Rate Laws Rate Laws n Chemists conduct experiments







![NO (g) + ½ Cl 2 (g) NOCl (g) Exp’t [NO]0 [Cl 2]0 Initial NO (g) + ½ Cl 2 (g) NOCl (g) Exp’t [NO]0 [Cl 2]0 Initial](https://slidetodoc.com/presentation_image_h2/475e4c2dca55b8bbafb232c06f4f58e1/image-9.jpg)

![Or, take Ratio of Rates Rate 2 = k[NH 4+]n[NO 2 -]m = k[0. Or, take Ratio of Rates Rate 2 = k[NH 4+]n[NO 2 -]m = k[0.](https://slidetodoc.com/presentation_image_h2/475e4c2dca55b8bbafb232c06f4f58e1/image-12.jpg)


- Slides: 14

Rate Laws

Rate Laws n Chemists conduct experiments to determine the rate law for a chemical reaction. n. A RATE LAW is a mathematical equation that shows how the reaction rate is related to the concentration of the reactants.

Rate Constant (k) n From collision theory, you know that one way to speed up a reaction is to increase reactant concentrations. n For two reactants, a rate law has this form: Rate = k [reactant A]m [reactant B]n n The term k is the rate constant – it is unique to each reaction; changes with catalyst – determined experimentally at specific temperature

Order of Reaction exponents m and n indicate the order of each reactant in the reaction. n The n For example, an exponent of 1 means the reaction is first order with respect to the given reactant. – Higher order, greater effect on reaction rate. sum of m + n gives the order of the overall reaction. n The

Reaction Order n 2 NO (g) n + O 2 (g) 2 NO 2 (g) Has rate equation: rate = k[NO]1[O 2]1 – Describe reaction – Describe reactants

Reaction Order n 2 NO (g) n + Cl 2 (g) 2 NOCl (g) Has rate equation: rate = k[NO]2[Cl 2] – Describe reaction – Describe reactants

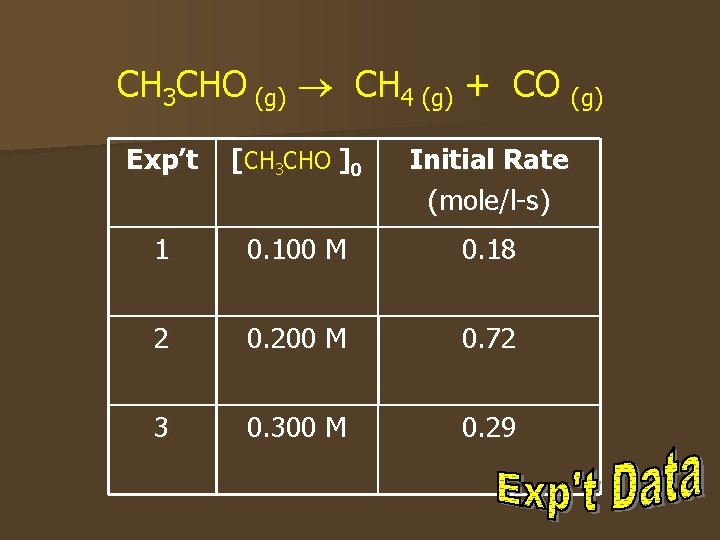
CH 3 CHO (g) CH 4 (g) + CO (g) Exp’t [CH 3 CHO ]0 Initial Rate (mole/l-s) 1 0. 100 M 0. 18 2 0. 200 M 0. 72 3 0. 300 M 0. 29

Rate Law n Using the data from the decomposition of acetaldehyde, CH 3 CHO, the order fo the reaction is determined in the following way: Rate = [CH 3 CHO]m determine the value of m, write a ratio of the rate expressions for two different concentrations. n To
![NO g ½ Cl 2 g NOCl g Expt NO0 Cl 20 Initial NO (g) + ½ Cl 2 (g) NOCl (g) Exp’t [NO]0 [Cl 2]0 Initial](https://slidetodoc.com/presentation_image_h2/475e4c2dca55b8bbafb232c06f4f58e1/image-9.jpg)
NO (g) + ½ Cl 2 (g) NOCl (g) Exp’t [NO]0 [Cl 2]0 Initial Rate (moles/L) (moles/L-s) 1 0. 250 1. 43 X 10 -6 2 0. 250 0. 500 2. 86 X 10 -6 3 0. 500 11. 4 X 10 -6

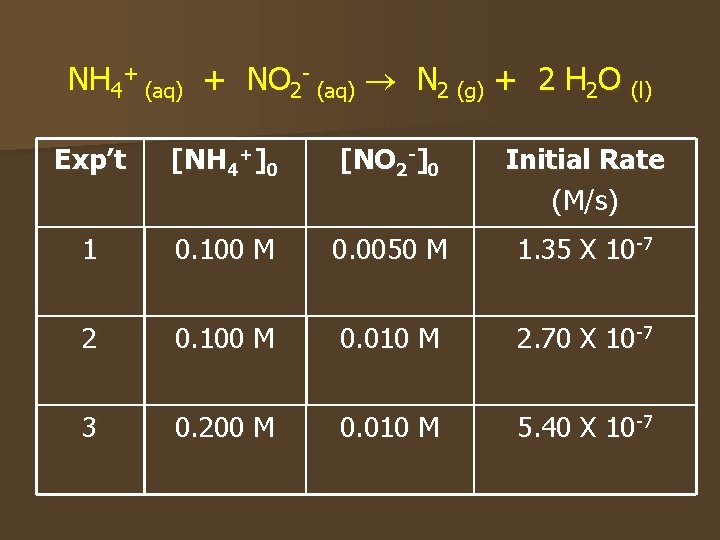
NH 4+ (aq) + NO 2 - (aq) N 2 (g) + 2 H 2 O (l) Exp’t [NH 4+]0 [NO 2 -]0 Initial Rate (M/s) 1 0. 100 M 0. 0050 M 1. 35 X 10 -7 2 0. 100 M 0. 010 M 2. 70 X 10 -7 3 0. 200 M 0. 010 M 5. 40 X 10 -7

Compare how the rate changes with concentration to find m, n n Compare expt’s 1 & 2 – [NH 4+] is constant, doubled [NO 2 -] – The rate doubled n Compare expt’s 2 & 3 – [NO 2 -] is constant, doubled [NH 4+] – The rate doubled
![Or take Ratio of Rates Rate 2 kNH 4nNO 2 m k0 Or, take Ratio of Rates Rate 2 = k[NH 4+]n[NO 2 -]m = k[0.](https://slidetodoc.com/presentation_image_h2/475e4c2dca55b8bbafb232c06f4f58e1/image-12.jpg)
Or, take Ratio of Rates Rate 2 = k[NH 4+]n[NO 2 -]m = k[0. 100]n[0. 010]m Rate 1 = k[NH 4+]n[NO 2 -]m = k[0. 100]n[0. 005]m 2. 70 X 10 -7 = [0. 010]m 1. 35 X 10 -7 = [0. 005]m 2. 00 = (2. 0)m m=1

Write a Rate Law 1. 2. 3. 4. Refer to balanced equation for the rate-determining step. Write a rate constant, k. Write the formula of each reactant in brackets. Write the equation coefficient of each reactant as an exponent (only if a single event).

Example: Write a Rate Law Step 1: NO + Cl 2 NOCl 2 Step 2: NOCl 2 + NO 2 NOCl (fast) (slow) n Determine the overall balanced equation. n What is the intermediate? n Which step is rate-determining? n What is the rate law for the rate-determining step? n What is the order of the reaction with respect to NO and Cl 2?