formation of hygrinic acid proves the N-methylpyrrolidine is attached to the pyridine nucleus through C 2. 5. From the above consideration the structural of nicotine may be written as, 6. The above structure of nicotine is confirmed by following reaction i. Nicotine when treated with bromine in acetic acid followed by sulphurous acid gives dibromonicotine. This upon oxidation with a mixture of sulphurous acid and sulphuric acid yields 3 -acetylpyridine, oxalic acid and methylamine.
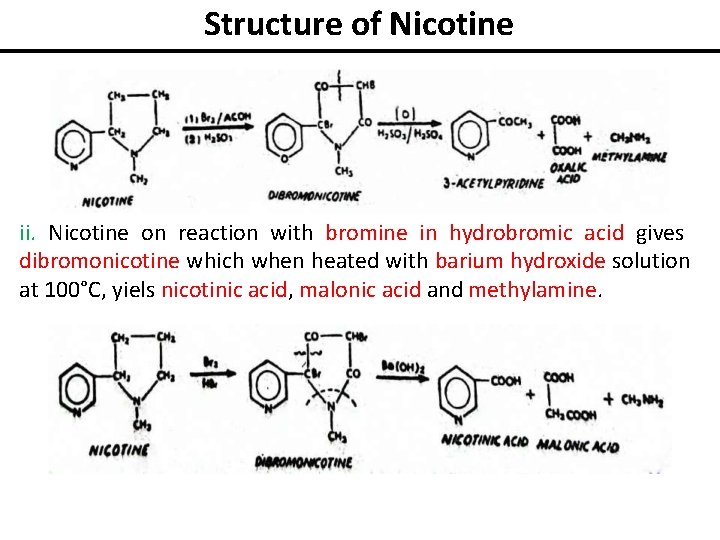
Structure of Nicotine ii. Nicotine on reaction with bromine in hydrobromic acid gives dibromonicotine which when heated with barium hydroxide solution at 100°C, yiels nicotinic acid, malonic acid and methylamine.

Reference BAHL A. and BAHL B. S. Advanced organic chemistry; S. CHAND & COMPANY LTD; ISBN: 81 -219 -0061 -1
- Slides: 11



















