Update Treatment and Prophylaxis of Pertussis with Macrolide






















- Slides: 23

Update Treatment and Prophylaxis of Pertussis with Macrolide Antibiotics Tejpratap Tiwari, M. D. , Bacterial Vaccine Preventable Diseases Branch Epidemiology and Surveillance Division National Immunization Program Centers for Disease Control and Prevention National Immunization Conference March 22, 2005

Background: Erythromycin • Accepted antibiotic of choice for pertussis treatment, prophylaxis • Usually recommended as 14 -day course to prevent bacteriologic relapse • Inexpensive

Erythromycin Compliance and Safety • Compliance: ~20% patients discontinue – Gastrointestinal side effects (~30%) – Demanding regimen (3 - 4 daily doses, 14 days) • Safety – Increased risk infantile hypertrophic pyloric stenosis (IHPS), infants <2 weeks old* – Drug-interactions, cytochrome P 450 *Mahon et al. J Pediatr 2001; 139: 380 – 4

Azithromycin and Clarithromycin vs Erythromycin • Good in vitro activity against Bordetella pertussis • More resistant to acid p. H, better absorbed • Greater tissue concentrations, longer plasma half-life • Fewer (1 or 2) daily doses, shorter course (5 days or 7 days)

Azithromycin (A) and Clarithromycin (C) Clinical Studies Author (Year) Study Design Aoyoma et al. 1996) Randomized Control Group Study Arms Historical control group A (8) , C( 9) v E(16, 18) Bace et al. (1999) Noncomparative Safety, two dose regime A (17) v A (20) Bace et al. (2000)* Randomized Erythromycin A(9) v E(15) Pichichero (2003) Noncomparative Historical studies Erythromycin group A (34) Langely et al. (2004) Randomized Lebel et al. (2001) *Abstract Randomized A( 58) v E (57) C (31) vs E (23)

Treatment for Pertussis by Age-group Age group Erythromycin Clarithromycin Azithromycin >6 mth 40 -50 mg/kg/d in 4 divided doses (max 2 gm/d) X 14 d 15 mg/kg/day in 2 divided doses (max 500 mg/dose) X 7 d 10 mg/kg/day (max 500 mg) single dose on day 1 then 5 mg/kg/day (max 250 mg) on day 2 – 5 1 -5 mth As above 10 mg/kg/day, single daily dose X 5 d <1 mth Use as alternate drug in (Safety data doses above (Risk of unavailable) IHPS) Preferred drug Dose as above

Summary • Macrolides, preferred antimicrobial agents – Erythromycin, clarithromycin, or azithromycin are appropriate first-line agents for persons age >1 month – Azithromycin, preferred choice for infants <1 month of age • Use TMX-SMZ as alternate agent • Consider safety, evaluate concurrent medications for potential interactions, adherence to the prescribed regimen, and cost

Acknowledgements • CDC/NIP – Colleagues at Bacterial VPD Branch, Epidemiology and Surveillance Division • AAP/COID • AAFP • HICPAC

Thank You

RED BOOK 2003: Pertussis Treatment • “The drug of choice is erythromycin estolate (40 – 50 mg per day, orally in 4 divided doses; maximum 2 g/day. The recommended duration of therapy to prevent relapse is 14 days. Studies have documented that the newer macrolides, azithromycin dihydrate (10– 12 mg kg per day, orally, in 1 dose for 5 days; maximum 600 mg day) or clarithromycin (15– 20 mg/kg per day, orally, in 2 divided doses; maximum 1 g/day for 7 days), may be as effective as erythromycin and have fewer adverse effects and better compliance. ”

EXTRA SLIDES

HICPAC Guidelines, 2004 Pertussis Treatment and Prophylaxis • Adults – Erythromycin estolate, 500 mg, 4 times/day – Erythromycin delayed-release tablet, 333 mg, 3 times daily, • Children age >2 weeks, – Erythromycin 40– 50 mg/kg day for 14 days • Infants aged <2 weeks, or adults intolerant to EE – Azithromycin 10– 12 mg/kg/day x 5– 7 days – Azithromycin 10 mg/kg on Day 1 then 5 mg/kg/day x 4 days (single dose) – Clarithromycin for 10– 14 days 15– 20 mg/kg/day in two divided doses

Control of Communicable Diseases, 2004 Pertussis Treatment and Prophylaxis • Treatment – Minimum 7 -day course of macrolide antibiotics • Chemoprophylaxis (selective) – A 7 -day course of erythromycin, clarithromycin or azithromycin

Langley et al. , 2004 * Azithromycin vs Erythromycin • Multi-center, randomized, equivalence trial • Suspect pertussis cases (n=477), aged 6 months – 16 years – Azithromycin (n=238) • 10 mg/kg single dose day 1; then 5 mg/kg/day single daily dose days 2– 5 – Erythromycin estolate (n=239) • 40 mg/kg/day, 3 doses/day x 10 days *Langley et al. Pediatrics 2004; 114: 96 – 101

Langley et al. , 2004* Safety Cohort (N=477) • Rate of gastrointestinal adverse events – Azithromycin group (n=238): 18% – Erythromycin group (n=239): 40% • Compliance with 100% doses – Azithromycin, 90% – Erythromycin, 55% *Langley et al. Pediatrics 2004; 114: 96 – 101

Efficacy Cohort* Culture-confirmed pertussis (N=114) • Bacterial eradication at end of treatment – Azithromycin (days 5 – 7) • (n=53), 100% (95% CI: 93– 100) – Erythromycin (days 10 – 12) • (n=53), 100% (95% CI: 93 – 100) • Recurrence, 1 week post-treatment – Azithromycin (n=51), 0% (95% CI: 0– 7) – Erythromycin (n=53), 0% (95% CI: 0– 6. 7) *Langley et al. Pediatrics 2004; 114: 96 – 101

Lebel et al. , 2001* Clarithromycin vs Erythromycin • • Randomized, prospective, single-blind Compared safety, efficacy Suspect pertussis (n=153) Ages 1 month – 16 years – Erythromycin estolate, 40 mg/kg/day (max 1 gm daily), 3 doses/day X 14 days (n=77) – Clarithromycin, 15 mg/kg/day (max 1 gm daily), 2 doses/day X 7 days (n=76) *Lebel et al. PIDJ 2001; 20: 1149 – 54.

Lebel et al. , 2001* Safety Cohort (N=153) • Rates of gastrointestinal adverse events – Clarithromycin (n=76): 32% – Erythromycin (n=77): 44% • Compliance (mean % of drug taken) – Clarithromycin, 98. 5% – Erythromycin, 88. 6% *Lebel et al. PIDJ 2001; 20: 1149 – 54.

Efficacy Cohort Culture-confirmed pertussis (N=62) • Post-treatment bacterial eradication similar – Clarithromycin (days 8– 10 ), (31/31) – Erythromycin (days 15– 18), (22/23) • Study limitation – Sample size insufficient to demonstrate equivalence Lebel et al. PIDJ, 2001; 20: 1149 – 54.

Azithromycin, Clarithromycin Safety in Infants Age <6 months • Not labeled for use in infants <6 months age* – No published report IHPS or severe adverse events in neonates *http: //pdrel. thomsonhc. com/

Macrolides Safety: FDA Pregnancy Label • Erythromycin, azithromycin (Category B) – Animal studies, no adverse effect on fetus – No clinical trials in pregnant women • Clarithromycin (Category C) – Animal studies, adverse effects on fetus – No clinical trials in pregnancy http: //pdrel. thomsonhc. com/

HIPAC Guidelines Hypersensitivity to Macrolides • Adults: – Trimethoprim (TMP) –sulfamethoxazole (SXT) 1 double-strength tablet, twice daily X 14 days • Children: – 8 mg/kg/day TMP, 40 mg/kg/day SXT, 2 divided doses • Exception: – Pregnant woman at term – Nursing mother – Infant aged <2 months)

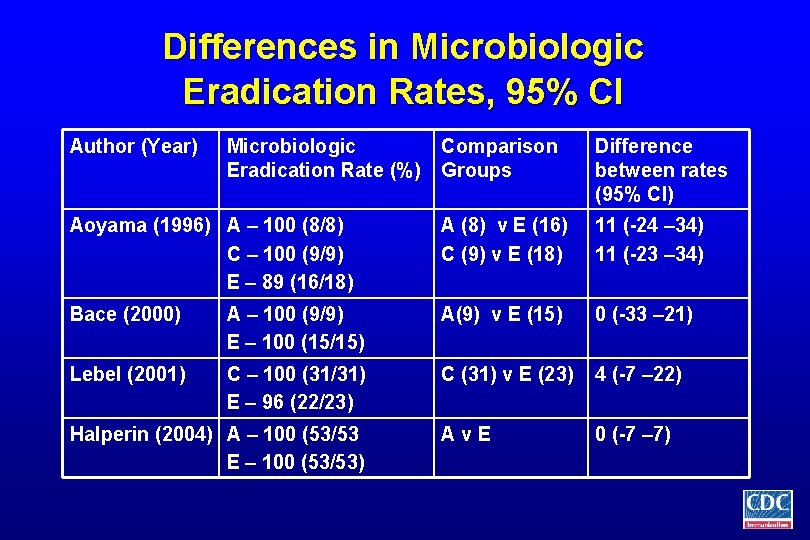
Differences in Microbiologic Eradication Rates, 95% CI Author (Year) Microbiologic Eradication Rate (%) Comparison Groups Difference between rates (95% CI) Aoyama (1996) A – 100 (8/8) C – 100 (9/9) E – 89 (16/18) A (8) v E (16) C (9) v E (18) 11 (-24 – 34) 11 (-23 – 34) Bace (2000) A – 100 (9/9) E – 100 (15/15) A(9) v E (15) 0 (-33 – 21) Lebel (2001) C – 100 (31/31) E – 96 (22/23) C (31) v E (23) 4 (-7 – 22) Av. E 0 (-7 – 7) Halperin (2004) A – 100 (53/53 E – 100 (53/53)