Scientific Discovery A mystery story Curiosity and inquisitiveness




































- Slides: 37

Scientific Discovery ( A mystery story) • Curiosity and inquisitiveness of how natural phenomena occur • Observe and interpret • Imagination – Hypothesize meaning of observations – Create models and theories • Test models and theories: experiments – Modify and retest September 8, 2009 Physics I Lesson 1 Dr J. Tison 1

Refine models and theories • Accepted theories – often proven incorrect by new evidence – create new or improved theories • Example… – Ptolemy (ca 100 AD) taught the earth is center of solar system – Copernicus, then Galileo (ca 1500 AD) determined that the sun is the center of the solar system. September 8, 2009 Physics I Lesson 1 Dr J. Tison 2

Symmetry in the universe? • A scientist looks for… – Symmetry or commonality – Applies accepted theories, hypotheses, or models. • Example… – Newton’s Law of Gravitational Force (2 masses): • Fg = Gm 1 m 2/r 2 – Coulomb’s Law of Electrostatic Force (2 charges): • Fe = Kq 1 q 2/r 2 September 8, 2009 Physics I Lesson 1 Dr J. Tison 3

Expand the range of theories • Newton’s law of motion – “classical” description of bodies in motion • Einstein’s theories of relativity – “modern” physics extends to the wider range of phenomena, in the universe Newton’s Laws of Motion are used to describe most natural phenomena that are observed in normal everyday situations. September 8, 2009 Physics I Lesson 1 Dr J. Tison 4

Who’s on first? Models, Theories, Laws & Principles • Model: Visual concept of what is observed • Theory: Detailed description with measurable results to test model • Law (e. g. Newton’s): Concise, general statements about how nature behaves • Principle: (e. g. Archimedes) is a less general statement than a law September 8, 2009 Physics I Lesson 1 Dr J. Tison 5

Measurement and Uncertainty Lord Kelvin (1824 -1907): • “… when you can measure what you are speaking about, and express it in numbers, you know something about it; • … when you cannot express it in numbers, your knowledge is of a meager and unsatisfactory kind; • … it may be the beginning of knowledge, but you have scarcely, in your thoughts, advanced the stage of Science, whatever the matter may be” September 8, 2009 Physics I Lesson 1 Dr J. Tison 6

Measurements • Some examples… – Distance, speed (and velocity) – Temperature – Time – Weight – And, many others. September 8, 2009 Physics I Lesson 1 Dr J. Tison 7

Measurement and Calculations • Examples. . . – Speed = (distance)/(time) e. g. mph, ft/sec, etc • 2 measurements: (1) distance; (2) time – Momentum = (mass)∙(velocity) = (mass)∙(distance)/time • 3 measurements: (1) mass; (2) distance; (3) time September 8, 2009 Physics I Lesson 1 Dr J. Tison 8

Undefinable Quantities • Some physical quantities… – length, time, mass, temperature, are fundamental and are not calculated from others – International committees (e. g. SI) determine the “standard” for measuring undefinable quantities September 8, 2009 Physics I Lesson 1 Dr J. Tison 9

SI Base units: Length: meter (m) – Length of the path traveled by light in a vacuum, – During the time interval of 1/299 792 458 of a second Note: Speed of light is 299, 792, 458 m/s September 8, 2009 Physics I Lesson 1 Dr J. Tison 10

SI Base units: Mass: kilogram (kg) – Mass of a platinum-iridium cylinder • (Bureau International des Poids et Mesures (BIPM), Sèvres, Paris (1 st CGPM (1889), CR 3438). – Only base unit with a prefix (kg); • Gram is derived unit, equal to 1/1000 of a kilogram; – Only unit still defined by a physical prototype • (instead of a measurable natural phenomenon ) September 8, 2009 Physics I Lesson 1 Dr J. Tison 11

SI Base units: Time : second (s) • Duration of exactly 9, 192, 631, 770 periods of the radiation corresponding to • Transition between two hyperfine levels of the ground state of the cesium-133 atom – at a temperature of 0º K September 8, 2009 Physics I Lesson 1 Dr J. Tison 12

SI Base Unit: Temperature, Kelvin (ºK) • Thermodynamic temperature, ºK – Absolute temperature – Fraction 1/273. 16 (exactly) of thermodynamic temperature at the triple point* of water *Triple Point of Water: water, ice, and water vapour coexisting in a stable equilibrium : 0. 01 °C; 6. 03 x 10 -3 atm) September 8, 2009 Physics I Lesson 1 Dr J. Tison 13

SI Base Unit: Ampere (A) • Ampere: Constant current maintained in – Two straight parallel conductors of • Infinite length and • Negligible cross-section, and • Placed 1 meter apart in a vacuum, – Produces a force between these conductors = 2× 10 -7 n/m September 8, 2009 Physics I Lesson 1 Dr J. Tison 14

Systems of Units • mks – SI – French Academy of Sciences – meter, kilogram, sec • cgs – centimeter, gram, sec • British – foot, lb, sec September 8, 2009 Physics I Lesson 1 Dr J. Tison 15

Conversion between Units Speed = distance/time – 1 m/s = (1 m/s)(100 cm/1 m) = 100 cm/s – 1 m/s = (1 m/s) (1 in/2. 54 cm)(100 cm/1 m) = 3. 94 in/s – 1 m/s = (1 m/s) (1 in/2. 54 cm)(100 cm/1 m (60 s/min)(60 min/h) = 14. 2 x 103 in/h September 8, 2009 Physics I Lesson 1 Dr J. Tison 16

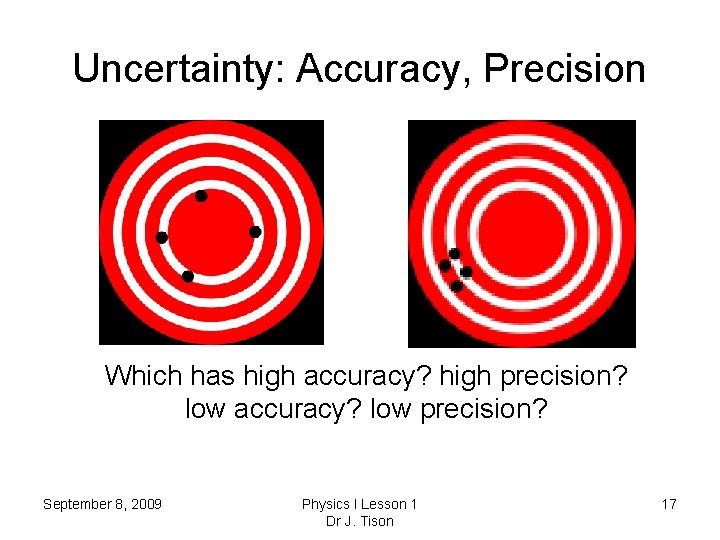
Uncertainty: Accuracy, Precision Which has high accuracy? high precision? low accuracy? low precision? September 8, 2009 Physics I Lesson 1 Dr J. Tison 17

X Y • X = average value – sometimes written as= <x> • Y = calculated difference from <x> – Y is measure of “precision” – % = y/x ∙ 100 • Multiple measurements as in a “Gage R&R” – 5 or 10 measurements. – Static or dynamic – 3 days September 8, 2009 Physics I Lesson 1 Dr J. Tison 18

All Measurements have Uncertainty • Random Errors: statistical – Repeatability and reproducibility – Unpredicable fluctuations in temperature or line voltage – Mechanical vibrations of an experimental setup – Unbiased estimates of measurements by observer • Systematic Errors: determinate – Measurement device: – Instrument calibration – Personal error, e. g. parallax reading of a gage September 8, 2009 Physics I Lesson 1 Dr J. Tison 19

How to… • Obtain greater accuracy – Minimize systematic errors • Obtain greater precision – Minimize random errors September 8, 2009 Physics I Lesson 1 Dr J. Tison 20

Metrology System Specification September 8, 2009 Physics I Lesson 1 Dr J. Tison 21

Hip Replacement Metrology September 8, 2009 Physics I Lesson 1 Dr J. Tison 22

Hip Joint Metrology Result September 8, 2009 Physics I Lesson 1 Dr J. Tison 23

Camshaft Metrology September 8, 2009 Physics I Lesson 1 Dr J. Tison 24

Gears September 8, 2009 Physics I Lesson 1 Dr J. Tison 25

Significant Figures • Number of directly read + one estimated number – All non-zero digits – Leading zeros not significant – Zeros (a) between non-zero, and (b) at end, if measured, are significant – Least count: • Smallest subdivision on the measurement scale • “Meter stick is calibrated in cm with a millimeter least count September 8, 2009 Physics I Lesson 1 Dr J. Tison 26

Significant Figures: Example • 12. 3 cm – # significant figures = 3 • 0. 0123 cm – # significant figures = 3 • 12. 03 cm – # significant figures = 4 • 12. 30 cm – # significant figures = 3 or 4? September 8, 2009 Physics I Lesson 1 Dr J. Tison 27

Significant Figures in Calculations • Calculating with Significant Figures – Use the least # significant figures – 12. 3 x 1. 2 = 14. 76 = 15 • there are 2 s. f. in 1. 2 – 12. 3 x 1. 20 = 14. 76 = 14. 8 • There are 3 s. f. in 1. 20 and 12 September 8, 2009 Physics I Lesson 1 Dr J. Tison 28

Significant Figures in Calculations • Calculating with Significant Figures – Use the least # significant figures – 12. 3 x 1. 2 = 14. 76 = 15 • there are 2 s. f. in 1. 2 – 12. 3 x 1. 20 = 14. 76 = 14. 8 • There are 3 s. f. in 1. 20 and 12 September 8, 2009 Physics I Lesson 1 Dr J. Tison 29

Scientific Notation • Powers of 10 – Usually expressed as 1 digit to left of decimal point – 1, 234. 0 = 1. 234 ∙ 103 – Alternative notation • Doesn’t require superscripts (simplifies typing) • 1, 234. 0 = 1. 234 E 3 • Scientific calculator display September 8, 2009 Physics I Lesson 1 Dr J. Tison 30

Scientific Notation Speed of light: – 186, 282 mi/s → 1. 86282 ∙ 105 mi/s – 299, 792, 458 m/s → 2. 99792458 ∙ 108 m/s Gravitational constant – Standard format: 0. 00000667300 m 3 kg 1 s-2 – Scientific notation: 6. 67300 ∙ 10 -11 m 3 kg-1 s-2 – September 8, 2009 Physics I Lesson 1 Dr J. Tison 31

Computations in Powers of 10 • Adding/subtracting – All values must be have same “power of ten” • Multiplying/dividing – Write each number in scientific notation, i. e. x. yz ∙ 10 a – Multiply the numbers as standard numbers – Add/subtract the exponents for the “power of ten term” • 12. 3 x 123. 0 = • (1. 23 ∙ 101) x (1. 23 ∙ 102) = • 5. 129 ∙ 103 = 5. 129 E 3 September 8, 2009 Physics I Lesson 1 Dr J. Tison 32

Powers of Ten Prefixes 10 -24 to 1024 • See names Table 1 -4 • Most common – 10 -12 pico – 109 giga – 10 -9 nano– 103 kilo – 106 mega September 8, 2009 Physics I Lesson 1 Dr J. Tison 33

Order of Magnitude Estimate value to approx. “Power of Ten” – Order of Magnitude (OM) of 100 is 102 – ≈ is mathematical operator meaning “roughly” September 8, 2009 Physics I Lesson 1 Dr J. Tison 34

Dimensions, Dimensional Analysis Convenient way to • Verify proper mathematical relationships • Check correctness of calculations • Make sure all terms to be added or subtracted have same ‘units of measure’ September 8, 2009 Physics I Lesson 1 Dr J. Tison 35

Lesson 1: Summary • Scientific Discovery – Models, Theories, Laws, Principles • Physical Quantities – Undefinables, standards, SI • Mass, Length, Time, Temperature, etc – Definables • Velocity, Acceleration, Force, Momentum, etc – Systems of Units • MKS • CGS • British September 8, 2009 Physics I Lesson 1 Dr J. Tison 36

Lesson 1: Summary • Measurements – Accuracy and Precision – Uncertainty • Random and Statistical Errors • Determinant and indeterminate • % Uncertainty – Significant Figures • Calculators and reported values • Calculations – Scientific Notation • Powers of 10 – Order of Magnitude (OM) – Dimensional Analysis September 8, 2009 Physics I Lesson 1 Dr J. Tison 37