Huntington Disease in the 21 st Century Moderator




![Cognitive Symptoms • Subcortical cognitive impairment[a] – Insidious decline in 21 measures[b] – Dysfunction Cognitive Symptoms • Subcortical cognitive impairment[a] – Insidious decline in 21 measures[b] – Dysfunction](https://slidetodoc.com/presentation_image/0dc534486e4b8b9975facdbea2f0055e/image-7.jpg)
![Psychiatric and Behavioral Symptoms • Often earliest symptoms[a] – Depression, apathy, and other psychiatric Psychiatric and Behavioral Symptoms • Often earliest symptoms[a] – Depression, apathy, and other psychiatric](https://slidetodoc.com/presentation_image/0dc534486e4b8b9975facdbea2f0055e/image-8.jpg)






![Treating Cognitive Symptoms • Neural compensation seen in preclinical stages[a] • No accepted cognitive Treating Cognitive Symptoms • Neural compensation seen in preclinical stages[a] • No accepted cognitive](https://slidetodoc.com/presentation_image/0dc534486e4b8b9975facdbea2f0055e/image-15.jpg)

![Treating Chorea • Treat chorea to – Improve quality of life[a, b] – Prevent Treating Chorea • Treat chorea to – Improve quality of life[a, b] – Prevent](https://slidetodoc.com/presentation_image/0dc534486e4b8b9975facdbea2f0055e/image-17.jpg)
![Pharmacologic Treatment of Chorea Associated With HD • FDA-approved drugs – Tetrabenazine[a] and deutetrabenazine[b] Pharmacologic Treatment of Chorea Associated With HD • FDA-approved drugs – Tetrabenazine[a] and deutetrabenazine[b]](https://slidetodoc.com/presentation_image/0dc534486e4b8b9975facdbea2f0055e/image-18.jpg)
![Tetrabenazine and Deutetrabenazine Parameter Approval Pharmacokinetics[a] Tetrabenazine Deutetrabenazine 2008 2017 P Cmax, ng/m. L Tetrabenazine and Deutetrabenazine Parameter Approval Pharmacokinetics[a] Tetrabenazine Deutetrabenazine 2008 2017 P Cmax, ng/m. L](https://slidetodoc.com/presentation_image/0dc534486e4b8b9975facdbea2f0055e/image-19.jpg)





- Slides: 24

Huntington Disease in the 21 st Century Moderator Victor Sung, MD Associate Professor of Neurology Director, HDSA Center of Excellence University of Alabama at Birmingham, Alabama Panelist Erin Furr-Stimming, MD Associate Professor of Neurology Director, HDSA Center of Excellence University of Texas Health Science Center in Houston, Texas

This program will include a discussion of off-label treatment and investigational agents not approved by the FDA for use in the United States.

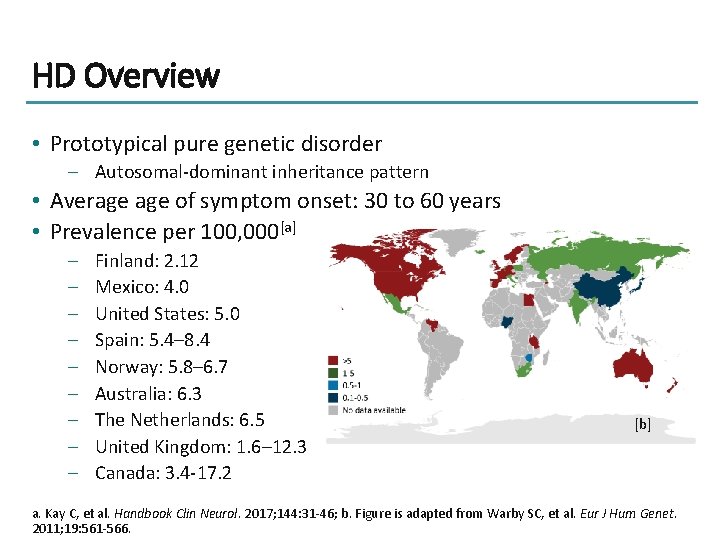
HD Overview • Prototypical pure genetic disorder – Autosomal-dominant inheritance pattern • Average of symptom onset: 30 to 60 years • Prevalence per 100, 000[a] – – – – – Finland: 2. 12 Mexico: 4. 0 United States: 5. 0 Spain: 5. 4– 8. 4 Norway: 5. 8– 6. 7 Australia: 6. 3 The Netherlands: 6. 5 United Kingdom: 1. 6– 12. 3 Canada: 3. 4 -17. 2 [b] a. Kay C, et al. Handbook Clin Neurol. 2017; 144: 31 -46; b. Figure is adapted from Warby SC, et al. Eur J Hum Genet. 2011; 19: 561 -566.

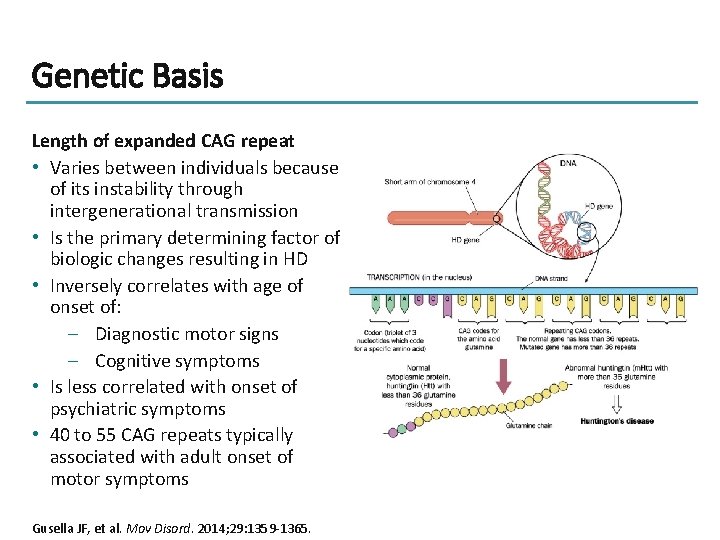
Genetic Basis Length of expanded CAG repeat • Varies between individuals because of its instability through intergenerational transmission • Is the primary determining factor of biologic changes resulting in HD • Inversely correlates with age of onset of: – Diagnostic motor signs – Cognitive symptoms • Is less correlated with onset of psychiatric symptoms • 40 to 55 CAG repeats typically associated with adult onset of motor symptoms Gusella JF, et al. Mov Disord. 2014; 29: 1359 -1365.

Symptom Triad in HD • May have lack of awareness (anosognosia) for the entire triad Motor Cognitive Psychiatric Mc. Cusker E, et al. Tremor Other Hyperkinet Mov (NY). 2014; 4: 257.

Motor Symptoms • Chorea – Abnormal, involuntary, purposeless, relatively random and quick movements – Affects gait, coordination, speech, and swallowing • Impairment of voluntary movements – Impersistence – Incoordination – Difficulty with fine motor skills • Less common – – – Bradykinesia Dystonia Myoclonus Slowing saccades Tics Jankovic J, et al. Mov Disord. 2014; 29: 1414 -1418; Nance M, et al. A Physicians’ Guide to the Management of Huntington’s Disease. ; 2011.
![Cognitive Symptoms Subcortical cognitive impairmenta Insidious decline in 21 measuresb Dysfunction Cognitive Symptoms • Subcortical cognitive impairment[a] – Insidious decline in 21 measures[b] – Dysfunction](https://slidetodoc.com/presentation_image/0dc534486e4b8b9975facdbea2f0055e/image-7.jpg)
Cognitive Symptoms • Subcortical cognitive impairment[a] – Insidious decline in 21 measures[b] – Dysfunction in attention, working memory, verbal learning, long-term memory, and learning of random associations may be earliest manifestations[b] – Memory relatively preserved[c] • Evident prior to motor dysfunction[b] • UHDRS TFC associated with cognitive performance[b] • SDMT can be used to track progression and detect onset of cognitive problems[c] • Cognitive reserve associated with slower rate of change in TMT-B and brain volume loss in prodromal HD[d] a. Papoutsi M, et al. Mov Disord. 2014; 29: 673 -683; b. Paulsen JS, et al. Mov Disord. 2014; 29: 1342 -1350; c. Lemiere J, et al. J Neurol. 2004; 251: 935 -942; d. Bonner-Jackson A, et al. J Int Neuropsychol Soc. 2014; 19: 739 -750.
![Psychiatric and Behavioral Symptoms Often earliest symptomsa Depression apathy and other psychiatric Psychiatric and Behavioral Symptoms • Often earliest symptoms[a] – Depression, apathy, and other psychiatric](https://slidetodoc.com/presentation_image/0dc534486e4b8b9975facdbea2f0055e/image-8.jpg)
Psychiatric and Behavioral Symptoms • Often earliest symptoms[a] – Depression, apathy, and other psychiatric symptoms as prodromal symptoms • Often most disruptive (though treatable) • Can include impulsivity, [b] depression, anxiety, hallucinations, delusion, perseveration, irritability, apathy, and personality change[c] • Combination of untreated depression and impulsivity can result in increased suicide risk[c] a. Martinez-Horta S, et al. Parkinsonism Relat Disord. 2016; 25: 58 -64; b. Johnson PL, et al. J Clin Exp Neuropsychol. 2017; 39: 694 -706; c. Cardoso F. Int Rev Neurobiol. 2017; 134: 1397 -1408.

HD Diagnosis • Based upon – Family history – Clinical presentation with typical motor, behavioral, or cognitive symptoms • Confirmed with definitive genetic testing • Supportive findings on neuroimaging – The presence of caudate atrophy is but not diagnostic in and of itself Pagan F, et al. Handb Clin Neurol. 2017; 144: 63 -67.

Genetic Testing and Counseling • Analysis of CAG repeat length • Clinical situations in which genetic testing is useful – Asymptomatic at-risk individual – Confirmation of suspected HD diagnosis – Prenatal and preimplantation genetic diagnosis • Large increases in CAG repeats more common with paternal transmission CAG Repeat Size Interpretation Alternate Terminology ≤ 26 Normal 27 -35 Normal, unstable Intermediate range 36 -39 Abnormal, reduced penetrance, unstable Indeterminate range ≥ 40 Abnormal, HD Nance MA. Am J Med Genet B Neuropsychiatr Genet. 2017; 174: 75 -92.

Recommended Components of HD Predictive Testing Process 1. Telephone Contact 2. Visit 1 – – – Carry out genetic counseling Document informed consent Assess mental health Perform neurologic examination with patient consent Review potential impact of the results Draw blood 3. Visit 2 – In-person disclosure of results – Make post-results follow-up arrangements 4. Follow-up – Conduct prearranged phone call or in-person visit Genetic Testing Protocol for Huntington’s Disease Society of America. 2016.

General Management Strategies • Identify most bothersome symptoms by interviewing patient and caregiver • Address each bothersome symptom with the most appropriate strategy – Neuro-driven therapy § Physical therapy § Occupational therapy § Speech therapy – Pharmacotherapy – Psychotherapy or counseling – Complementary medicine Nance M, et al. A Physician’s Guide to the Management of Huntington’s Disease. 2011.

Treating Psychiatric Symptoms: Nonpharmacologic Approaches Multidisciplinary Team Nonpharmacologic Approaches • Neuropsychiatrist • Psychologist • Social worker • Identify and alleviate potential external drivers of mood changes • Counseling • Mindfulness training or meditation Education • Patient • Family and caregivers • Community Nance M, et al. A Physician’s Guide to the Management of Huntington’s Disease. 2011.

Treating Psychiatric Symptoms: Pharmacotherapy • Depression – Antidepressants • Hallucinations or delusions – Neuroleptics* • Irritability – SSRIs – Aripiprazole • Labile mood, impulsivity – – Valproate Lamotrigine Topiramate Carbamazepine • Obsessive-compulsive symptoms – SSRIs – Clomipramine *May also have benefit on movements and may cause weight gain and apathy. Nance M, et al. A Physician’s Guide to the Management of Huntington’s Disease. 2011.
![Treating Cognitive Symptoms Neural compensation seen in preclinical stagesa No accepted cognitive Treating Cognitive Symptoms • Neural compensation seen in preclinical stages[a] • No accepted cognitive](https://slidetodoc.com/presentation_image/0dc534486e4b8b9975facdbea2f0055e/image-15.jpg)
Treating Cognitive Symptoms • Neural compensation seen in preclinical stages[a] • No accepted cognitive battery[b] – Mo. CA may be useful • Break tasks into smaller steps or use checklist[c] • Ensure patient participates in activities and uses new information[c] a. Papoutsi M, et al. Mov Disord. 2014; 29: 673 -683; b. Paulsen JS. Curr Neurol Neurosci Rep. 2011; 11: 474 -483; c. Johnson AC, et al. Understanding Behavior in Huntington's Disease: A Guide for Professionals; 2014.

Treating Motor Symptoms • Identify most bothersome symptoms from patient and caregiver perspective • Understand that patients may have anosognosia related to the movements; informant input may be necessary • Assess movements using the UHDRS to establish baseline – Four domains: motor, cognitive, behavioral, function – Motor domain § Includes standardized ratings of oculomotor function, dysarthria, chorea, dystonia, gait, and postural stability § Higher scores indicate more severe motor impairment Huntington Study Group. Mov Disord. 1996; 11: 136 -142.
![Treating Chorea Treat chorea to Improve quality of lifea b Prevent Treating Chorea • Treat chorea to – Improve quality of life[a, b] – Prevent](https://slidetodoc.com/presentation_image/0dc534486e4b8b9975facdbea2f0055e/image-17.jpg)
Treating Chorea • Treat chorea to – Improve quality of life[a, b] – Prevent injuries[b] – Increase daily functioning at work and home[a, b] • Educate patients and caregivers – About purpose of treatment[b] – The impact of chorea on functioning[b]* § Tripping § Swallowing problems § Dropping things *Manifestations of chorea may include these and other indicators a. Jankovic J, et al. Mov Disord. 2014; 29: 1414 -1418; b. Coppen EM, et al. Drugs. 2017; 77: 29 -46.
![Pharmacologic Treatment of Chorea Associated With HD FDAapproved drugs Tetrabenazinea and deutetrabenazineb Pharmacologic Treatment of Chorea Associated With HD • FDA-approved drugs – Tetrabenazine[a] and deutetrabenazine[b]](https://slidetodoc.com/presentation_image/0dc534486e4b8b9975facdbea2f0055e/image-18.jpg)
Pharmacologic Treatment of Chorea Associated With HD • FDA-approved drugs – Tetrabenazine[a] and deutetrabenazine[b] § VMAT-2 inhibitors § Include boxed warning for increased risk of depression and suicidality • Off-label treatments – Neuroleptics[c] – Benzodiazepines[c] – Levitiracetam[d] a. Xenazine® PI 2008; b. Austedo® PI 2017; c. Coppen EM, et al. Drugs. 2017; 77: 29 -46; d. Zesiewicz TA, et al. Mov Disord. 2006; 21: 1998 -2001.
![Tetrabenazine and Deutetrabenazine Parameter Approval Pharmacokineticsa Tetrabenazine Deutetrabenazine 2008 2017 P Cmax ngm L Tetrabenazine and Deutetrabenazine Parameter Approval Pharmacokinetics[a] Tetrabenazine Deutetrabenazine 2008 2017 P Cmax, ng/m. L](https://slidetodoc.com/presentation_image/0dc534486e4b8b9975facdbea2f0055e/image-19.jpg)
Tetrabenazine and Deutetrabenazine Parameter Approval Pharmacokinetics[a] Tetrabenazine Deutetrabenazine 2008 2017 P Cmax, ng/m. L 65. 1 22. 5 Tmax, h 1. 0 2. 3 T 1/2, h 4. 5 9. 4 Dosing regimen 3 X daily 2 x daily Serious AEs* 5. 44 (0. 28, 104. 49) 1. 00 (0. 06, 16. 50) Depression* 11. 15 (0. 62, 200. 33) 0. 65 (0. 10, 4. 10) UHDRS chorea score -3. 5 (-5. 2, -1. 9) -2. 5 (-3. 7, -1. 3) UHDRS total motor score -3. 3 (-7. 0, 0. 3) -4. 0 (-6. 5, -1. 5) Pivotal study results[b] *Odds ratio vs placebo (95% confidence interval) in clinical trials. a. Stamler D, et al. Mov Disord. 2013; 28(Suppl 1): 765; b. Rodrigues FB, et al. Mov Disord. 2017; 4: 582 -585.

Huntingtin-Lowering Therapies HTTRx WVE • Antisense oligonucleotide to reduce the production of huntingtin • Completed enrollment in RDBPC, dose-escalation, Phase 1/2 a, multicenter, 13 -week trial in June 2017[a] – 46 patients with early manifest HD – Intrathecally administered 1 of 5 doses of HTTRx or placebo q 4 weeks • Primary outcome – Safety – Tolerability • Secondary outcome – CSF PK • Antisense oligonucleotides designed to selectively target the m. HTT m. RNA transcript of SNP rs 362307 and SNP rs 362331, respectively • Phase 1 b/2 a RDBPC studies of single and multiple doses of WVE-120101[b] and WVE-120102[c] – 48 patients with early manifest HD in each study – Intrathecally administered 1 of 4 doses or placebo Clinical. Trials. gov. a. NCT 02519036; b. NCT 03225846; c. NCT 03225833 • • Primary outcome – Safety – Tolerability Secondary outcome – Plasma and CSF PK/PD – Clinical effects

Other Investigational Therapies for HD Treatment Phase 2 NCT# Treatment Phase 3 NCT# SRX 246 NCT 02507284 Resveratrol NCT 02336633 Cellavita HD NCT 03252535 Deutetrabenazine NCT 01897896 Laquinimod NCT 02215616 Triheptanoin oil NCT 02453061 VX 15/2503 NCT 02481674 Treatment Phase 4 NCT# Pridopidine Clinical. Trials. gov. NCT 02494778 NCT 01306929 Tetrabenazine NCT 02509793 NCT 01834911 Treatment NCT# GPi-DBS NCT 02535884 Subthalamic DBS NCT 02263430

Clinical Trial Participation • Many new therapies are emerging and require participants for clinical trials – HDSA can assist in matching participants with appropriate trials through HDTrialfinder. org • Participation in a trial – Does not mean that patients will not receive excellent care – Required to ensure new therapies can move forward • Clinicians must address patient concerns and understand why they are not enrolling

Conclusion • HD is an autosomal dominant disease characterized by a CAG repeat – ≥ 40 repeats is positive for HD • Symptoms are motor (especially chorea), cognitive, and psychiatric/behavioral – Can include lack of executive functioning, labile mood, depression, impulsivity, and hallucinations • Management strategies for chorea include FDAapproved VMAT-2 inhibitors

Thank you for participating in this activity. To proceed to the online CME test, click on the Earn Credit link on this page.